Full Paper
doi.org/10.1002/ejic.202000670
EurJIC
European Journal of Inorganic Chemistry
ent temperature, the reaction mixture was concentrated and
washed with CH2Cl2/diethyl ether. The product B was afforded as a
white solid in 88 % yield.
gram in energy technology (No. 20154010200820) of the Korea
institute of Energy Technology Evaluation and Planning (KETEP),
which receives financial resources from the Ministry of Trade,
Industry & Energy, Republic of Korea.
Synthetic Procedure for the Synthesis of B′: A mixture of A (1.8 g,
3.4 mmol) and 1-ethylimidazole (1.3 mL, 13.4 mmol) was refluxed in
MeCN (0.05 M, 67.2 mL) for 18 hours. After cooled down to ambient
Keywords: Carbene ligands · Iridium · Homogeneous
catalysis · Hydrogen · Lactate · Glycerol
temperature, the reaction mixture was concentrated and washed
with CH2Cl2/diethyl ether. The product B′ was afforded as a white
solid in 87 % yield.
[1] L. Barelli, G. Bidini, F. Gallorini, S. Servili, Energy 2008, 33, 554–570.
[2] H. Ahmad, S. K. Kamarudin, L. J. Minggu, M. Kassim, Renewable Sustaina-
ble Energy Rev. 2015, 43, 599–610.
[3] M. Trincado, D. Banerjee, H. Grützmacher, Energy Environ. Sci. 2014, 7,
2464–2503.
[4] E. Alberico, M. Nielsen, Chem. Commun. 2015, 51, 6714–6725.
[5] G. Bottari, K. Barta, Recycl. Catal. 2015, 2, 70–77.
[6] R. H. Crabtree, ACS Sustainable Chem. Eng. 2019, 7, 15845–15853.
[7] Y. Li, M. Nielsen, B. Li, P. H. Dixneuf, H. Junge, M. Beller, Green Chem.
2015, 17, 193–198.
[8] W. A. Herrmann, M. Elison, J. Fischer, C. Köcher, G. R. J. Artus, Angew.
Chem. Int. Ed. Engl. 1995, 34, 2371–2374; Angew. Chem. 1995, 107, 2602.
[9] J. A. Mata, M. Poyatos, E. Peris, Coord. Chem. Rev. 2007, 251, 841–859.
[10] M. Poyatos, J. A. Mata, E. Peris, Chem. Rev. 2009, 109, 3677–3707.
[11] J. A. Mata, F. E. Hahn, E. Peris, Chem. Sci. 2014, 5, 1723–1732.
[12] E. Peris, Chem. Rev. 2018, 118, 9988–10031.
[13] E. Mas-Marzá, M. Poyatos, M. Sanaú, E. Peris, Inorg. Chem. 2004, 43, 2213–
2219.
[14] C. Tubaro, A. Biffis, E. Scattolin, M. Basato, Tetrahedron 2008, 64, 4187–
4195.
[15] A. Biffis, G. G. Lobbia, G. Papini, M. Pellei, C. Santini, E. Scattolin, C. Tubaro,
J. Organomet. Chem. 2008, 693, 3760–3766.
[16] C. E. Ellul, G. Reed, M. F. Mahon, S. I. Pascu, M. K. Whittlesey, Organometal-
lics 2010, 29, 4097–4104.
Synthetic Procedure for the Synthesis of 1 and 1′: Sodium
hydride (35 mg, 0.875 mmol) was dissolved in ethanol (4.4 mL) and
slowly added to a suspension of [Ir(COD)Cl]2 (144 mg, 0.22 mmol)
in ethanol (2.9 mL). The reaction mixture was stirred at room tem-
perature for 1 h and B (348 mg, 0.5 mmol) was added and stirred
at room temperature for 12 h. Then, the mixture was filtered, and
the filtrate was evaporated in vacuo. Potassium hexafluorophos-
phate (158 mg, 0.9 mmol) in DCM/EtOH (v:v, 1:1) was added and
stirred at room temperature for 1 h. The mixture was concentrated
and purified by column chromatography. Complex 1′ was obtained
as an orange solid with 26 % yield (eluent: CH2Cl2 and acetone),
and complex 1 was obtained with 23 % yield (eluent: CH2Cl2 and
MeOH).
Synthetic Procedure for the Synthesis of 2 and 2′: Sodium
hydride (35 mg, 0.875 mmol) was dissolved in ethanol (4.4 mL) and
added dropwise to
a suspension of [Ir(COD)Cl]2 (144 mg,
0.22 mmol) in ethanol (2.9 mL). The reaction mixture was stirred at
room temperature for 1 h and B′ (375 mg, 0.5 mmol) was added
and stirred at room temperature for 12 h. Then, the mixture was
filtered, and the filtrate was evaporated in vacuo. Potassium hexa-
fluorophosphate (158 mg, 0.9 mmol) in DCM/EtOH (v:v, 1:1) was
added and stirred at room temperature for 1 h. The mixture was
concentrated and purified by column chromatography. Complex 2′
was obtained as an orange solid with 27 % yield (eluent: CH2Cl2
and acetone), and complex 2 was obtained with 11 % yield (eluent:
CH2Cl2 and MeOH).
[17] M. Raynal, C. S. J. Cazin, C. Vallée, H. Olivier-Bourbigou, P. Braunstein,
Dalton Trans. 2009, 3824–3832.
[18] M.-M. Gan, J.-Q. Liu, L. Zhang, Y.-Y. Wang, F. E. Hahn, Y.-F. Han, Chem. Rev.
2018, 118, 9587–9641.
[19] H.-Y. Kuo, Y.-H. Liu, S.-M. Peng, S.-T. Liu, Organometallics 2012, 31, 7248–
7255.
Synthetic Procedure for 3: Complex 2 (40 mg, 0.043 mmol) was
dissolved in CH2Cl2 (1.4 mL) under CO (1 atm) and stirred for 24 h
at room temperature. The solvent was removed in vacuo. The yellow
solid was washed with diethyl ether to afford 3 with 93 % yield.
[20] A. E. Díaz-Álvarez, V. Cadierno, Appl. Sci. 2013, 3, 55–69.
[21] D. Wang, D. Astruc, Chem. Rev. 2015, 115, 6621–6686.
[22] L. S. Sharninghausen, J. Campos, M. G. Manas, R. H. Crabtree, Nat. Com-
mun. 2014, 5, 5084–5092.
[23] Z. Sun, Y. Liu, J. Chen, C. Huang, T. Tu, ACS Catal. 2015, 5, 6573–6578.
[24] Z. Lu, I. Demianets, R. Hamze, N. J. Terrile, T. J. Williams, ACS Catal. 2016,
6, 2014–2017.
Synthetic Procedure for 3′: Complex 2′ (41 mg, 0.033 mmol) was
dissolved in CH2Cl2 (1.4 mL) under CO (1 atm) and stirred for 24 h
at room temperature. The solvent was removed in vacuo. The yellow
solid was washed with diethyl ether to afford 3′ with 91 % yield.
[25] M. Finn, J. A. Ridenour, J. Heltzel, C. Cahill, A. Voutchkova-Kostal, Organo-
metallics 2018, 37, 1400–1409.
[26] W. Oberhauser, C. Evangelisti, A. Liscio, A. Kovtun, Y. Cao, F. Vizza, J. Catal.
2018, 368, 298–305.
[27] L. Li, F. Shen, R. L. Smith, X. Qi, Green Chem. 2017, 19, 76–81.
[28] Z. Lu, V. Cherepakhin, I. Demianets, P. J. Lauridsen, T. J. Williams, Chem.
Commun. 2018, 54, 7711–7724.
[29] Y.-J. Cheong, K. Sung, S. Park, J. Jung, H.-Y. Jang, ACS Sustainable Chem.
Eng. 2020, 8, 6972–6978.
[30] Y. Wang, J. Zhou, X. Guo, RSC Adv. 2015, 5, 74611–74628.
[31] P. Buchwalter, J. Rosé, P. Braunstein, Chem. Rev. 2015, 115, 28–126.
[32] W.-H. Wang, J. F. Hull, J. T. Muckerman, E. Fujita, T. Hirose, Y. Himeda,
Chem. Eur. J. 2012, 18, 9397–9404.
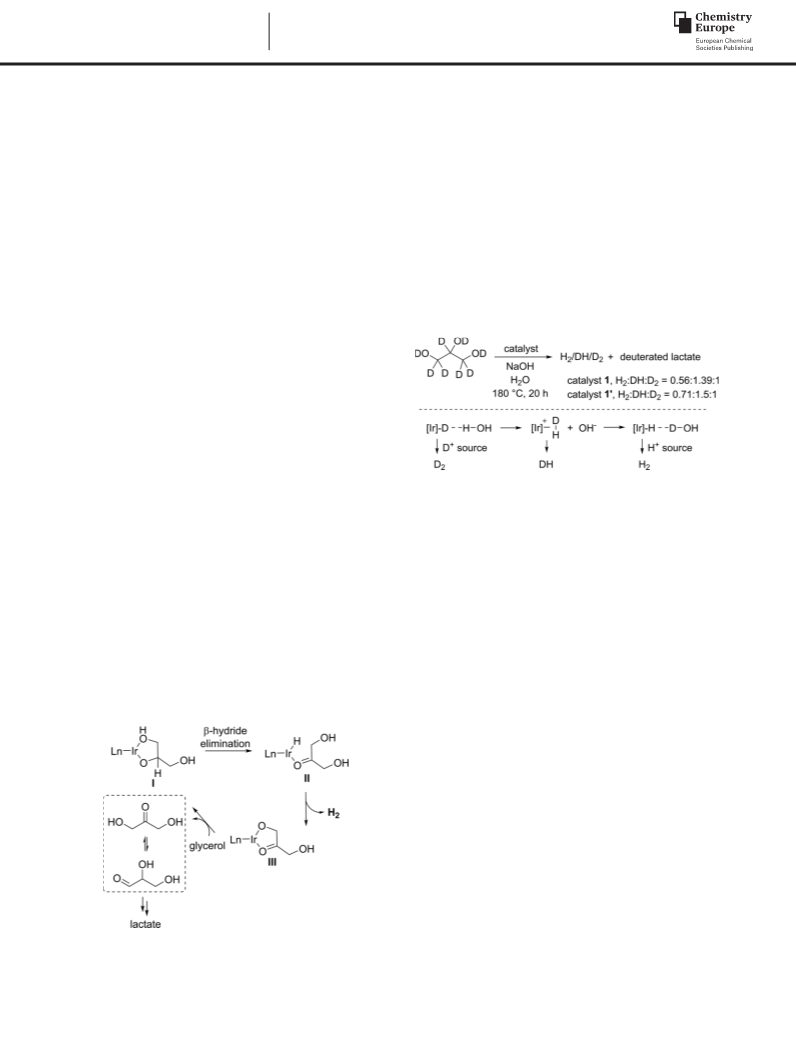
Catalytic Procedure for Dehydrogenation of Glycerol: The cata-
lyst (for monometallic complexes 0.02 μmol, for bimetallic com-
plexes 0.01 μmol), base (12 mmol), H2O (0.5 mL), and glycerol
(2.66 g, 28.9 mmol) were added into an autoclave. The mixture was
heated at 180 °C for 20 h. Then, the reaction mixture was cooled
down to ambient temperature, and diluted with H2O. The TONs
of lactates were calculated by 1H NMR spectroscopy in D2O using
isonicotinic acid as an internal standard.
Acknowledgments
This study was supported by National Research Foundation
(NRF) programs (C1 gas refinery No. 2015M3D3A1A01065436
and No. 2019R1A2C1084021) and the human resources pro-
[33] M. C. Carrión, M. Ruiz-Castaneda, G. Espino, C. Aliende, L. Santos, A. M.
Rodríguez, B. R. Manzano, F. A. Jalón, A. Lledós, ACS Catal. 2014, 4, 1040–
1053.
Received: July 14, 2020
Eur. J. Inorg. Chem. 0000, 0–0
5
© 2020 Wiley-VCH GmbH

 Cheong, Yeon-Joo
Cheong, Yeon-Joo
 Sung, Kihyuk
Sung, Kihyuk
 Kim, Jin-A
Kim, Jin-A
 Kim, Yu Kwon
Kim, Yu Kwon
 Jang, Hye-Young
Jang, Hye-Young