Notes
J . Org. Chem., Vol. 62, No. 23, 1997 8253
(HPLC, SiO2, 2:2:1 CHCl3/n-hexane/MeOH) to afford compounds
4 (37 mg, 37%), 5 (44 mg, 44%), and 6 (7.7 mg, 7.7%). In the
same manner, compound 3 (100 mg) gave 4 (32 mg, 32%), 5 (36
mg, 36%), and 6 (7.6 mg, 7.6%).
values being 0.017, 0.92, 1.9, 4.0, 15, 6.0, 6.2, and 1.5
µg/mL, respectively. Both N-oxides 2 and 3, being
regarded as the conformationally locked analogues of 1,
showed weaker activity compared with that of 1, which
has a nitrogen lone pair. Thus, the nitrogen lone pair
on the cephalotaxine skeleton appears to be essential for
its activity. Also, the weaker activity of analogues 4-8
compared with that of 1 is accounted for by the changes
in the topology of the cephalotaxine skeleton through
chemical modification.
1-[(1R,4S,5S)-3-Meth oxy-9,11,21-tr ioxa-17-azapen tacyclo-
[15.3.1.01,5.06,14.08,12]h en icosa -2,6,8(12),13-t et r a en -4-yl] 4-
m eth yl (2R)-2-h yd r oxy-2-(4-h yd r oxy-4-m eth ylp en tyl)su c-
cin a te (4): a colorless oil; [R]D ) -209˚ (c 0.55, MeOH); UV
(MeOH) λmax 292 nm (log ꢀ 3.70); IR (film) 3516 br, 1743, 1648,
1
1507, 1490 cm-1; H NMR (CDCl3) δ 1.16 (s, 3H), 1.17 (s, 3H),
1.37 (m, 2H), 1.44 (m, 2H), 1.56 (m, 1H), 1.68 (m, 2H), 1.79 (m,
2H), 2.04 (td, J ) 12.8, 6.0 Hz, 1H), 2.46 (ddd, J ) 14.0, 12.8,
2.0 Hz, 1H), 2.54 (d, J ) 16.5 Hz, 1H), 2.56 (ddd, J ) 13.5, 10.8,
4.0 Hz, 1H), 2.70 (br dd, J ) 17.5, 9.0 Hz, 1H), 2.77 (d, J ) 16.5
Hz, 1H), 2.84 (ddd, J ) 13.5, 10.8, 2.0 Hz, 1H), 2.89 (m, 1H),
3.32 (d, J ) 7.2 Hz, 1H), 3.45 (s, 3H), 3.64 (m, 1H), 3.65 (s, 3H),
4.97 (s, 1H), 5.78 (d, J ) 7.2 Hz, 1H), 5.89 (d, J ) 1.5 Hz, 1H),
5.90 (d, J ) 1.5 Hz, 1H), 6.59 (s, 1H), 6.66 (s, 1H); Key NOESY
correlations: H-2/OMe-3, H-2/H-19â, H-2/H-20â, H-4/H-5, H-5/
H-7, H-5/H-20R, H-13/H-15R; 13C NMR (CDCl3) δ 17.8 (t), 19.5
(t), 29.0 (q), 29.3 (q), 29.7 (t), 35.6 (t), 39.3 (t), 43.0 (t), 43.8 (t),
49.4 (t), 51.4 (q), 55.5 (t), 57.1 (q), 59.2 (d), 70.8 (s), 74.7 (s), 77.5
(d), 87.5 (s), 101.0 (t), 102.8 (d), 111.7 (d), 114.4 (d), 129.4 (s),
134.7 (s), 145.3 (s), 146.3 (s), 161.4 (s), 170.2 (s), 174.7 (s); EIMS
m/z (%) 561 (15, M+), 545 (13), 530 (6), 314 (100), 298 (72);
HRFABMS [M + H]+ calcd for C29H40NO10 562.2652, found
562.2650.
Exp er im en ta l Section
Gen er a l. Analytical high performance liquid chromatogra-
phy (HPLC) was carried out on a TSKgel silica 60 column (5
µm, 4.6 × 250 mm, TOSOH) at a flow rate of 1 mL/min.
Semipreparative HPLC was carried out on a LiChrosorb Si 60
column (7 µm, 25 × 250 mm, Merck) at a flow rate of 7 mL/min.
The effluent was monitored at 290 nm. Rotations were recorded
in units of 10-1 deg cm2 g-1 1H and 13C NMR spectra were
;
collected using a 500 or 125 MHz instrument, respectively.
Chemical shifts (δ) were reported in ppm. 1H chemical shifts
were calibrated using the tetramethylsilane resonance as inter-
nal standard. 13C chemical shifts were referenced to the solvent
(CDCl3, 77.0 ppm). The chemical shift assignment was carried
out by homonuclear correlation (COSY) and heteronuclear
correlation (HMQC, HMBC) experiments. Mass spectroscopy
was performed using EI and FAB ion sources.
1-[(1R,4S,5S,15S)-3-Meth oxy-15-m eth yl-9,11,20-tr ioxa -16-
azapen tacyclo[14.3.1.01,5.06,14.08,12]icosa-2,6,8(12),13-tetr aen -
4-yl] 4-m eth yl (2R)-2-h ydr oxy-2-(4-h ydr oxy-4-m eth ylpen tyl)-
su ccin a te (5): an amorphous solid; [R]D ) -148˚ (c 1.00,
MeOH); UV (MeOH) λmax 292 nm (log ꢀ 3.62); IR (film) 3210 br,
1744, 1656, 1489 cm-1; 1H NMR (CDCl3) δ 1.15 (s, 3H), 1.18 (m,
1H), 1.18 (s, 3H), 1.3-1.45 (m, 5H), 1.56 (m, 1H), 1.83 (d, J )
6.8 Hz, 3H), 1.85 (d, J ) 16.4 Hz, 1H), 1.88 (ddd, J ) 13.0, 7.5,
5.0 Hz, 1H), 2.27 (m, 1H), 2.29 (d, J ) 16.4 Hz, 1H), 2.80 (dt, J
) 13.0, 8.3 Hz, 1H), 3.01 (ddd, J ) 11.5, 8.9, 7.0 Hz, 1H), 3.24
(ddd, J ) 11.5, 8.1, 5.8 Hz, 1H), 3.60 (d, J ) 8.1 Hz, 1H), 3.60
(s, 3H), 3.79 (s, 3H), 4.39 (q, J ) 6.8 Hz, 1H), 5.29 (s, 1H), 5.91
(d, J ) 8.1 Hz, 1H), 5.96 (d, J ) 1.4 Hz, 1H), 5.99 (d, J ) 1.4 Hz,
1H), 6.62 (s, 1H), 6.83 (br s, 1H); Key NOESY correlations: H-2/
OMe-3, H-2/H-15, H-2/H-19â, H-4/H-5, H-5/H-7, H-5/H-19R,
H-13/Me-15, Me-15/H-17R, Me-15/H-17â, H-15/H-17â; 13
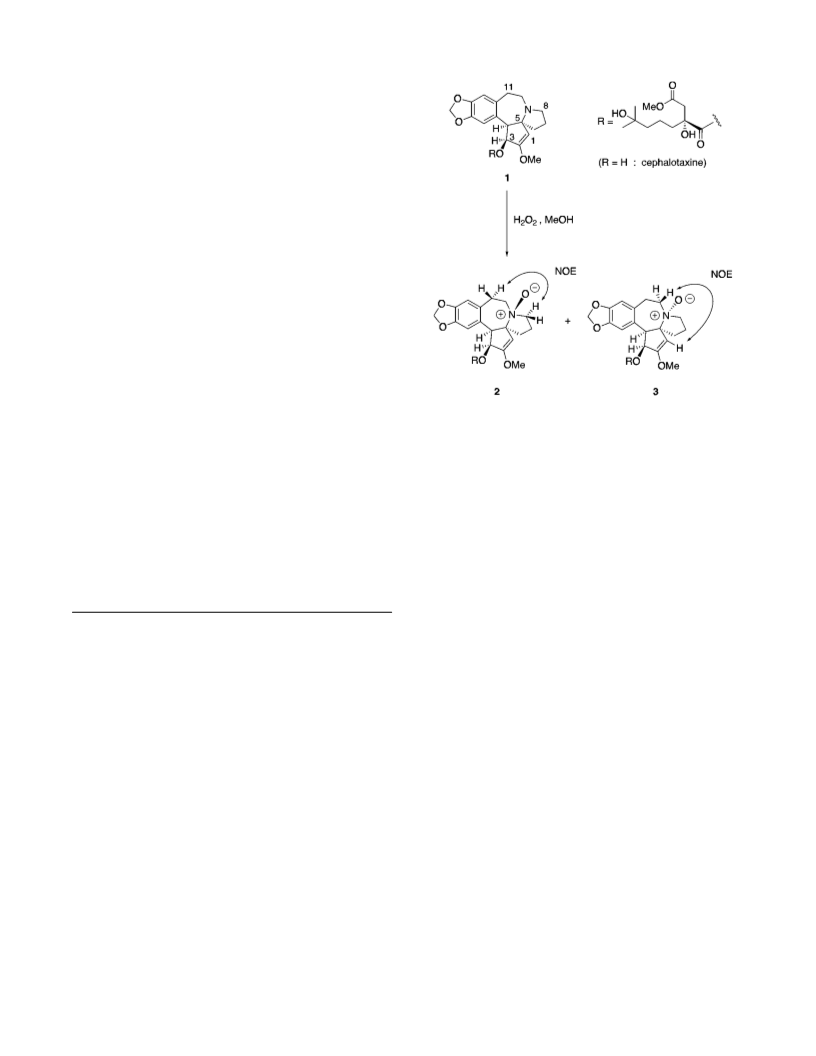
Hom oh a r r in gton in e â-N-oxid e (2) a n d r-N-oxid e (3). To
a solution of homoharringtonine (1) (100 mg) in MeOH (0.5 mL)
was added 30% aqueous H2O2 (0.1 mL), and the mixture was
stirred at room temperature for 24 h. Methyl sulfide (50 µL)
was added to the solution at 0 °C, and the mixture was stirred
at room temperature for 3 h. The reaction mixture was
concentrated under reduced pressure to give a residue, which
was chromatographed (HPLC, SiO2, 1:1:2 CHCl3/n-hexane/
MeOH) to provide N-oxides 2 (27 mg, 26%) and 3 (37 mg, 36%).
2: a colorless oil; [R]D ) -90° (c 0.22, MeOH); UV (MeOH)
λmax 292 nm (log ꢀ 3.66); IR (film) 3350 br, 1742, 1652, 1505,
1490 cm-1 1H NMR (CDCl3) δ 1.16 (s, 3H), 1.18 (s, 3H), 1.23
;
(m, 1H), 1.38 (m, 2H), 1.40 (m, 1H), 1.50 (m, 2H), 1.71 (m, 1H),
1.86 (m, 1H), 2.25 (m, 1H), 2.29 (d, J ) 16.5 Hz, 1H), 2.43 (d, J
) 16.5 Hz, 1H), 2.74 (m, 1H), 2.99 (td, J ) 10.0, 7.6 Hz, 1H),
3.06 (td, J ) 10.0, 7.0 Hz, 1H), 3.37 (td, J ) 10.0, 5.0 Hz, 1H),
3.54 (s, 3H), 3.64 (m, 2H), 3.73 (d, J ) 8.4 Hz, 1H), 3.81 (s, 3H),
3.93 (m, 1H), 5.43 (s, 1H), 5.90 (d, J ) 8.4 Hz, 1H), 5.94 (br s,
2H), 6.60 (s, 1H), 6.61 (s, 1H); Key NOESY correlations: H-1/
H-6â, H-1/Me-19, H-3/H-4, H-4/H-6R, H-4/H-14, H-8R/H-11R,
H-11R/H-17; 13C NMR (CDCl3) δ 17.7 (t), 19.3 (t), 28.8 (q), 29.7
(q), 30.3 (t), 38.3 (t), 39.4 (t), 42.7 (t), 43.5 (t), 51.5 (q), 55.8 (d),
57.9 (q), 62.5 (t), 66.6 (t), 70.4 (s) × 2, 75.9 (d), 89.3 (s), 101.3 (t),
101.6 (d), 109.3 (d), 112.1 (d), 125.8 (s), 130.7 (s), 146.7 (s), 147.9
(s), 160.7 (s), 170.3 (s), 173.8 (s); EIMS m/z (%) 561 (6, M+), 545
(7), 530 (10), 314 (62), 298 (100); HRFABMS [M + H]+ calcd for
C NMR
(CDCl3) δ 12.2 (q), 17.6 (t), 21.9 (t), 28.7 (q), 29.8 (q), 37.9 (t),
39.4 (t), 42.8 (t), 43.6 (t), 51.6 (q), 51.7 (d), 58.0 (q), 62.5 (t), 65.3
(d), 70.2 (s), 74.5 (s), 76.2 (d), 89.0 (s), 101.5 (t), 102.5 (d), 106.2
(d), 109.7 (d), 125.4 (s), 130.2 (s), 147.7 (s), 147.8 (s), 160.1 (s),
170.2 (s), 173.5 (s); EIMS m/z (%) 561 (1, M+), 543 (2), 530 (18),
314 (18), 298 (100); HRFABMS [M + H]+ calcd for C29H40NO10
562.2652, found 562.2669.
1-[(1R,4S,5S,15R)-3-Met h oxy-15-m et h yl-9,11,20-t r ioxa -
16-a za p en ta cyclo[14.3.1.01,5.06,14.08,12]icosa -2,6,8(12),13-tet-
r a en -4-yl] 4-m eth yl (2R)-2-h yd r oxy-2-(4-h yd r oxy-4-m eth -
ylp en tyl)su ccin a te (6): a colorless oil; [R]D ) -141˚ (c 0.20,
MeOH); UV (MeOH) λmax 292 nm (log ꢀ 3.62); IR (film) 3360 br,
1744, 1661, 1488 cm-1; 1H NMR (CDCl3) δ 1.16 (s, 3H), 1.17 (s,
3H), 1.18 (m, 1H), 1.32 (m, 2H), 1.35 (m, 1H), 1.44 (m, 2H), 1.71
(d, J ) 7.3 Hz, 3H), 1.71 (m, 1H), 1.82 (m, 1H), 1.92 (d, J ) 16.0
Hz, 1H), 2.30 (m, 1H), 2.33 (d, J ) 16.0 Hz, 1H), 2.96 (dt, J )
12.5, 9.2 Hz, 1H), 3.51 (m, 1H), 3.63 (ddd, J ) 11.5, 10.0, 6.1
Hz, 1H), 3.68 (s, 3H), 3.72 (s, 3H), 3.78 (d, J ) 8.9 Hz, 1H), 4.39
(q, J ) 7.3 Hz, 1H), 5.50 (s, 1H), 5.91 (d, J ) 1.3 Hz, 1H), 5.97
(d, J ) 1.3 Hz, 1H), 6.12 (d, J ) 8.9 Hz, 1H), 6.54 (s, 1H), 6.59
(s, 1H); Key NOESY correlations: H-2/OMe-3, H-2/Me-15, H-2/
H-19â, H-4/H-5, H-5/H-7, H-5/H-19R, H-13/H-15, H-15/H-17R,
H-15/H-17â; 13C NMR (CDCl3) δ 17.7 (t), 18.4 (q), 20.7 (t), 29.2
(q), 29.3 (q), 39.1 (t), 40.6 (t), 42.8 (t), 43.7 (t), 50.4 (d), 51.8 (q),
58.0 (q), 70.7 (s), 71.5 (d), 72.0 (t), 74.7 (s), 76.8 (d), 84.7 (s),
101.4 (t), 104.5 (d), 107.9 (d), 110.2 (d), 122.1 (s), 131.0 (s), 147.4
(s) × 2, 156.6 (s), 170.4 (s), 173.8 (s); EIMS m/z (%) 530 (20),
314 (35), 300 (100), 298 (95); HRFABMS [M + H]+ calcd for
C
29H40NO10 562.2652, found 562.2637.
3: a colorless oil; [R]D ) -179° (c 0.42, MeOH); UV (MeOH)
λmax 294 nm (log ꢀ 3.54); IR (film) 3365 br, 1746, 1659, 1505,
1489 cm-1 1H NMR (CDCl3) δ 1.18 (s, 3H), 1.19 (s, 3H), 1.2-
;
1.4 (m, 6H), 1.86 (d, J ) 16.5 Hz, 1H), 1.89 (m, 1H), 1.97 (br t,
J ) 11.0 Hz, 1H), 2.22 (m, 1H), 2.27 (d, J ) 16.5 Hz, 1H), 2.51
(dd, J ) 14.7, 6.6 Hz, 1H), 2.94 (m, 1H), 3.45 (m, 1H), 3.61 (m,
3H), 3.61 (s, 3H), 3.74 (m, 1H), 3.75 (s, 3H), 3.94 (d, J ) 9.7 Hz,
1H), 4.92 (s, 1H), 5.83 (br s, 1H), 5.86 (br s, 1H), 6.02 (d, J ) 9.7
Hz, 1H), 6.51 (s, 1H), 6.64 (s, 1H); Key NOESY correlations: H-1/
H-6â, H-1/H-10â, H-1/Me-19, H-3/H-4, H-4/H-6R, H-4/H-14,
H-11R/H-17; 13C NMR (CDCl3) δ 17.7 (t), 18.3 (t), 28.6 (t), 29.0
(q), 29.4 (q), 38.9 (t), 39.4 (t), 42.3 (t), 43.6 (t), 51.6 (q), 52.1 (d),
58.0 (q), 62.6 (t), 70.7 (t), 70.7 (s), 72.8 (d), 74.7 (s), 84.9 (s), 100.0
(d), 100.7 (t), 109.2 (d), 111.5 (d), 128.5 (s), 130.4 (s), 145.7 (s),
145.9 (s), 161.7 (s), 170.6 (s), 173.6 (s); EIMS m/z (%) 561 (1,
M+), 545 (4), 530 (4), 314 (53), 298 (100); HRFABMS [M + H]+
calcd for C29H40NO10 562.2652, found 562.2648.
C
29H40NO10 562.2652, found: 562.2667.
Red u ction of 4 to Hom oh a r r in gton in e (1). To a solution
of 4 (20 mg) in AcOH (1 mL) was added zinc powder (20 mg),
and the mixture was stirred at 47 °C for 1 h. The reaction
mixture was filtrated and concentrated. The residue was
dissolved in CHCl3 (35 mL) and washed successively with
saturated aqueous NaHCO3 (5 mL) and brine (5 mL). The
Th er m a l Rea r r a n gem en ts of 2 a n d 3. A solution of 2 (100
mg) in 1,2-dimethoxyethane (1 mL) was heated at 105 °C in a
sealed tube for 2 h. The mixture was concentrated under
reduced pressure to give a residue, which was chromatographed

 Takano
Takano
 Yasuda
Yasuda
 Nishijima
Nishijima
 Hitotsuyanagi
Hitotsuyanagi
 Takeya
Takeya
 Itokawa
Itokawa