1302
Vol. 57, No. 11
that can be used for acetylation without any further purification step.
b) Shapiro Reaction: The trisylhydrazone 7 (10.3 g, 25.3 mmol, 1 eq) is
first dissolved in anhydrous THF (50 ml) and the resulting homogeneous so-
lution is cooled to ꢄ78 °C. n-Butyllithium (47.5 ml, 76 mmol, 3 eq) is then
added dropwise over a period of 30 min. The red solution is then stirred for
2 h at ꢄ78 °C, and cooled to 0 °C. Nitrogen evolution takes place over a pe-
Hadley M. S., Hawkins J., Loudon J. M., Naylor C. B., Orlek B. S.,
Riley G. J., J. Med. Chem., 40, 4265—4280 (1997).
8) Bos M., Canesso R., Heterocycles, 38, 1889 (1994).
9) Clark R. D., Weinhardt K. K., Berger J., Lee C. H., Leung E., Wong E.
H. F., Smith W. L., Eglen R. M., Bioorg. Med. Chem. Lett., 3, 1375—
1378 (1993).
riod of 15 min, while the reaction mixture turns from orange to yellow. Solid 10) Brown G. R., Foubister A. J., Freeman S., Mc Taggart F., Mirrlees D.
paraformaldehyde (2.28 g, 76 mmol, 3 eq) is then added at 0 °C and the reac-
tion medium is stirred at room temperature for 3 h. During this period of
J., Reid, A. C., Smith G. J., Taylor M. J., Thomason D. A., Whittamore
P. R. O., Bioorg. Med. Chem. Lett., 7, 597—600 (1997).
time, the mixture turns to clear yellow color. The reaction is then quenched 11) Persi L., Dupin O., Arnaud B., Trinquand C., Michel F.-B., Bousquet
with an aqueous solution of K2CO3 (10%, 40 ml) and extracted with Et2O J., Allergy, 52, 451—454 (1997).
(3ꢂ50 ml). The combined organic phases were dried over Na2SO4, filtered 12) Theunissen E. L., Vermeeren A., van Oers A. C. M., van Maris I., Ra-
and evaporated to dryness. The crude is then purified by flash chromatogra-
phy on silica (eluting with: CH2Cl2 : MeOH : NEt3, 9 : 1 : 0.1, v/v/v) to yield
maekers J. G., Clin. Exp. Allergy, 34, 250—258 (2004).
13) Buksa M., Cernobrovijs A., Materialzinatne un Lietiska Kimija, 12,
27—32 (2006).
6 as a yellow oil (6.4 g, 91%).
1
Rfꢅ0.1 (CH2Cl2 : MeOH, 9 : 1, v/v); H-NMR (CDCl3, 300 MHz) d: 6.15 14) Yamazaki S., Yumoto H., Igi M., Eur. Pat. Appl., EP 1074552 A2
(s, 1H), 4.08 (s, 2H), 2.78—2.87 (m, 2H), 2.42—2.54 (m, 3H), 1.50—1.60
(m, 2H), 1.30—1.50 (m, 2H); 13C-NMR (CDCl3, 75 MHz) d: 148.8, 133.6, 15) Guminski Y., Imbert Y., Lesimple P., FR 2777278 A1 19991015
61.0, 49.2, 27.8, 27.5; IR (KBr) cmꢄ1: 3387, 2949, 2873, 1651, 1456, 1306,
(1999).
1129, 1056, 1017, 765, 669; HR-MS (m/z): Calcd for C8H13NO: 139.0997, 16) Guminski Y., Imbert Y., Lesimple P., Org. Prep. Proc. Int., 31, 319—
Found: 139.0994. 323 (1999).
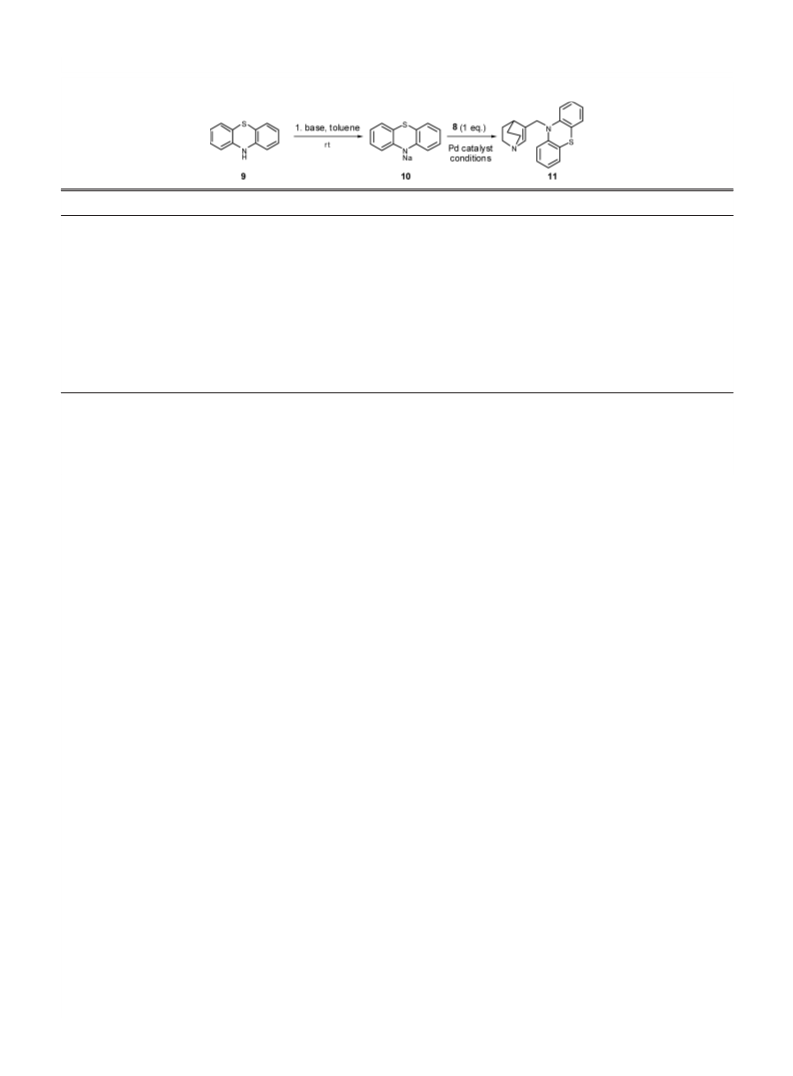
Typical Procedure for the Synthesis of 11 a) Preparation of sodium 17) Guminski Y., Imbert Y., Lesimple P., WO 9929692 A1 19990617
20010207 (2001).
phenothiazinate 10: To a stirred THF (8 ml) solution of phenothiazine 9
(150 mg, 0.75 mmol, 3 eq) and under an atmosphere of Argon, NaH (60% in
oil, 33 mg, 0.83 mmol, 3.3 eq) is added portion wise. The reaction mixture is
stirred for 25 min while the medium becomes dark green.
b) In an another flame dried flask under an Argon atmosphere, containing
THF (2 ml) and the allylic acetate 7 (45.3 mg, 0.25 mmol, 1 eq), the palla-
dium catalyst Pd(PPh3)4 is added (29 mg, 0.025 mmol, 10 mol%). The result-
ing red mixture is stirred for 10—15 min at room temperature and then
transfered, via a canula to the flask containing the sodium phenothiazinate
10. After 3 h at room temperature, the reaction is quenched with a saturated
aqueous solution of K2CO3 (6 ml) and extracted with CH2Cl2. The resulting
(1999).
18) Mioskowski C., Gonnot V., Baati R., Nicolas M., WO 2008107545 A1
20080912 (2008).
19) Corey E. J., Chaykovsky M., J. Am. Chem. Soc., 87, 1345—1353
(1965).
20) Corey E. J., Chaykovsky M., J. Am. Chem. Soc., 87, 1354—1364
(1965).
21) Lauffer D. J., Moos W. H., Tecle H., US 89-310229, 4937239,
19890213, 1990, 12.
22) Yamauchi H., Sugiyama I., Saito I., Nomoto S., Kamya T., Machida
Y., Negi S., JP 84-114484, 60258187, 19840606, 1985, 7.
organic phases were dried with Na2SO4, and concentrated under vacuum. 23) Treves G. R., Baum B. M., US 69-800350, 4467095, 19690210, 1984,
The crude green oil is then purified by flash chromatography on silica (elut- 9.
ing with: CH2Cl2 : MeOH : NEt3, 96 : 4 : 0.5, v/v/v) to afford a slightly yellow 24) Crandall J. K., Lin L.-H. C., J. Org. Chem., 32, 435—439 (1967).
solid (74 mg, 92%).
25) Crandall J. K., Lin L.-H. C., J. Org. Chem., 33, 2375—2378 (1968).
26) Yasuda A., Tanaka S., Oshima K., Yamamoto H., Nozaki H., J. Am.
Chem. Soc., 96, 6513—6514 (1974).
Rfꢅ0.1 (CH2Cl2 : MeOH, 9 : 1, v/v); mp 149 °C; 1H-NMR (CDCl3,
3
4
300 MHz) d: 7.06—7.15 (m, 4H), 6.90 (td, Jꢅ7.4 Hz, Jꢅ1 Hz, 2H), 6.82
3
(d, Jꢅ8.0 Hz, 2H), 6.52 (s, 1H), 4.58 (s, 2H), 2.84—3.00 (m, 2H), 2.77 (s, 27) Bertilsson S. K., Andersson P. G., Tetrahedron, 58, 4665—4668
1H), 2.40—2.60 (m, 2H), 1.50—1.65 (m, 2H), 1.10—1.34 (m, 2H); 13C-
(2002).
NMR (CDCl3, 75 MHz) d: 144.9, 143.1, 138.4, 127.2, 127.1, 125.0, 122.8, 28) Sdergren M. J., Bertlisson S. K., Andersson P. G., J. Am. Chem. Soc.,
115.9, 49.5, 49.2, 28.0, 27.8; IR (KBr) cmꢄ1: 2943, 2867, 1593, 1571, 1463,
1365, 1319, 1286, 1255, 1219, 1127, 1038, 751; HR-MS (m/z): Calcd for
C20H20N2S: 320.1348, Found: 320.1452.
122, 6610—6618 (2000).
29) Esmieu W. R., Worden S. M., Catterick D., Wilson C., Hayes C. J.,
Org. Lett., 10, 3045—3048 (2008).
Typical Procedure for the Preparation of 1 by Hydrogenation of 11 30) Shapiro R. H., Lipton M. F., Kolonto K. J., Buswell R. L., Capuano L.
A methanolic solution (2 ml) of 11 (35 mg, 0.11 mmol) containing 3.5 mg A., Tetrahedron Lett., 16, 1811—1814 (1975).
(10%, w/w) was first degassed at room temperature using a standard water 31) Adlington R. M., Barret A. G. M., Acc. Chem. Res., 16, 55—59
pump. The flask atmosphere was then kept under an atmosphere of H2 (1983).
(1 atm) by charging a balloon containing hydrogen on the top of the flask. 32) Nilsson B. M., Sundquist S., Johansson G., Nordvall G., Glas G., Nil-
The reaction mixture is stirred at room temperature for 24 h. After total con- vebrant L., Hacksell U., J. Med. Chem., 38, 473—487 (1995).
version of the starting material, the catalyst was filtered off, and the solvent 33) Norvall G., Sundquist S., Nilvebrant L., Hacksell U., Bioorg. Med.
was evaporated to dryness. The crude slurry was purified by column chro- Chem. Lett., 4, 2837—2840 (1994).
matography on neutral alumina eluting with ethyl acetate yielding 32 mg 34) Hua D. H., Huang X., Chen Y., Battina S. K., Tamura M., Noh S. K.,
(91%) of analytically pure rac-mequitazine 1.
Koo S. I., Namatame I., Tomoda H., Perchellet E. M., J. Org. Chem.,
69, 6065—6078 (2004).
Acknowledgments We are grateful to Les Instituts de Recherche Pierre 35) Nagano T., Kobayashi S., J. Am. Chem. Soc., 131, 4200—4201 (2009).
Fabre and the CNRS for financial support to VG.
36) Lu Z., Ma S., Angew. Chem. Int. Ed., 47, 258—297 (2008).
37) Trost B. M., Crawley M. L., Chem, Rev., 103, 2921—2944 (2003).
38) Byström S. E., Aslanian R., Bäckvall J.-E., Tetrahedron Lett., 26,
1749—1752 (1985).
References and Notes
1) Deceased.
2) Federsel H.-J., Acc. Chem. Res., 42, 671—680 (2009).
3) Federsel H.-J., Drug Discov. Today, 11, 966 (2006).
4) Trost B. M., Acc. Chem. Res., 35, 695—705 (2002).
5) Frankenpohl J., Hoffmann H. M. R., J. Org. Chem., 65, 3982—3996
(2000).
6) Yang D., Soulier J.-L., Sicsic S., Marte-Allainmat M., Bremont B.,
Croci T., Cardamone R., Aureggi G., Langlois M., J. Med. Chem., 40,
608—621 (1997).
39) Inoue Y., Taguchi M., Toyofuku M., Hashimoto H., Bull. Chem. Soc.
Jpn., 57, 3021—3022 (1984).
40) Hsu Y.-C., Gan K.-H.,. Yang S.-C., Chem. Pharm. Bull., 53, 1266—
1269 (2005).
41) Weihofen R., Tverskoy O., Helmchen G., Angew. Chem. Int. Ed., 45,
5546—5549 (2006).
42) Control experiments were also performed without Pd(PPh3)4 catalyst
with 9 and 10 and were completely ineffective at room temperature
even after prolonged reaction time.
7) Bromide S. M., Brown F., Cassidy F., Clark M. S. G., Dabbs S.,

 Gonnot, Vanessa
Gonnot, Vanessa
 Nicolas, Marc
Nicolas, Marc
 Mioskowski, Charles
Mioskowski, Charles
 Baati, Rachid
Baati, Rachid