8
02
S. M. Firestine et al. / Bioorg. Med. Chem. 17 (2009) 794–803
0
þ
4
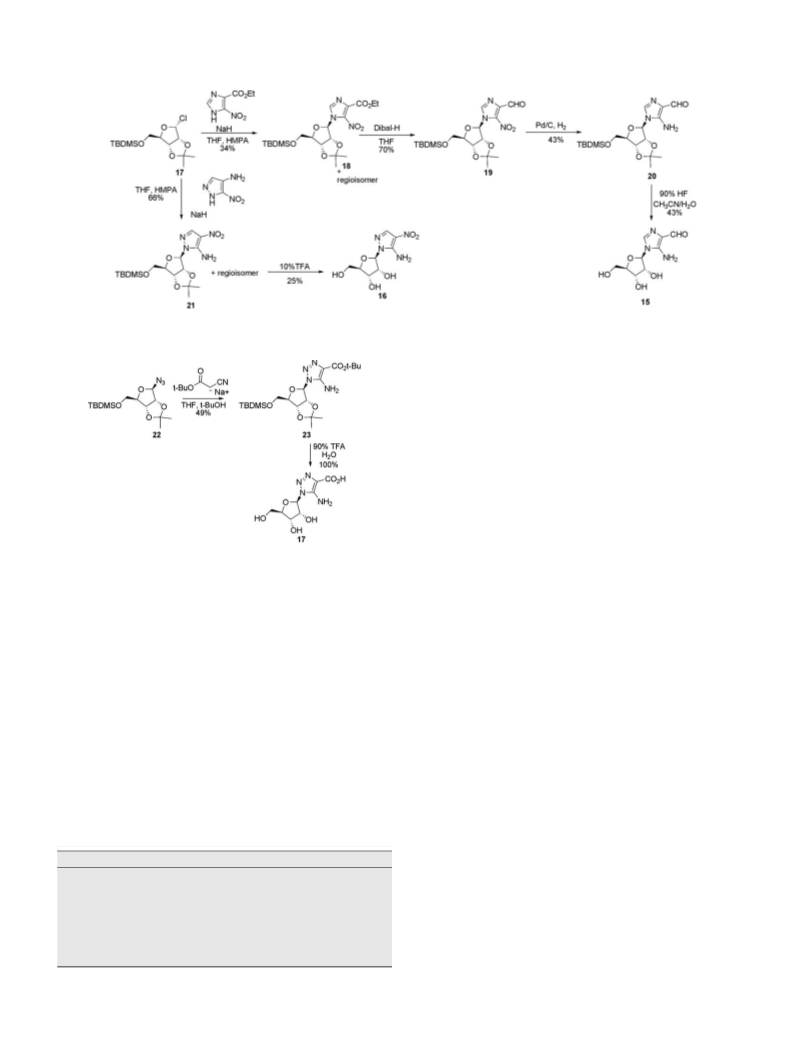
.12. 4-Nitro-1-(-b-
D
-ribofuranosyl)pyrazole 5 -phosphate (7)
0.94; HRMS (FABM+NH4 calcd for C
8 17 5 9
H N O P 358.0764, found
3
68.0760.
Compound 13 (35 mg, 0.14 mmol) was phosphorylated in
.0 mL of triethyl phosphate with POCl (0.13 mL, 1.4 mmol) for
h and purified by Q-sepharose anion exchange chromatography
1
3
3
4.16. Testing for inhibition against AIR carboxylase
to give the desired product (42 mg, 85%). UV (kmax in 0.1 N HCl)
All compounds were analyzed as inhibitors as previously de-
1
2
02, 273 nm; H NMR (300 MHz, D
2
O) d 8.84 (1H, s, C3), 8.23
scribed. Briefly, the inhibitor (10 nM to 100
cuvette containing 50 mM Tris-HCl, 0.5 mM EDTA, pH 8.0 and CAIR
(10–100 M). The reaction was initiated by the addition of 23–
46 ng of enzyme and the change in absorbance at 260 nm was
monitored. The K value was determined from a series of 1/v versus
lM) was added to a
0
(
1H, s, C5), 5.81 (1H, d, J = 4.2 Hz, C1 ), 4.56 (1H,
w
t, J = 5.1 Hz,
0
0
C2 ), 4.34 (1H,
C4 ), 3.96 (2H, m, C5 ); C NMR (75 MHz, D
w
t, J = 5.2 Hz, C3 ), 4.24 (1H, dd, J = 4.2 and 4.6 Hz,
l
0
0
13
2
O) d 140.0, 132.8,
9
6.6, 87.87.0 (d, Jc–p = 7.6 Hz), 77.3, 73.0, 66.8 (d, Jc–p = 3.4 Hz);
i
3
1
P NMR (121 MHz, D
2
O) 0.61; HRMS (FAB): calcd for C
8
11
H N
4
O
9
P
1/S plots using the program Enzyme Kinetics (Trinity Software). For
each compound, three different inhibitor concentrations were used
based upon preliminary experiments to determine the IC50 value.
ꢀ
3
24.0233 [MꢀH] , found 324.0263.
0
4
.13. 5-Amino-1-(b-D-ribofuranosyl)pyrazole-4-carboxylate 5 -
phosphate (8)
4.17. Molecular electrostatic potential calculations
Compound 14 (248 mg, 0.86 mmol) was phosphorylated in
5 mL of triethyl phosphate with POCl (403 L, 4.33 mmol) for
.5 h and was purified by boronate affinity gel column to give a
white solid (249 mg, 72%). 65.6 mg of the ethyl ester (0.163 mmol)
was dissolved in 10 mL of water containing NaOH (110 mg,
The geometries of each compound were determined by minimi-
zation using GAUSSIAN 03 with the B3LyP 6-31G basis set. The elec-
*
1
1
3
l
trostatic potential was calculated from the wave functions
generated for the minimized structure using the CUBE option from
Gaussian. The color map for each compound was set to the same
range of charges (ꢀ0.07 to 0.07 in gas phase; ꢀ0.1 to 0.1 for water).
The range was established to maximize contrast between the
compounds.
2
.75 mmol). The reaction was heated to reflux for 4 h, cooled to
room temperature and then diluted to 50 mL with water. The solu-
ꢀ
tion was applied to a Q Sepharose column (1.5 ꢁ 13 cm, HCO ),
3
the column washed with water and then eluted with a gradient
ꢀ1
4 3
of 0–500 mM NH HCO over 30 min at a flow rate of 4.0 mL min .
Supplementary data
The eluent, monitored at 260 nm, was collected in fractions and the
appropriate fractions were combined and dried by lyophilization
(
3ꢁ). The solid was dissolved in water (10 mL) and the solution
þ
passed through Chelex (NH ) to remove metal contaminants
4
1
(
5
59 mg, 93%). TLC; H NMR (300 MHz, D
2
O) d 7.51 (1H, s, C3),
References and notes
0
0
.56 (1H, d, J = 6.7 Hz, C1 ), 4.60 (1H, dd, J = 5.5 and 6.7 Hz, C2 ),
.25 (1H, dd, J = 3.3 and 5.5 Hz, C3 ), 4.08 (1H, m, C4 ), 3.76 (2H,
0
0
4
1
.
Meyer, E.; Leonard, N. J.; Bhat, B.; Stubbe, J.; Smith, J. M. Biochemistry 1992, 31,
5022–5032.
2. Firestine, S. M.; Davisson, V. J. Biochemistry 1994, 33, 11917–11926.
0
13
m, C5 ); C NMR (75 MHz, D
2
O) d 169.3, 152.9, 143.5, 98.1, 66.3,
0
0
0
0
0
7
1.6 (C5 ), 73.2 (C2 or C3 ), 84.7 (C2 or C3 ), 90.1 (JC, P = 8 Hz,
3
.
Firestine, S. M.; Misialek, S.; Toffaletti, D. L.; Klem, T. J.; Perfect, J. R.; Davisson,
V. J. Arch. Biochem. Biophys. 1998, 351, 123–134.
31
C4’), 98.1 (C4), 143.5 (C3), 152.9 (C5), 169.3 (C@O); P NMR
(
3
121 MHz,
40.0546 [M+H] , found 340.0545.
D
2
O)
d
0.38; HRMS (ESI) calcd for
C
9
H
15
N
3
O
9
P
4. Firestine, S. M.; Poon, S. W.; Mueller, E. J.; Stubbe, J.; Davisson, V. J. Biochemistry
1994, 33, 11927–11934.
+
5.
Mueller, E. J.; Meyer, E.; Rudolph, J.; Davisson, V. J.; Stubbe, J. Biochemistry 1994,
3, 2269–2278.
3
0
4
.14. 5-Amino-1-(b-
D
-ribofuranosyl)-4-nitropyrazole 5 -
6. Hoskins, A. A.; Morar, M.; Kappock, T. J.; Mathews, A.; Zaugg, J. B.; Barder, T. E.;
Peng, P.; Okamoto, A.; Ealick, S. E.; Stubbe, J. Biochemistry 2007, 46, 2842–2855.
phosphate (9)
7.
Constantine, C. Z.; Starks, C. M.; Mill, C. P.; Ransome, A. E.; Karpowicz, S. J.;
Francois, J. A.; Goodman, R. A.; Kappock, T. J. Biochemistry 2006, 45, 8193–8208.
8. Firestine, S. M.; Davisson, V. J. J. Med. Chem. 1993, 36, 3484–3486.
Bhat, B.; Groziak, M. P.; Leonard, N. J. J. Am. Chem. Soc. 1990, 112, 4891–4897.
0. Jordan, F. Nat. Prod. Rep. 2003, 20, 184–201.
1. Miller, B. G.; Wolfenden, R. Annu. Rev. Biochem. 2002, 71, 847–885.
12. Blazevic, N.; Kajfez, F.; Sunjic, V. J. Heterocycl. Chem. 1970, 7, 227–229.
13. Li, S. X.; Tong, Y. P.; Xie, X. C.; Wang, Q. H.; Zhou, H. N.; Han, Y.; Zhang, Z. Y.;
Gao, W.; Li, S. G.; Zhang, X. C.; Bi, R. C. J. Mol. Biol. 2007, 366, 1603–1614.
Compound 16 was phosphorylated in 2.0 mL OP(OEt)
3
with
9
1
1
.
POCl3 (154 L, 1.65 mmol) for 4 h and was purified on a Q Sephar-
l
ose column to give a white solid (53 mg, 85%). TLC (cellulose, 7:3
CH
8
3
CN/100 mM NH
.36 (1 H, s, C3), 6.10 (1H, d, J = 5.8 Hz, C1 ), 5.07 (1H, dd,
4
HCO
3
) R
f
0.32; 1H NMR (300 MHz, D
2
O) d
0
0
0
J = 5.8 Hz, J = 5.5 Hz, C2 ), 4.74 (1H, dd, J = 3.9 Hz, J = 5.5, C3 ), 4.60
1
4. Mathews, I. I.; Kappock, T. J.; Stubbe, J.; Ealick, S. E. Structure 1999, 7, 1395–
0
0
13
(
(
(
(
1H, m, C4 ), 4.32 (2H, m, C5 ); C NMR (75 MHz, D
2
O) d 64.2
1406.
0
0
0
0
0
J
J
C,P = 4.3 Hz, C5 ), 69.9 (C2 or C3 ), 71.3 (C2 or C3 ), 83.4
15. Chavis, C.; Grodenic, F.; Imbach, J. L. Eur. J. Med. Chem. 1979, 14, 123–131.
16. Harki, D. A.; Graci, J. D.; Korneeva, V. S.; Ghosh, S. K. B.; Hong, Z.; Cameron, C. E.;
Peterson, B. R. Biochemistry 2002, 41, 9026–9033.
7. Barascut, J. L.; Tamby, C.; Imbach, J. L. J. Carbohydr. Nucleosides, Nucleotides
1974, 1, 77–84.
8. Sanghvi, Y. S.; Larson, S. B.; Robins, R. K.; Revankar, G. R. J. Chem. Soc., Perkin
Trans 1990, 11, 2943–2950.
19. Rosemeyer, H.; Seela, F. Helv. Chim. Acta 1988, 71, 1573–1785.
0
31
C,P = 8.6 Hz, C4 ), 118.3 (C4), 136.5 (C3), 147.0 (C5); P NMR
O) 1.23; HRMS (ESI): calcd for
41.0498 [M+H] , found 341.0500.
121 MHz,
D
2
d
8 14 4 9
C H N O P
1
+
3
1
4
.15. 5-Amino-1-(b-d-ribofuranosyl)-1,2,3-triazole-4-
0
carboxylate 5 -phosphate (10)
20. Kissman, H. M.; Child, R. G.; Weiss, M. J. J. Am. Chem. Soc. 1957, 79, 1185–1188.
2
2
2
2
1. Acevedo, O. L.; Krawczyk, S. H.; Townsend, L. B. J. Org. Chem. 1986, 51, 1050–
058.
1
Compound 17 (19.7 mg, 0.071 mmol) was phosphaorylated in
.0 mL of OP(OEt) with POCl (14 L, 0.15 mmol) for 6 h and
2. Wamhoff, H. In Comprehensive Heterocyclic Chemistry; Katritzky, A. R., Rees, C.
W., Eds.; Pergamon Press: Oxford, 1984; pp 669–732.
3. Camarasa, M. J.; Alonso, R.; De Las Heras, F. G. Carbohydr. Res. 1980, 83, 152–
2
3
3
l
was purified first on a Q Sepharose column then on a semi-pre-
156.
parative PRP-1 column to give the desired product (2.7 mg, 20%).
4. Yoshikawa, M.; Kato, T.; Takenishi, T. Tetrahedron Lett. 1967, 50, 5065–5068.
25. Yoshikawa, M.; Kato, T. Bull. Chem. Soc. Jpn. 1967, 40, 2849–2853.
26. Yoshikawa, M.; Kato, T.; Takenishi, T. Bull. Chem. Soc. Jpn. 1969, 42, 3505–3508.
7. Wu, W.; Bergstrom, D. E.; Davisson, V. J. J. Org. Chem. 2003, 68, 3860–3865.
8. Acevedo, O.; Jorgensen, W. L. J. Org. Chem. 2006, 71, 4896–4902.
1
0
H NMR (300 MHz, D
2
O) d 5.81 (1H, d, J = 5.9 Hz, C1 ), 4.73 (1H,
0
0
dd, J = 5.3 and 5.9 Hz, C2 ), 4.36 (1H, dd, J = 3.8 and 5.3 Hz, C3 ),
.21 (1H, m, C4 ), 3.91 (2H, m, C5 ); P NMR (121 MHz, D O) d
2
2
2
0
0
31
4

 Firestine, Steven M.
Firestine, Steven M.
 Wu, Weidong
Wu, Weidong
 Youn, Hasik
Youn, Hasik
 Jo Davisson
Jo Davisson