Full Papers
gave pure product as an orange solid (1.60 g, a quantitative yield).
Purity: 99.07%.
pure product as a yellow oil (1.40 g, a quantitative yield). Purity:
98.18%.
1
2
3
4
5
6
7
8
9
3
3
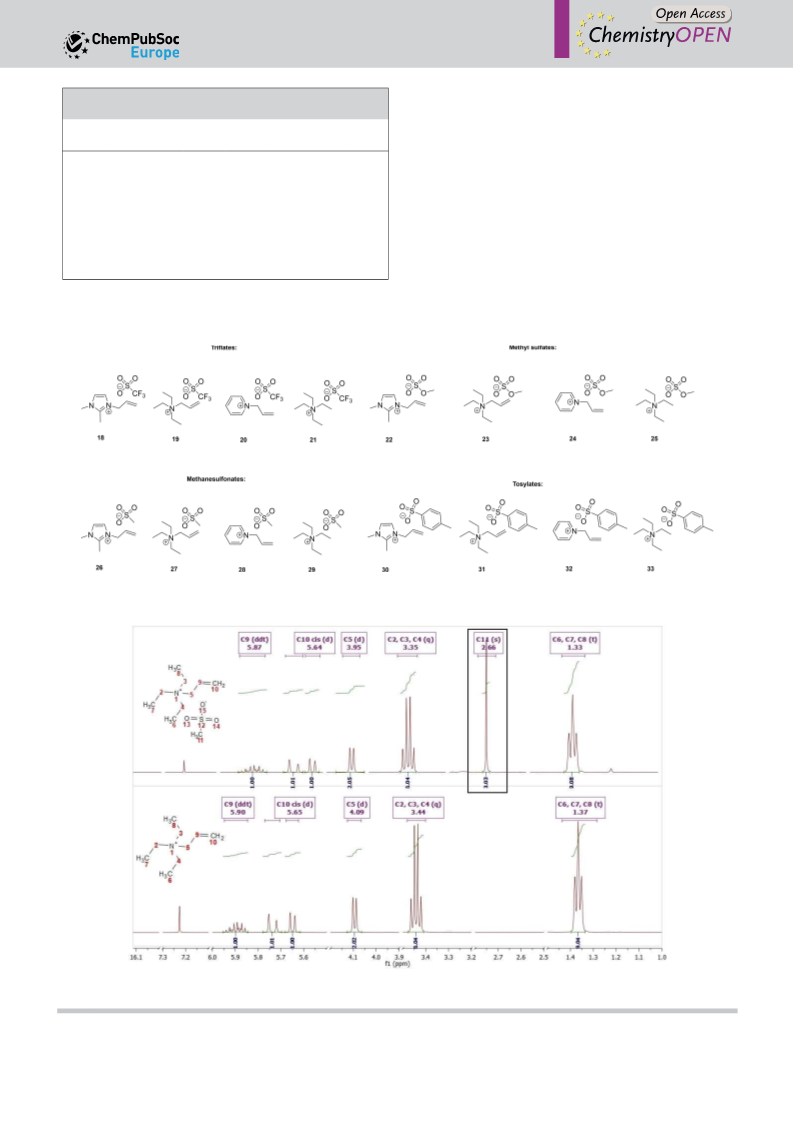
1H NMR (500 MHz, CDCl3): δ=7.27 (d, J=2.1 Hz, 1H), 7.19 (d, J=
1H NMR (400 MHz, CDCl3): δ=5.94 (ddt, 3J=17.1 Hz, 3J=10.0 Hz,
2.2 Hz, 1H), 5.86 (dd, 3J=17.1 Hz, 3J=10.3 Hz, 1H), 5.34 (dt, 3J=
3J=7.2 Hz, 1H), 5.76 (dd, J=16.8 Hz, J=0.9, Hz, 1H), 5.69 (d, J=
3
2
3
2
3
2
3
10.4 Hz, J=1.5 Hz, 1H), 5.20 (dt, J=17.1 Hz, J=1.7, Hz, 1H), 4.67
(dt, 3J=5.8 Hz, 2J=1.5 Hz, 2H), 3.78 (s, 3H), 2.56 (s, 3H); 13C {1H} NMR
(100 MHz, DMSO-d6): δ=145.03, 131.94, 122.92, 121.48, 121.17 (q,
1JCF =322.8 Hz), 119.27, 50.07, 35.19, 9.59; 19F NMR (376 MHz, CDCl3):
δ=-78.50; IR (ATR, νmax, cmÀ 1): 3142 (w), 3119 (w), 1650 (w), 1592
(w), 1544 (w), 1460 (w), 1415 (w), 1254 (vs), 1224 (s), 1150 (s), 1027
(s), 992 (w), 917 (m), 775 (m), 677 (w); MS (ESI)+: m/z (%)=137
10.2 Hz, 1H), 3.95 (d, 3J=7.2 Hz, 2H), 3.70 (s, 3H), 3.37 (q, J=7.3 Hz,
6H), 1.38 (t, 3J=7.3 Hz, 9H); 13C {1H} NMR (100 MHz, CDCl3): δ=
128.53, 124.03, 59.43, 54.58, 52.86, 7.52; IR (ATR, νmax, cmÀ 1): 3182
(s), 3141 (vs), 3090 (m), 3057 (s), 3020 (s), 2985 (vs), 2947 (s), 2164
(m), 1591 (m), 1559 (m), 1520 (m), 1477 (s), 1429 (m), 1397 (s), 1379
(s); MS (ESI)+: m/z (%)=142 ([M]+, 100); HRMS (ESI)+ m/z: Calcd for
C9H20N+ 142.1596; Found 142.1597, À 0.7034; MS (ESI) : m/z (%)=
À
~
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
+
([M]+, 100); HRMS (ESI)+ m/z: Calcd for C8H13N2 137.1079; Found
111([M+], 100), 96 ([MÀ CH3]+, 27); HRMS (ESI)À m/z: Calcd for
~
137.1078,
0.7294; MS (ESI)À : m/z (%)=149 ([M+], 100); HRMS
CH3O4SÀ 110.9752; Found 110.9756, Δ À 3.6044; ICÀ (min): tR =3.31.
(ESI)À m/z: Calcd for CF3O3SÀ 148.9520; Found 148.9520, Δ 0; ICÀ
(min): tR =12.11.
Allyltriethylammonium Triflate (19)
Compound was obtained according to general procedure C with
use of allyltriethylammonium chloride (2.00 g, 11.30 mmol), methyl
triflate (1.85 g, 11.30 mmol) and MeCN (10 mL). The reaction gave
pure product as a yellow solid (3.30 g, a quantitative yield). Purity:
96.40%.
1-Allyl-2,3-Dimethylimidazolium Tosylate (30)
Compound was obtained according to general procedure D with
use of 1-allyl-2,3-dimethylimidazolium chloride (1.00 g, 6.31 mmol),
methyl tosylate (1.18 g, 6.31 mmol) and MeCN (15 mL). The reaction
gave pure product as a beige solid (1.75 g, a quantitative yield).
Purity: 98.18%.
1H NMR (600 MHz, CDCl3): δ=5.91 (ddt, 3J=17.3 Hz, 3J=10.0 Hz,
3
3
3
3J=7.3 Hz, 1H), 5.74 (dd, J=21.7 Hz, J=5.4 Hz, 2H), 3.87 (d, J=
3
3
3
3
1H NMR (500 MHz, CDCl3): δ=7.60 (d, J=8.1 Hz, 2H), 7.45 (d, J=
7.3 Hz, 2H), 3.32 (q, J=7.3 Hz, 6H), 1.36 (t, J=7.3 Hz, 9H); 13C {1H}
2.1 Hz, 1H), 7.28 (d, 3J=2.1 Hz, 1H), 7.03 (d, 3J=7.9 Hz, 2H), 5.79
(ddt, J=16.1 Hz, J=10.3 Hz, J=5.8 Hz, 1H), 5.23 (d, J=10.3 Hz,
NMR (100 MHz, CDCl3): δ=128.97, 123.59, 120.75 (q, JCF
=
1
3
3
3
3
322.10 Hz), 59.58, 52.95, 7.49; 19F NMR (376 MHz, CDCl3): δ=-78.44;
IR (ATR, νmax, cmÀ 1): 2999 (w), 1459 (w), 1258 (vs), 1224 (s), 1150 (s),
1029 (s), 950 (w), 800 (w), 636 (m), 572 (m), 516 (s); MS (ESI)+: m/z
(%)=142 ([M]+, 100); HRMS (ESI)+ m/z: Calcd for C9H20N+ 142.1596;
~
3
3
2
1H), 5.10 (d, J=17.1 Hz, 1H), 4.68 (dt, J=5.7 Hz, J=1.4 Hz, 2H),
3.76 (s, 3H), 2.53 (s, 3H), 2.24 (s, 3H); 13C {1H} NMR (100 MHz, DMSO-
d6): δ=146.19, 145.01, 138.1, 132.03, 128.52, 125.92, 122.93, 121.49,
119.27, 50.05, 35.19, 21.25, 9.62; IR (ATR, νmax, cmÀ 1): 3130 (s), 3100
(s), 3086 (s), 3016 (w), 1645 (w), 1587 (m), 1539 (m), 1493 (w), 1457
(m), 1417 (m), 1366 (w), 1282 (m), 1243 (m), 1206 (s), 1186 (vs), 1118
( s), 1030 (m), 1009 (m), 995 (s), 938 (m), 816 (s), 785 (s), 763 (m),
Found 142.1597,
À 0.7034; MS (ESI)À : m/z (%)=149 ([M+], 100);
HRMS (ESI)À m/z: Calcd for CF3O3SÀ 148.9520; Found 148.9521, Δ
À 0.6714; ICÀ (min): tR =12.62.
679 (s), 640 (m); MS (ESI)+: m/z (%)=137 ([M]+, 100); HRMS (ESI)+
+
~
Allyltriethylammonium Tosylate (31)
m/z: Calcd for C8H13N2 137.1079; Found 137.1078,
0.7294; MS
(ESI)À : m/z (%)=171 ([M+], 100); HRMS (ESI)À m/z: Calcd for
C7H7O3SÀ 171.0116; Found 171.0118, Δ À 1.1695; ICÀ (min): tR =4.88.
Compound was obtained according to general procedure D with
use of allyltriethylammonium chloride (2.00 g, 11.30 mmol), methyl
tosylate (2.10 g, 11.30 mmol) and MeCN (15 mL). The reaction gave
pure product as a yellow solid (3.50 g, a quantitative yield). Purity:
98.16%.
35 1-Allyl-2,3-Dimethylimidazolium Methanesulfonate (26)
36
37
Compound was obtained according to general procedure E with
use of 1-allyl-2,3-dimethylimidazolium chloride (1.00 g, 6.31 mmol),
methyl methanesulfonate (0.70 g, 6.31 mmol) and MeCN (10 mL).
3
3
1H NMR (600 MHz, CDCl3): δ=7.68 (d, J=8.1 Hz, 2H), 7.05 (d, J=
3
3
3
8.0 Hz, 2H), 5.77 (ddt, J=17.2 Hz, J=10.0 Hz, J=7.2 Hz, 1H), 5.60
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54
55
56
57
3
2
3
(dd, J=16.8 Hz, J=0.5 Hz, 1H), 5.53 (d, J=10.2 Hz, 1H), 3.81 (d,
3J=7.2 Hz, 2H), 3.21 (q, 3J=7.3 Hz, 6H), 2.25 (s, 3H), 1.20 (t, 3J=
7.3 Hz, 9H); 13C {1H} NMR (100 MHz, CDCl3): δ=144.52, 138.86,
128.43, 128.30, 125.89, 124.06, 59.33, 52.75, 21.20, 7.54; IR (ATR,
νmax, cmÀ 1): 2985 (m), 2946 (w), 1642 (w), 1598 (w), 1478 (m), 1458
(m), 1397 (w), 1300 (m), 1192 (vs), 1119 (s), 1030 (s), 1010 (s), 817
(m), 797 (m); MS (ESI)+: m/z (%)=142 ([M]+, 100); HRMS (ESI)+ m/Àz:
The reaction gave pure product as
quantitative yield). Purity: 98.30%.
a
yellow oil (1.75 g, a
3
3
1H NMR (500 MHz, CDCl3): δ=7.55 (d, J=2.1 Hz, 1H), 7.40 (d, J=
3
3
3
2.1 Hz, 1H), 5.89 (ddd, J=16.2 Hz, J=10.9 Hz, J=5.8 Hz, 1H), 5.31
(d, 3J=10.3 Hz, 1H), 5.19 (d, 3J=17.1 Hz, 1H), 4.80 (d, 3J=5.7 Hz,
2H), 3.87 (s, 3H), 2.64 (s, 3H), 2.57 (s, 3H); 13C {1H} NMR (100 MHz,
CDCl3): δ=144.2, 130.19, 122.88, 121.34, 120.25, 50.61, 39.43, 35.38,
9.83; IR (ATR, νmax, cmÀ 1): 3444 (w), 3133 (m), 3010 (w), 2933 (w),
1647 (w), 1588 (m), 1538 (m), 1459 (w), 1419 (m), 1332 (w), 1177
(vs), 1037 (vs), 946 (m), 766 (s), 676 (w); MS (ESI)+: m/z (%)=137
Calcd for C9H20N+ 142.1596; Found 142.1597, À 0.7034; MS (ESI) :
~
m/z (%)=171 ([M+], 100); HRMS (ESI)À m/z: Calcd for C7H7O3SÀ
171.0116; Found 171.0107, Δ 5.2628; ICÀ (min): tR =4.76.
+
([M]+, 100); HRMS (ESI)+ m/Àz: Calcd for C8H13N2 137.1079; Found
+
À
~
Allyltriethylammonium Methanesulfonate (27)
137.1078, 0.7294; MS (ESI) : m/z (%)=95 ([M ], 100); HRMS (ESI)
m/z: Calcd for CH3O3SÀ 94.9803; Found 94.9807, Δ À 4.2114; ICÀ
Compound was obtained according to general procedure E with
use of allyltriethylammonium chloride (2.00 g, 11.30 mmol), methyl
methanesulfonate (1.25 g, 11.30 mmol) and MeCN (10 mL). The
reaction gave pure product as a yellow solid (2.65 g, a quantitative
yield). Purity: 96.13%.
(min): tR =1.76.
Allyltriethylammonium Methyl Sulfate (23)
Compound was obtained according to general procedure B with
use of allyltriethylammonium chloride (1.00 g, 4.51 mmol), dimethyl
sulfate (0.57 g, 4.51 mmol) and MeCN (10 mL). The reaction gave
1H NMR (500 MHz, CDCl3): δ=5.87 (ddt, 3J=17.2 Hz, 3J=10.0 Hz,
3
3
3J=7.2 Hz, 1H), 5.71 (d, J=16.8 Hz, 1H), 5.64 (d, J=10.1 Hz, 1H),
ChemistryOpen 2019, 8, 972–983
980
© 2019 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Szpecht, Andrea
Szpecht, Andrea
 Zajac, Adrian
Zajac, Adrian
 Zielinski, Dawid
Zielinski, Dawid
 Maciejewski, Hieronim
Maciejewski, Hieronim
 Smiglak, Marcin
Smiglak, Marcin