738
Y. Y. Rusakov et al.
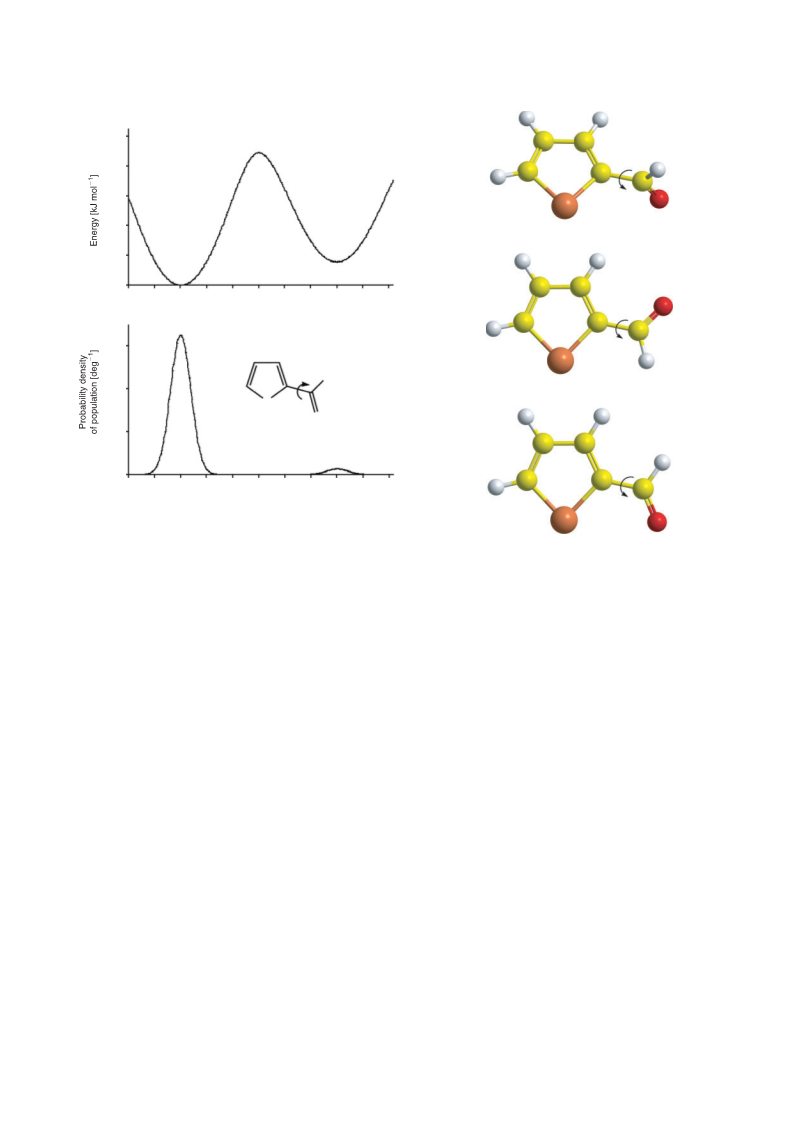
predominance of the s-cis conformer compared with the s-trans
one originates in several hyperconjugative interactions resulting
in the increased π-stabilization of the former.
References
[1] Yu. Yu. Rusakov, L. B. Krivdin, N. V. Istomina, V. A. Potapov,
S. V. Amosova, Magn. Reson. Chem. 2008, 46, 979. doi:10.1002/
MRC.2291
[2] C. W. Nogueira, G. Zeni, J. B. T. Rocha, Chem. Rev. 2004, 104, 6255.
doi:10.1021/CR0406559
Experimental
[3] (a) L. B. Krivdin, Yu. Yu. Rusakov, E. Yu. Schmidt, A. I. Mikhaleva,
B. A. Trofimov, Aust. J. Chem. 2007, 60, 583. doi:10.1071/CH07086
(b) S. V. Fedorov, L. B. Krivdin, Yu. Yu. Rusakov, I. A. Ushakov,
N. V. Istomina, N. A. Belogorlova, S. F. Malysheva, N. K. Gusarova,
B. A. Trofimov, Magn. Reson. Chem. 2009, 47, 288. doi:10.1002/
MRC.2386
[4] M. P. Simonnin, M. J. Pouet, J. M. Cense, C. Paulmier, Org. Magn.
Reson. 1976, 8, 508. doi:10.1002/MRC.1270081005
[5] (a) E. S. Nielsen, P. Jørgensen, J. Oddershede, J. Chem. Phys. 1980,
73, 6238. doi:10.1063/1.440119
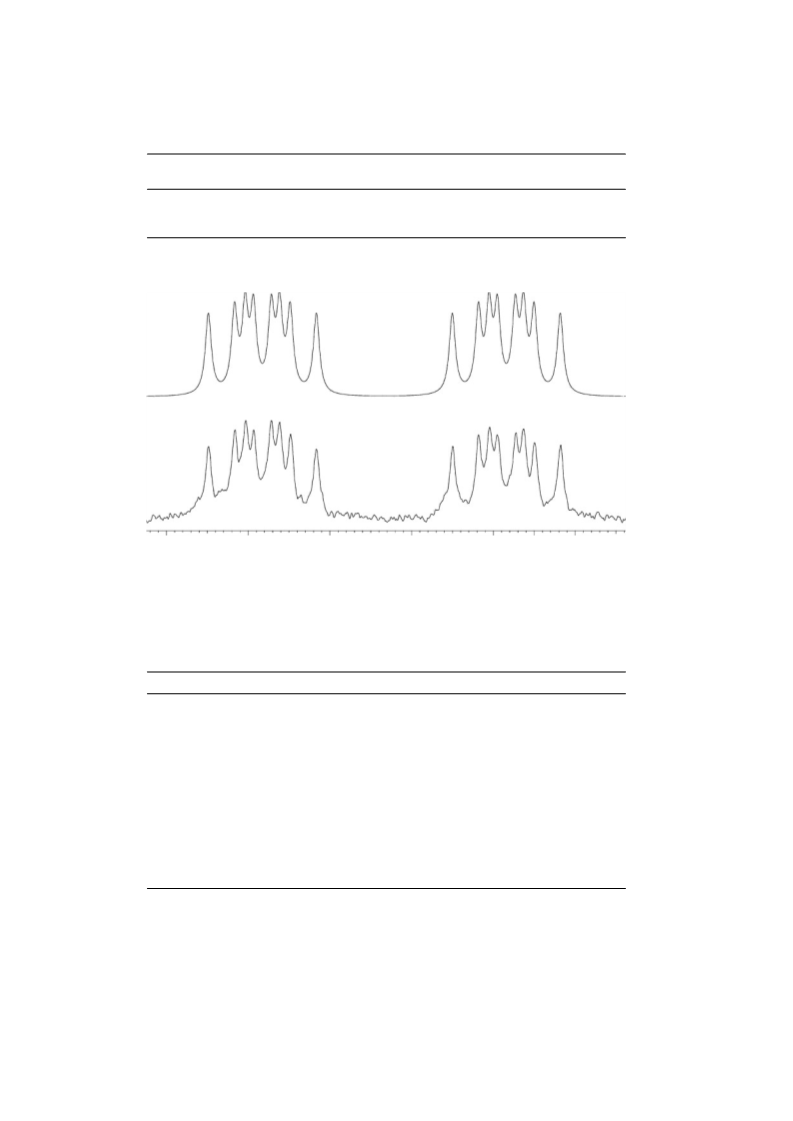
NMR Measurements
1H, 13C, and 77Se NMR spectra were recorded on a Bruker DPX
400 MHz spectrometer (1H, 400.13 MHz; 13C, 100.62 MHz;
77Se, 76.34 MHz) in a 5-mm broadband probe at 25◦C in CDCl3
with hexamethyldisiloxane (HMDS) as an internal standard.
77Se–1H coupling constants were measured from the proton-
coupled 77Se NMR spectra using the spectral settings as follows:
90◦ pulse length, 13 µs; spectral width, 8 kHz; acquisition time,
8.5 s; relaxation delay, 3 s; digital resolution, 0.06 Hz per pt;
accumulation time, 1 h. Carbon–proton and carbon–carbon cou-
plings were measured from 1D and 2D proton-coupled and
INADEQUATE 13C NMR spectra respectively using standard
spectral parameters providing 0.05 Hz per pt digital resolution.
(b) K. L. Bak, H. Koch, J. Oddershede, O. Christiansen, S. P. A. Sauer,
J. Chem. Phys. 2000, 112, 4173. doi:10.1063/1.480963
(c) M. J. Packer, E. K. Dalskov, T. Enevoldsen, H. J. A. Jensen,
J. Oddershede, J. Chem. Phys. 1996, 105, 5886. doi:10.1063/1.472430
(d) T. Enevoldsen, J. Oddershede, S. P. A. Sauer, Theor. Chem. Acc.
1998, 100, 275. doi:10.1007/S002140050388
Computational Details
[6] P. F. Provasi, G. A. Aucar, S. P. A. Sauer, J. Chem. Phys. 2001, 115,
1324. doi:10.1063/1.1379331
All geometry optimizations and calculation of the rotational
potential energy curve were performed with the GAMESS
code[8] at the MP2 perturbation level[9] using the 6–311G**
basis set of Pople and coworkers[10] without symmetry con-
straints, i.e. assumingtheC1 symmetrypointgroup. Calculations
of spin–spin coupling constants were carried out taking into
account all four non-relativistic coupling contributions with the
DALTON package[11] at the SOPPA level[5] using the station-
ary equilibrium geometries located at the MP2/6–311G** level,
taking into account solvent effect of CDCl3 within the PCM
model. The correlation-consistent basis set aug-cc-pVTZ-J of
Sauer et al.[6] was used for selenium and coupled carbons and
hydrogens whereas all uncoupled atoms were specified with
cc-pVDZ of Woon and Dunning.[12]
[7] (a) R. H. Contreras, J. E. Peralta, Prog. Nucl. Magn. Reson. Spectrosc.
2000, 37, 321. doi:10.1016/S0079-6565(00)00027-3
(b) R. H. Contreras, V. Barone, J. C. Facelli, J. E. Peralta, Ann. Rep.
NMR Spectrosc. 2003, 51, 167. doi:10.1016/S0066-4103(03)51004-3
(c) L. B. Krivdin, R. H. Contreras, Ann. Rep. NMR Spectrosc. 2007,
61, 133. doi:10.1016/S0066-4103(07)61103-X
(d) L. B. Krivdin, G. A. Kalabin, Prog. Nucl. Magn. Reson. Spectrosc.
1989, 21, 293. doi:10.1016/0079-6565(89)80007-X
[8] M. W. Schmidt, K. K. Baldridge, J. A. Boatz, S. T. Elbert,
M. S. Gordon, J. H. Jensen, S. Koseki, N. Matsunaga, K. A. Nguyen,
S. Su, T. L. Windus, M. Dupuis, J. A. Montgomery, Jr, J. Comput.
Chem. 1993, 14, 1347. doi:10.1002/JCC.540141112
[9] C. Møller, M. S. Plesset, Phys. Rev. 1934, 46, 618. doi:10.1103/
PHYSREV.46.618
[10] R. Krishnan, J. S. Binkley, R. Seeger, J.A. Pople, J. Chem. Phys. 1980,
72, 650. doi:10.1063/1.438955
[11] Dalton,A Molecular Electronic Structure Program, Release 2.0 2005,
May 2009).
Synthesis
2-Formylselenophene ( 1) was synthesized from selenophene via
theVilsmeier–Haack reaction, as described in Ref. [13], whereas
selenophene was obtained by the thermolysis of diethyl selenide
and acetylene in the presence of methanol.[14]
[12] (a) T. H. Dunning, Jr, J. Chem. Phys. 1989, 90, 1007.
doi:10.1063/1.456153
(b) R. A. Kendall, T. H. Dunning, Jr, R. J. Harrison, J. Chem. Phys.
1992, 96, 6796. doi:10.1063/1.462569
(c) D. E. Woon, T. H. Dunning, Jr, J. Chem. Phys. 1993, 98, 1358.
doi:10.1063/1.464303
Acknowledgements
Financial support from the Russian Foundation for Basic Research (Grant
no. 08–03–00021) is acknowledged. L.B.K. thanks Professor Sauer for pro-
viding him with the extended aug-cc-pVTZ-J basis set including selenium.
[13] Yu. K. Yur’ev, N. N. Mezentseva, Zh. Obsch. Khim. 1957, 27, 179.
[14] E. N. Deryagina, E. N. Sukhomazova, E. P. Levanova, N.A. Korchevin,
A. P. Danilova, Russ. J. Gen. Chem. 2004, 40, 290.

 Rusakov, Yury Yu.
Rusakov, Yury Yu.
 Krivdin, Leonid B.
Krivdin, Leonid B.
 Istomina, Natalia V.
Istomina, Natalia V.
 Levanova, Ekaterina P.
Levanova, Ekaterina P.
 Levkovskaya, Galina G.
Levkovskaya, Galina G.