Journal of the American Chemical Society
Page 4 of 5
University and the Max Planck Society are greatly appre-
ciated.
1
2
3
4
5
6
7
8
9
1
1
1
1
1
1
1
1
1
1
2
2
2
2
2
2
2
2
2
2
3
3
3
3
3
3
3
3
3
3
4
4
4
4
4
4
4
4
4
4
5
5
5
5
5
5
5
5
5
5
6
REFERENCES
(1)Rinaldi, R.; Schuth, F. Energy Environ. Sci 2009, 2, 610.
(2)Beaumont, S. K. J. Chem. Technol. Biotechnol. 2012, 87, 595.
(
3)Thomas, J. M.; Johnson, B. F. G.; Raja, R.; Sankar, G.; Midgley, P.
A. Acc. Chem. Res. 2003, 36, 20.
4)Wang, Y.; Yao, J.; Li, H.; Su, D.; Antonietti, M. J. Am. Chem. Soc.
011, 133, 2362.
(5)Robin J. White; Rafael Luque; Vitaliy L. Budarin; Clark, J. H.;
Macquarrie, D. J. Chem. Soc. Rev. 2009, 38, 481.
(
2
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
(
(
6)Martono, E.; Vohs, J. M. J. Catal. 2012, 291, 79.
7)Bond, J. Q.; Alonso, D. M.; Wang, D.; West, R. M.; Dumesic, J. A.
Figure 4. Products distribution at different temperatures.
Reaction condition: vanillin 1000 mg, S/C=350, water 80 mL,
Science 2010, 327, 1110.
8)Peng, B. X.; Yuan, X. G.; Zhao, C.; Lercher, J. A. J. Am. Chem. Soc.
(
H
2
pressure 1.0 MPa, 1h.
2012, 134, 9400.
(9)Enache, D. I.; Edwards, J. K.; Landon, P.; Solsona-Espriu, B.;
Carley, A. F.; Herzing, A. A.; Watanabe, M.; CKiely, C. J.; Knight, D.
W.; Hutchings, G. J. Science 2006, 311, 362.
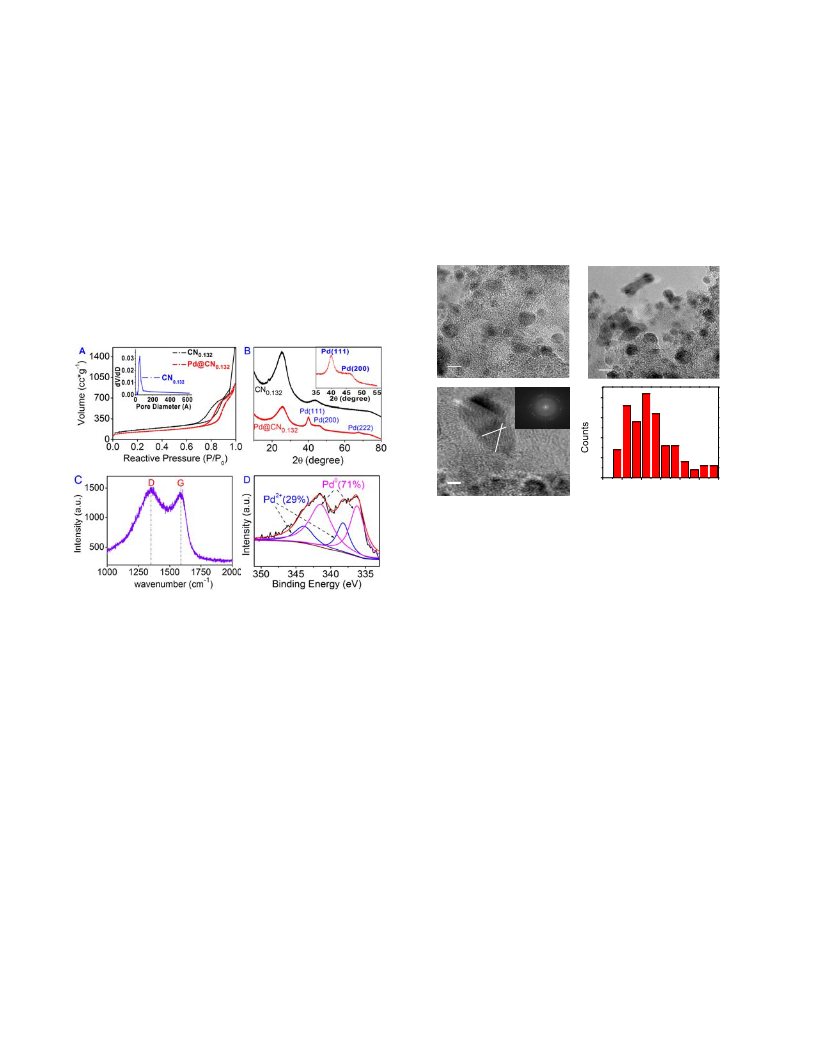
Furthermore, the catalyst can easily be separated from the
reaction solution by simple filtration. The catalyst is highly
stable and can be reused for several cycles without losing
activity, which is a prerequisite for practical applications.
(Table S3). The concentration of Pd in reaction solution was
determined by ICP-AES, and it was below 0.1 ppm, meaning
the leaching of the Pd in solvent is neglectable. Since nitro-
gen is strong electronegative and usually has a lone pair of
electrons, the nitrogen functionalities on the surface might
act as Lewis base sites and are expected to be more effective
in retaining metal nanoparticles.
(
(
10)Zhang, C.; Lercher, J. A. Angew. Chem., Int. Ed. 2012, 51, 5935.
11)Deng, W. P.; Liu, M.; Tan, X. S.; Zhang, Q. H.; Wang, Y. J. Catal.
2010, 271, 22.
(12)Zhao, C.; Kou, Y.; Lemonidou, A. A.; Li, X. B.; Lercher, J. A.
Angew. Chem., Int. Ed. 2009, 48, 3987.
(
13)Clippel, F.; Dusselier, M.; Van Rompaey, R.; Vanelderen, P.;
Dijkmans, J.; Makshina, E.; Giebeler, L.; Oswald, S.; Baron, G. V.;
Denayer, J. F. M.; Pescarmona, P. P.; Jacobs, P. A.; Sels, B. F. J. Am.
Chem. Soc. 2012, 134, 10089.
(14)Crossley, S.; Faria, J.; Shen, M.; Resasco, D. E. Science 2010, 327,
68.
In conclusion, Pd nanoparticles have been heterogeneously
supported on mesoporous N-doped carbon with its special
base properties and electronic behavior. The novel Pd cata-
lyst exhibits a high activity for the hydrodeoxygenation of
vanillin, a common component in lignin-derived bio-oil, un-
der mild reaction conditions and using water as a clean sol-
vent. The high catalytic performance has been attributed to
the special structure of the catalytic N-doped carbon-metal
heterojunction, which leads not only to a very stable and
uniform dispersion of Pd nanoparticles but also to additional
electronic activation of the metal nanoparticles and well dis-
persion of catalyst in water. This Pd@CN0.132 catalyst hold
promising potential for the biofuel upgrade and further work
will be directed toward the applications to natural lignin and
other model systems.
(
15)Radkevich, V. Z.; Senko, T. L.; Wilson, K.; Grishenko, L. M.;
Zaderko, A. N.; Diyuk, V. Y. Appl. Catal., A 2008, 335, 241.
16)Yang, S. B.; Feng, X. L.; Wang, X. C.; Mullen, K. Angew. Chem.,
(
Int. Ed. 2011, 50, 5339.
(17)Lee, J. S.; Wang, X. Q.; Luo, H. M.; Baker, G. A.; Dai, S. J. Am.
Chem. Soc. 2009, 131, 4596.
(
18)Yang, W.; Fellinger, T. P.; Antonietti, M. J. Am. Chem. Soc. 2011,
133, 206.
(19)Fulvio, P. F.; Lee, J. S.; Mayes, R. T.; Wang, X. Q.; Mahurin, S.
M.; Dai, S. Phys. Chem. Chem. Phys. 2011, 13, 13486.
(20)Liu, R.; Mahurin, S. M.; Li, C.; Unocic, R. R.; Idrobo, J. C.; Gao,
H. J.; Pennycook, S. J.; Dai, S. Angew. Chem., Int. Ed. 2011, 50, 6799.
(
21)Zhao, L.; Fan, L. Z.; Zhou, M. Q.; Guan, H.; Qiao, S. Y.;
Antonietti, M.; Titirici, M. Adv. Mater. 2010, 22, 5202.
22)Paraknowitsch, J. P.; Zhang, J.; Su, D. S.; Thomas, A.;
(
Antonietti, M. Adv. Mater. 2010, 22, 87.
(23)Wang, X. Q.; Dai, S. Angew. Chem., Int. Ed. 2010, 49, 6664.
(24)Lee, J. S.; Wang, X. Q.; Luo, H. M.; Dai, S. Adv. Mater. 2010, 22,
ASSOCIATED CONTENT
Supporting Information
1
004.
(
25)Yuan, J. Y.; Giordano, C.; Antonietti, M. Chem. Mater. 2010, 22,
5
003.
Experimental and characterization sections, Scheme S1 and
S2, Figures S1-6, Tables S1-4. This material is available free of
charge via the Internet at http://pubs.acs.org.
(
26)Vispute, T. P.; Zhang, H. Y.; Sanna, A.; Xiao, R.; Huber, G. W.
Science 2010, 330, 1222.
(27)Huber, G. W.; Iborra, S.; Corma, A. Chem. Rev. 2006, 106, 4044.
(
28)Zakzeski, J.; Bruijnincx, P. C. A.; Jongerius, A. L.; Weckhuysen,
B. M. Chem. Rev. 2010, 110, 3552.
29)Mohan, D.; Pittman, C. U., Jr.; Steele, P. H. Energy Fuels 2006,
0, 848.
30)Nimmanwudipong, T.; Runnebaum, R. C.; Block, D. E.; Gates,
B. C. Energy Fuels 2011, 25, 3417.
31)Nimmanwudipong, T.; Runnebaum, R. C.; Ebeler, S. E.; Block,
AUTHOR INFORMATION
(
2
Corresponding Author
(
(
ACKNOWLEDGMENT
D. E.; Gates, B. C. Catal. Lett. 2012, 142, 151.
(32)Yan, N.; Yuan, Y.; Dykeman, R.; Kou, Y.; Dyson, P. J. Angew.
Chem., Int. Ed. 2010, 49, 5549.
Financial support from the Joint Petroleum and Petrochemi-
cal Funds of the National Natural Science Foundation of Chi-
na and China National Petroleum Corporation (U1162124), the
Fundamental Reasearch Funds for the Central Universities,
the Program for Zhejiang Leading Team of S&T Innovation
(
33)Prochazkova, D.; Zamostny, P.; Bejblova, M.; Cerveny, L.; Cejka,
J. Appl. Catal., A 2007, 332, 56.
(
2011R50007), and the Partner Group Program of the Zhejiang
4
ACS Paragon Plus Environment

 Xu, Xuan
Xu, Xuan
 Li, Yi
Li, Yi
 Gong, Yutong
Gong, Yutong
 Zhang, Pengfei
Zhang, Pengfei
 Li, Haoran
Li, Haoran
 Wang, Yong
Wang, Yong