‘‘brush-like’’ TriDAP-PS. The topographic structures together
with adsorption and desorption of the polymer film can be readily
controlled by appropriate choice of solvent systems. This study
provides new directions in corresponding fields in terms of
renewability based on the reversible nature of hydrogen bonding,
and orthogonal modification associated with the specific recogni-
tion interaction. Furthermore, the Tri-DAP unit can be easily
incorporated into other functional polymers to impart desired
mechanical, optical, electronic or biological properties onto various
substrates. The investigations are underway and will be reported in
due course.
This research was supported by the National Science
Foundation (US) CHE-0518487. We thank Prof. Thomas J.
McCarthy’s group for help in surface analysis. T. B. N acknowl-
edges NSERC (Canada) for a postdoctoral fellowship.
Notes and references
1 (a) S. T. Milner, Science, 1991, 251, 905; (b) B. Zhao and W. J. Brittain,
Prog. Polym. Sci., 2000, 25, 677; (c) I. Luzinov, S. Minko and
V. V. Tsukruk, Prog. Polym. Sci., 2004, 29, 635; (d) T. P. Russell,
Science, 2002, 297, 964; (e) Y. Nagasaki and K. Kataoka, Trends Polym.
Sci., 1996, 4, 59; (f) Y. Liu, L. Mu, B. Liu and J. Kong, Chem. Eur. J.,
2005, 11, 2622.
2 (a) S. Khongtong and G. S. Ferguson, J. Am. Chem. Soc., 2002, 124,
7254; (b) R. R. Netz and D. Andelman, Phys. Rep., 2003, 380, 1; (c)
K. Ichimura, S. K. Oh and M. Nakagawa, Science, 2000, 288, 1624; (d)
D. Julthongpiput, Y. H. Lin, J. Teng, E. R. Zubarev and V. V. Tsukruk,
J. Am. Chem. Soc., 2003, 125, 15912.
3 (a) G. Sharma and M. Ballauff, Macromol. Rapid Commun., 2004, 25,
547; (b) S. Minko, M. Muller, M. Motornov, M. Nitschke, K. Grundke
and M. Stamm, J. Am. Chem. Soc., 2003, 125, 3896; (c) Y. Ito,
Y. Ochiai, Y. S. Park and Y. Imanishi, J. Am. Chem. Soc., 1997, 119,
1619; (d) M. Ornatska, S. E. Jones, R. R. Naik, M. O. Stone and
V. V. Tsukruk, J. Am. Chem. Soc., 2003, 125, 12722; (e) M. C. Woodle,
Adv. Drug Deliv. Rev., 1998, 32, 139.
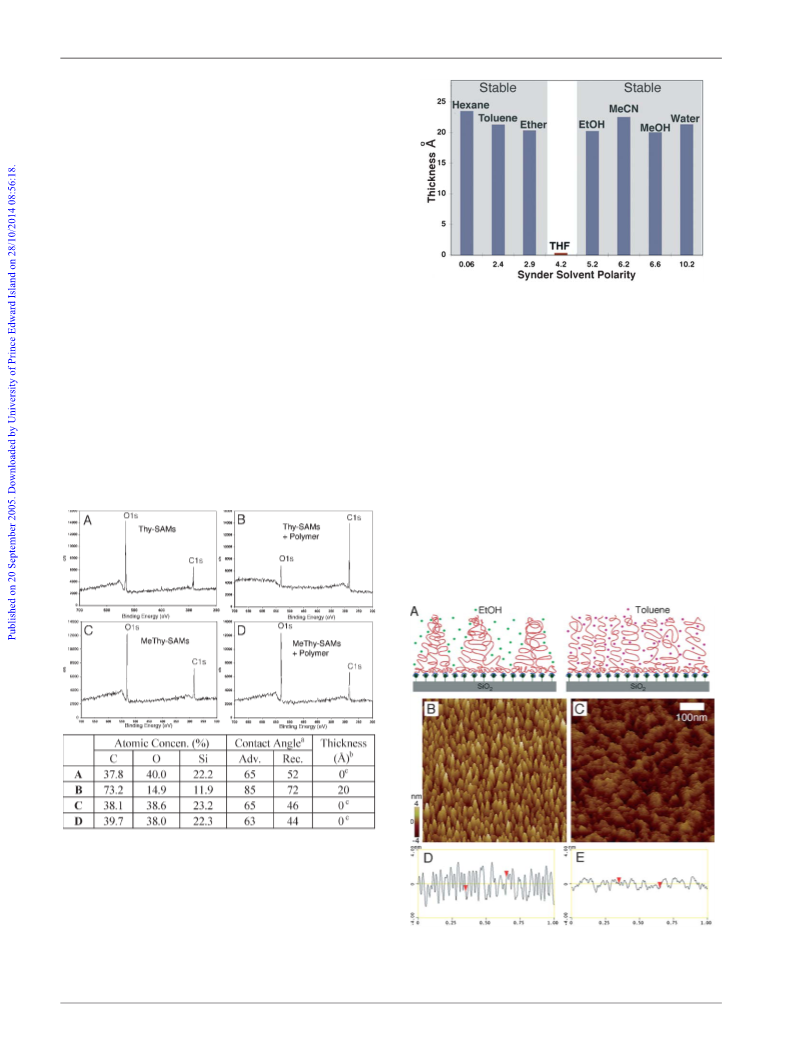
Fig. 5 Schematic illustration (A), AFM height profiles (B), and thickness
and contact angle changes (C), following (a) SAMs, and subsequent (b)
adsorption, (c) desorption, (d) readsorption, (e) redesorption of polymer
layer. See ESI{ for more detailed AFM information, including the second
adsorption–desorption cycles.
4 R. Chen, R. P. J. Bronger, P. C. J. Kamer, P. van Leeuwen and
J. N. H. Reek, J. Am. Chem. Soc., 2004, 126, 14557.
Fig. S6 in ESI{) The relative intensity of Si 2s and O 1s, compared
to C 1s, increased after the surface was treated with ethanol due to
the collapse of the uniform PS layer, which allowed the emitted Si
and O photoelectrons (from the substrate) more opportunity to
escape the polymer layer.
5 (a) B. Zhao, W. J. Brittain, W. S. Zhou and S. Z. D. Cheng,
J. Am. Chem. Soc., 2000, 122, 2407; (b) B. Zhao, R. T. Haasch and
S. MacLaren, J. Am. Chem. Soc., 2004, 126, 6124; (c) M. Husemann,
D. Mecerreyes, C. J. Hawker, J. L. Hedrick, R. Shah and
N. L. Abbott, Angew. Chem., Int. Ed., 1999, 38, 647; (d) X. G. Liu,
S. W. Guo and C. A. Mirkin, Angew. Chem., Int. Ed., 2003, 42,
4785.
An important attribute of supramolecular chemistry is the
reversible binding process that can be employed to recover the
original surface. Although the modified surfaces were stable under
most solvent conditions, rinsing with THF (a good solvent for PS
and a competitive solvent for H-bonding) can efficiently remove
the adsorbed polymer layers. The resulting surfaces show the same
thickness as the original SAMs and the advancing contact angles
decreased from 83 to 65u as a result of the top layer switching from
the hydrophobic PS backbone to more hydrophilic Thy-surfaces.
AFM images indicate the removal of the polymer layer as the
surface becomes smooth and all the micelles were erased,
displaying similar features to the original SAMs. This renewable
modification displays good cyclability as the regenerated surface
was found to perform similar functions multiple times without
affecting its recognition ability (Fig. 5).
6 (a) G. Fytas, S. H. Anastasiadis, R. Seghrouchni, D. Vlassopoulos,
J. B. Li, B. J. Factor, W. Theobald and C. Toprakcioglu, Science, 1996,
274, 2041; (b) P. Brandani and P. Stroeve, Macromolecules, 2003, 36,
9492.
7 (a) L. J. Prins, D. N. Reinhoudt and P. Timmerman, Angew. Chem., Int.
Ed., 2001, 40, 2383; (b) R. P. Sijbesma, F. H. Beijer, L. Brunsveld,
B. J. B. Folmer, J. Hirschberg, R. F. M. Lange, J. K. L. Lowe and
E. W. Meijer, Science, 1997, 278, 1601; (c) F. H. Beijer, H. Kooijman,
A. L. Spek, R. P. Sijbesma and E. W. Meijer, Angew. Chem., Int. Ed.,
1998, 37, 75; (d) E. A. Fogleman, W. C. Yount, J. Xu and S. L. Craig,
Agew. Chem., Int. Ed., 2002, 41, 4026.
8 J. M. Pollino, L. P. Stubbs and M. Weck, J. Am. Chem. Soc., 2004, 126,
563.
9 (a) T. B. Norsten, E. Jeoung, R. J. Thibault and V. M. Rotello,
Langmuir, 2003, 19, 7089; (b) A. Sanyal, T. B. Norsten, O. Uzun and
V. M. Rotello, Langmuir, 2004, 20, 5958; (c) A. Ulman, Chem. Rev.,
1996, 96, 1533.
10 J. Dao, D. Benoit and C. J. Hawker, J. Polym. Sci. Polym. Chem., 1998,
36, 2161.
11 E. Zhulina and A. C. Balazs, Macromolecules, 1996, 29, 2667.
In summary, we demonstrated the reversible functionaliza-
tion of silica surfaces via specific H-bonding interaction using
This journal is ß The Royal Society of Chemistry 2005
Chem. Commun., 2005, 5157–5159 | 5159

 Xu, Hao
Xu, Hao
 Norsten, Tyler B.
Norsten, Tyler B.
 Uzun, Oktay
Uzun, Oktay
 Jeoung, Eunhee
Jeoung, Eunhee
 Rotello, Vincent M.
Rotello, Vincent M.