
Journal of Medicinal Chemistry p. 6658 - 6661 (2004)
Update date:2022-08-11
Topics:
 Lombardo, Louis J.
Lombardo, Louis J.
 Lee, Francis Y.
Lee, Francis Y.
 Chen, Ping
Chen, Ping
 Norris, Derek
Norris, Derek
 Barrish, Joel C.
Barrish, Joel C.
 Behnia, Kamelia
Behnia, Kamelia
 Castaneda, Stephen
Castaneda, Stephen
 Cornelius, Lyndon A. M.
Cornelius, Lyndon A. M.
 Das, Jagabandhu
Das, Jagabandhu
 Doweyko, Arthur M.
Doweyko, Arthur M.
 Fairchild, Craig
Fairchild, Craig
 Hunt, John T.
Hunt, John T.
 Inigo, Ivan
Inigo, Ivan
 Johnston, Kathy
Johnston, Kathy
 Kamath, Amrita
Kamath, Amrita
 Kan, David
Kan, David
 Klei, Herbert
Klei, Herbert
 Marathe, Punit
Marathe, Punit
 Pang, Suhong
Pang, Suhong
 Peterson, Russell
Peterson, Russell
 Pitt, Sidney
Pitt, Sidney
 Schieven, Gary L.
Schieven, Gary L.
 Schmidt, Robert J.
Schmidt, Robert J.
 Tokarski, John
Tokarski, John
 Wen, Mei-Li
Wen, Mei-Li
 Wityak, John
Wityak, John
 Borzilleri, Robert M.
Borzilleri, Robert M.
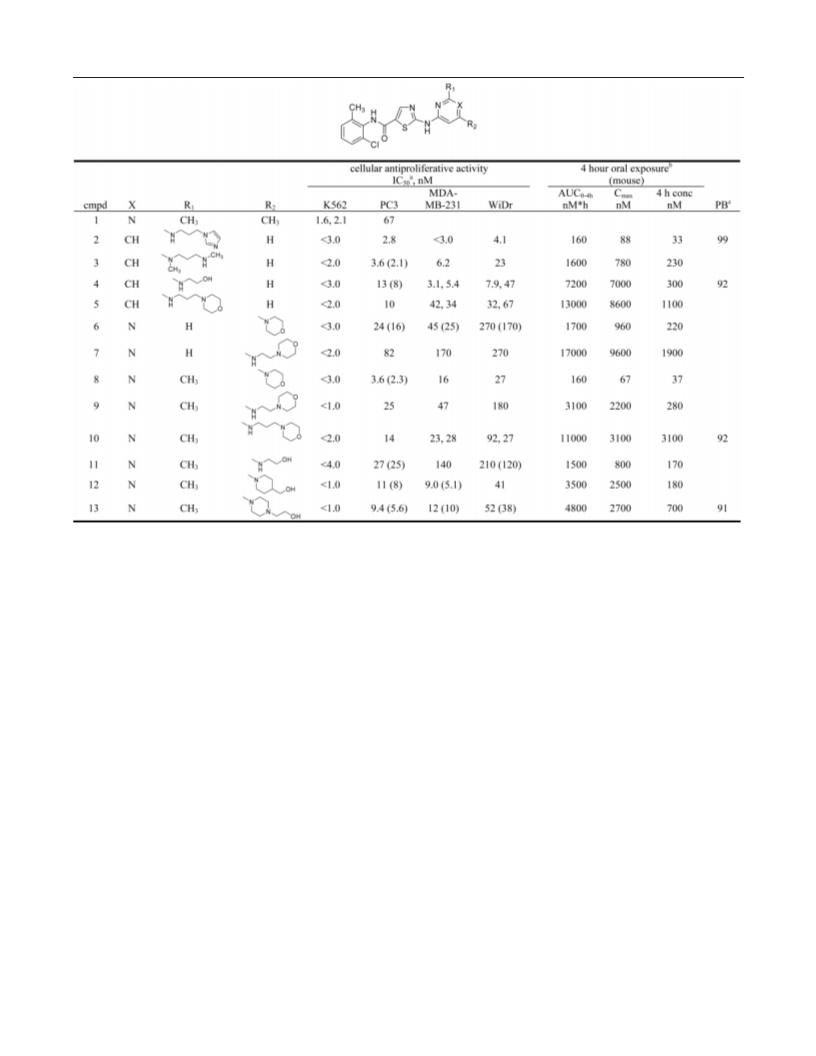
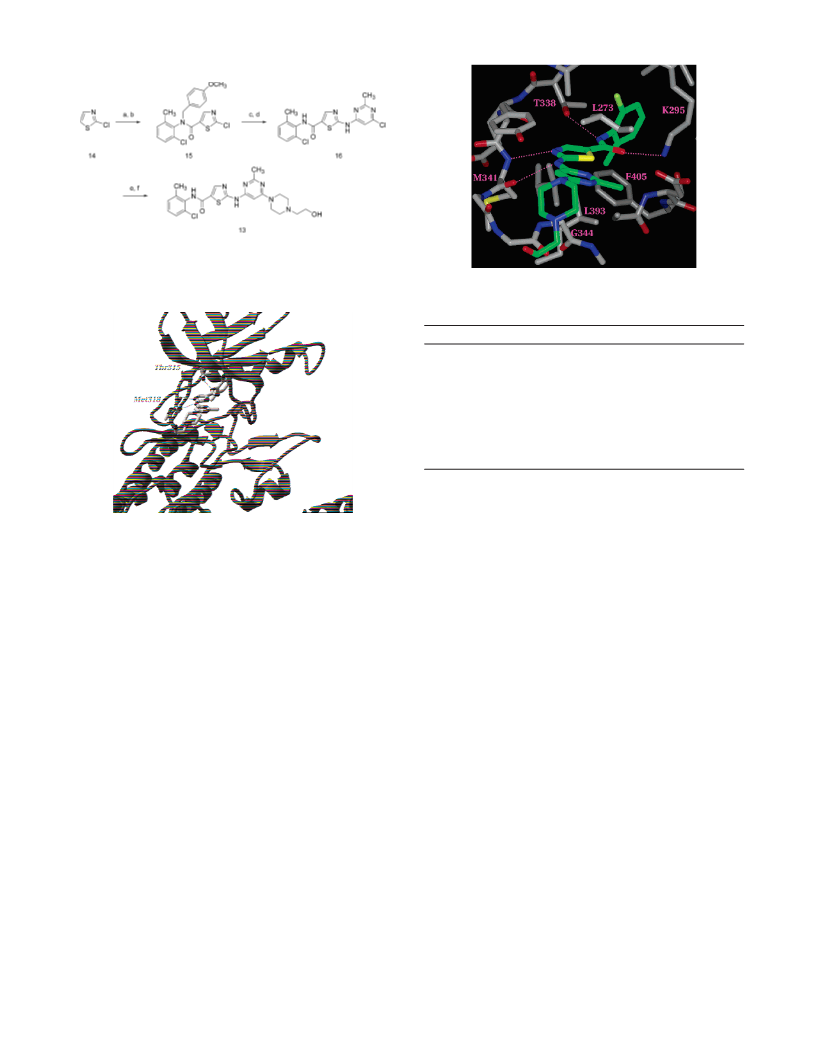
A series of substituted 2-(aminopyridyl)- and 2-(aminopyrimidinyl)thiazole- 5-carboxamides was identified as potent Src/Abl kinase inhibitors with excellent antiproliferative activity against hematological and solid tumor cell lines. Compound 13 was orally active in a K562 xenograft model of chronic myelogenous leukemia (CML), demonstrating complete tumor regressions and low toxicity at multiple dose levels. On the basis of its robust in vivo activity and favorable pharmacokinetic profile, 13 was selected for additional characterization for oncology indications.
View More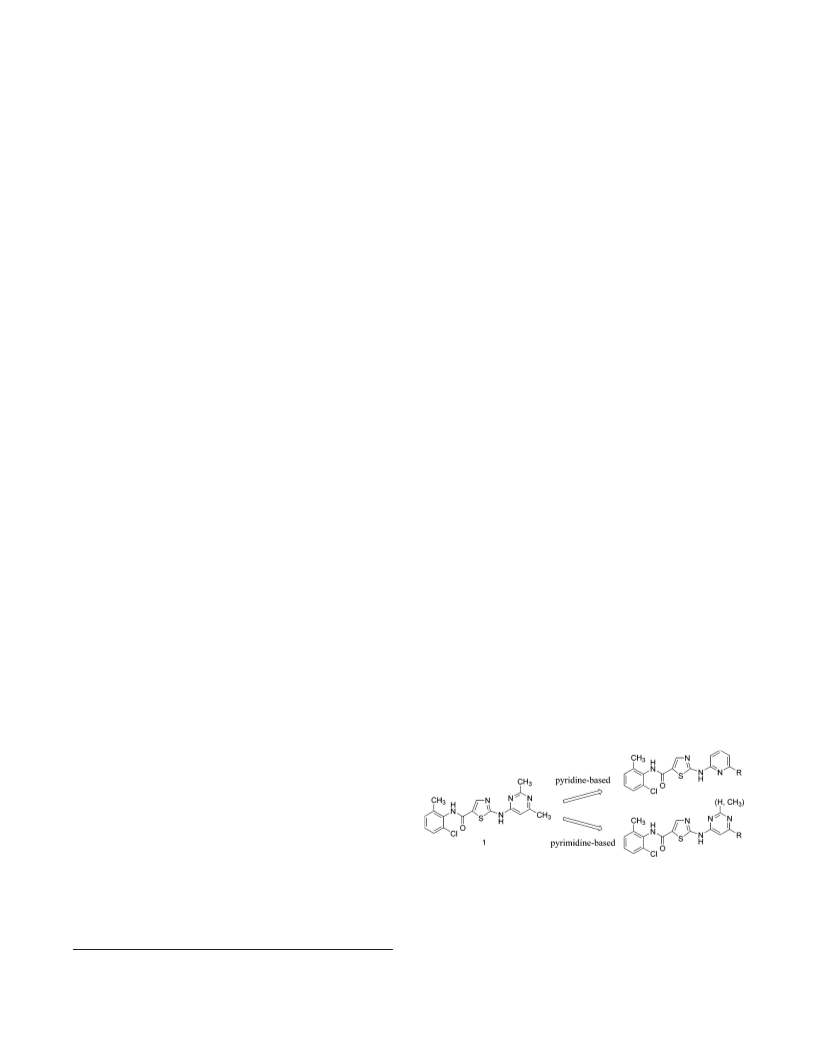

Improve Medical Technology(Nanxiong) Co., Ltd
Contact:86-751-3836997
Address:No.33, Pingan First Road, Fine Chemical Industry Base, Nanxiong City, Shaoguan, Guangdong, China
Liao Cheng All Win Chemicals Co.,LTD
Contact:86+0635-2991582
Address:Room 402,Unit 1,No.27 building.Zhong tong shi dai haoyuan,liaocheng city,Shan dong Province.China
Tianjin Te-An Chemtech Co., Ltd.
Contact:+86-22-65378638
Address:A5-8, No.80 Haiyun Street, TEDA
Contact:+86-838-5655598
Address:Guanghan Nanfeng Industrial Zone
Yuan Shi(SuQian)Biotechnology Co.,Ltd
website:http://www.yuanshibio.com
Contact:+86-527-84226672
Address:jiangsu suqian
Doi:10.1271/bbb.64.1988
(2000)Doi:10.1016/S0040-4020(98)00103-3
(1998)Doi:10.1246/cl.1982.219
(1982)Doi:10.1021/jo300879r
(2012)Doi:10.1021/jo980189l
(1998)Doi:10.1016/j.electacta.2021.138528
(2021)