Journal of the American Chemical Society
Communication
(5) Goto, Y.; Ohta, A.; Sako, Y.; Yamagishi, Y.; Murakami, H.; Suga, H.
ACS Chem. Biol. 2008, 3, 120.
(6) Bionda, N.; Cryan, A. L.; Fasan, R. ACS Chem. Biol. 2014, 9, 2008.
(7) Horne, W. S.; Olsen, C. A.; Beierle, J. M.; Montero, A.; Ghadiri, M.
R. Angew. Chem., Int. Ed. 2009, 48, 4718.
(8) Wuo, M. G.; Mahon, A. B.; Arora, P. S. J. Am. Chem. Soc. 2015, 137,
11618.
(9) Blackwell, H. E.; Sadowsky, J. D.; Howard, R. J.; Sampson, J. N.;
Chao, J. A.; Steinmetz, W. E.; O’Leary, D. J.; Grubbs, R. H. J. Org. Chem.
2001, 66, 5291.
(10) Kim, Y. W.; Grossmann, T. N.; Verdine, G. L. Nat. Protoc. 2011, 6,
761.
(11) Rohrbacher, F.; Deniau, G.; Luther, A.; Bode, J. W. Chem. Sci.
2015, 6, 4889.
(12) Mendive-Tapia, L.; Preciado, S.; Garcia, J.; Ramon, R.; Kielland,
N.; Albericio, F.; Lavilla, R. Nat. Commun. 2015, 6, 7160.
(13) Lian, W.; Upadhyaya, P.; Rhodes, C. A.; Liu, Y.; Pei, D. J. Am.
Chem. Soc. 2013, 135, 11990.
(14) Heinis, C.; Rutherford, T.; Freund, S.; Winter, G. Nat. Chem. Biol.
2009, 5, 502.
(15) Branco, M. C.; Sigano, D. M.; Schneider, J. P. Curr. Opin. Chem.
Biol. 2011, 15, 427.
(16) Zhou, J.; Xu, B. Bioconjugate Chem. 2015, 26, 987.
(17) Bowerman, C. J.; Nilsson, B. L. J. Am. Chem. Soc. 2010, 132, 9526.
(18) Hutin, M.; Bernardinelli, G.; Nitschke, J. R. Chem. - Eur. J. 2008,
14, 4585.
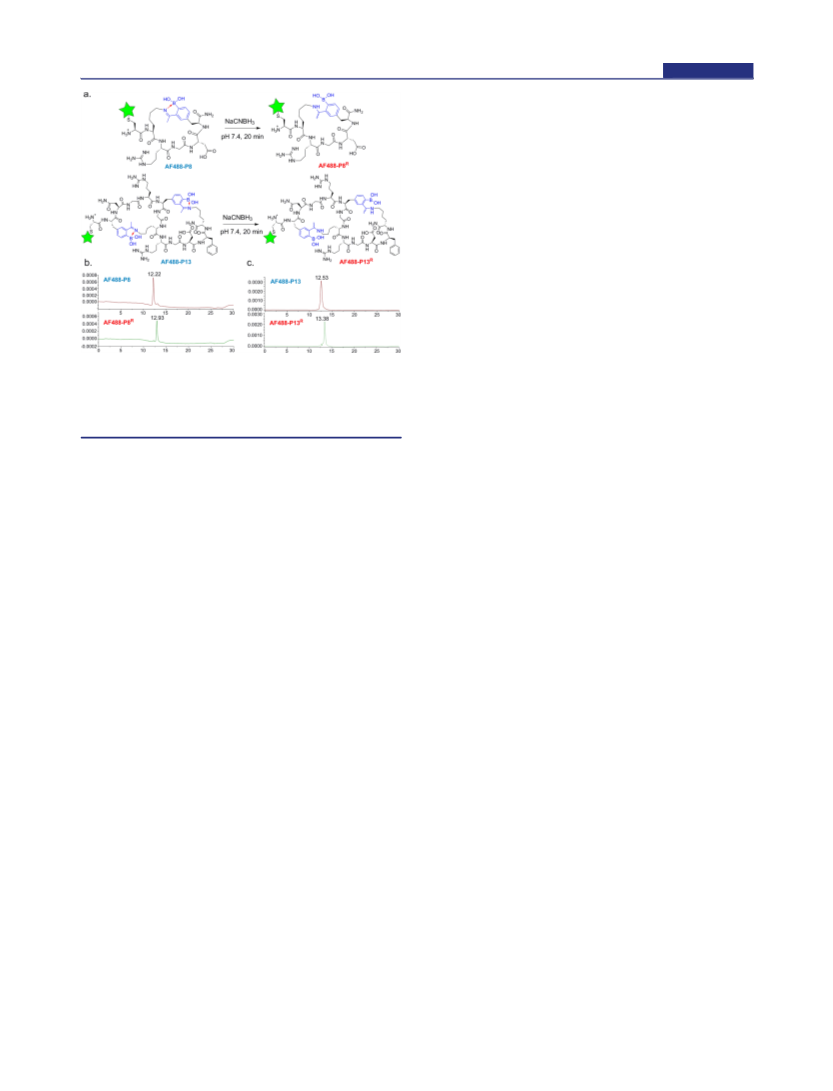
Figure 5. Iminoboronate reduction yielding permanently cyclized
peptides. (a) Schematic illustration of the iminoboronate reduction to
give irreversible peptide cyclization; (b) LC chromatograms of AF488-
P8 before (top) and after (bottom) reduction; (c) LC chromatograms
of AF488-P13 before (top) and after reduction (bottom) reduction.
(19) Galbraith, E.; Kelly, A. M.; Fossey, J. S.; Kociok-Kohn, G.;
Davidson, M. G.; Bull, S. D.; James, T. D. New J. Chem. 2009, 33, 181.
(20) Gutierrez-Moreno, N. J.; Medrano, F.; Yatsimirsky, A. K. Org.
Biomol. Chem. 2012, 10, 6960.
(21) Cal, P. M.; Vicente, J. B.; Pires, E.; Coelho, A. V.; Veiros, L. F.;
Cordeiro, C.; Gois, P. M. J. Am. Chem. Soc. 2012, 134, 10299.
(22) Bandyopadhyay, A.; McCarthy, K. A.; Kelly, M. A.; Gao, J. Nat.
Commun. 2015, 6, 6561.
(23) Stubbs, M.; McSheehy, P. M.; Griffiths, J. R.; Bashford, C. L. Mol.
Med. Today 2000, 6, 15.
(24) Andreev, O. A.; Dupuy, A. D.; Segala, M.; Sandugu, S.; Serra, D.
A.; Chichester, C. O.; Engelman, D. M.; Reshetnyak, Y. K. Proc. Natl.
Acad. Sci. U. S. A. 2007, 104, 7893.
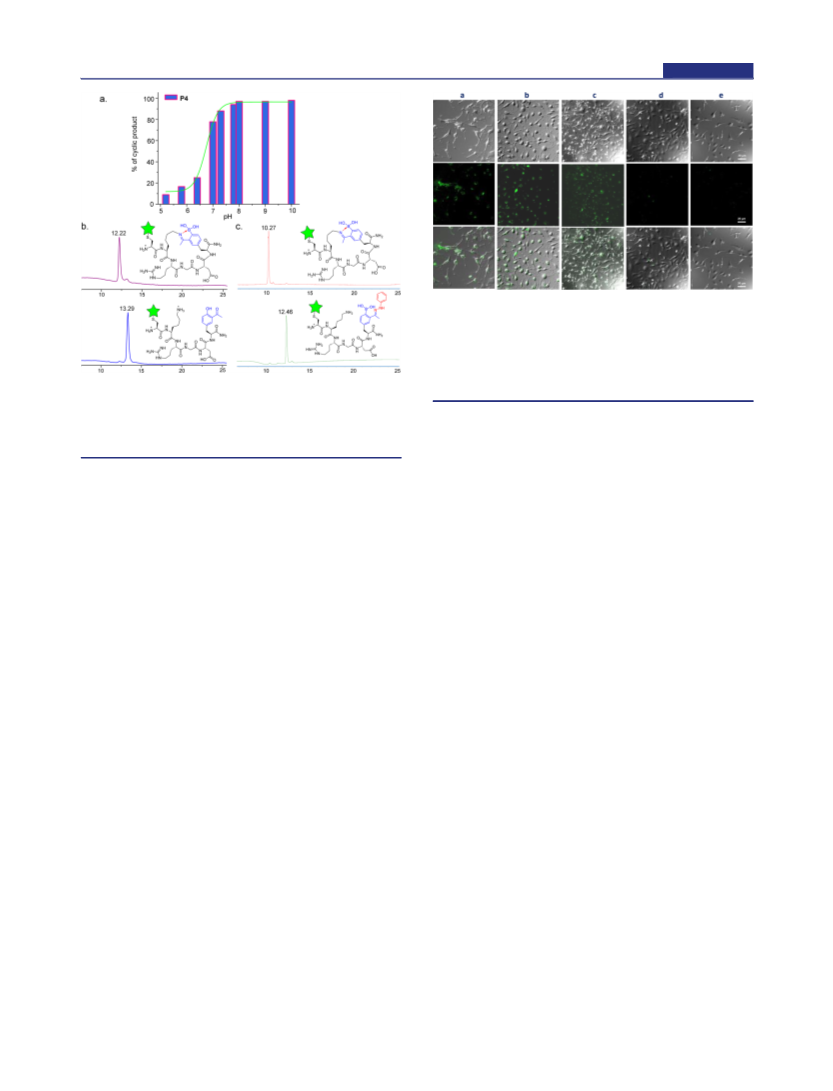
extremely sensitive to the amino acid composition of the
peptides.30 This is perhaps not surprising given the unfavorable
thermodynamic equilibrium of imine formation in aqueous
media. In contrast to simple imines, iminoboronates enjoy much
greater thermodynamic stability due to stabilization by the dative
bond. At neutral pH, the iminoboronate-cyclized peptides
display surprisingly robust stability against commonly seen
biomolecules. Excitingly, despite the robustness in biological
milieu, the iminoboronate-mediated cyclization can be readily
reversed with acidification, oxidation, and addition of exogenous
small molecule modulators. We believe the quick response to
multiple stimuli will make the iminoboronate cyclization strategy
useful for a wide range of applications in biotechnology.
(25) Lippert, A. R.; De Bittner, G. C. V.; Chang, C. J. Acc. Chem. Res.
2011, 44, 793.
(26) Chen, Z. J.; Ren, W.; Wright, Q. E.; Ai, H. W. J. Am. Chem. Soc.
2013, 135, 14940.
(27) Bandyopadhyay, A.; Gao, J. Chem. - Eur. J. 2015, 21, 14748.
(28) Mas-Moruno, C.; Rechenmacher, F.; Kessler, H. Anti-Cancer
Agents Med. Chem. 2010, 10, 753.
(29) Becker, J. E.; Moore, R. E.; Moore, B. S. Gene 2004, 325, 35.
(30) Kopp, F.; Mahlert, C.; Grunewald, J.; Marahiel, M. A. J. Am. Chem.
Soc. 2006, 128, 16478.
ASSOCIATED CONTENT
* Supporting Information
The Supporting Information is available free of charge on the
■
S
Details of synthesis and characterization (PDF)
(31) Evans, B. S.; Ntai, I.; Chen, Y.; Robinson, S. J.; Kelleher, N. L. J.
Am. Chem. Soc. 2011, 133, 7316.
AUTHOR INFORMATION
Corresponding Author
■
Notes
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
The authors acknowledge the financial support from the
National Institutes of Health via the grant GM102735.
■
REFERENCES
■
(1) Baeriswyl, V.; Heinis, C. ChemMedChem 2013, 8, 377.
(2) Bock, J. E.; Gavenonis, J.; Kritzer, J. A. ACS Chem. Biol. 2013, 8,
488.
(3) Passioura, T.; Katoh, T.; Goto, Y.; Suga, H. Annu. Rev. Biochem.
2014, 83, 727.
(4) Heinis, C.; Winter, G. Curr. Opin. Chem. Biol. 2015, 26, 89.
D
J. Am. Chem. Soc. XXXX, XXX, XXX−XXX

 Bandyopadhyay, Anupam
Bandyopadhyay, Anupam
 Gao, Jianmin
Gao, Jianmin