A.R. Bayguzina, et al.
JournalofFluorineChemistry226(2019)109346
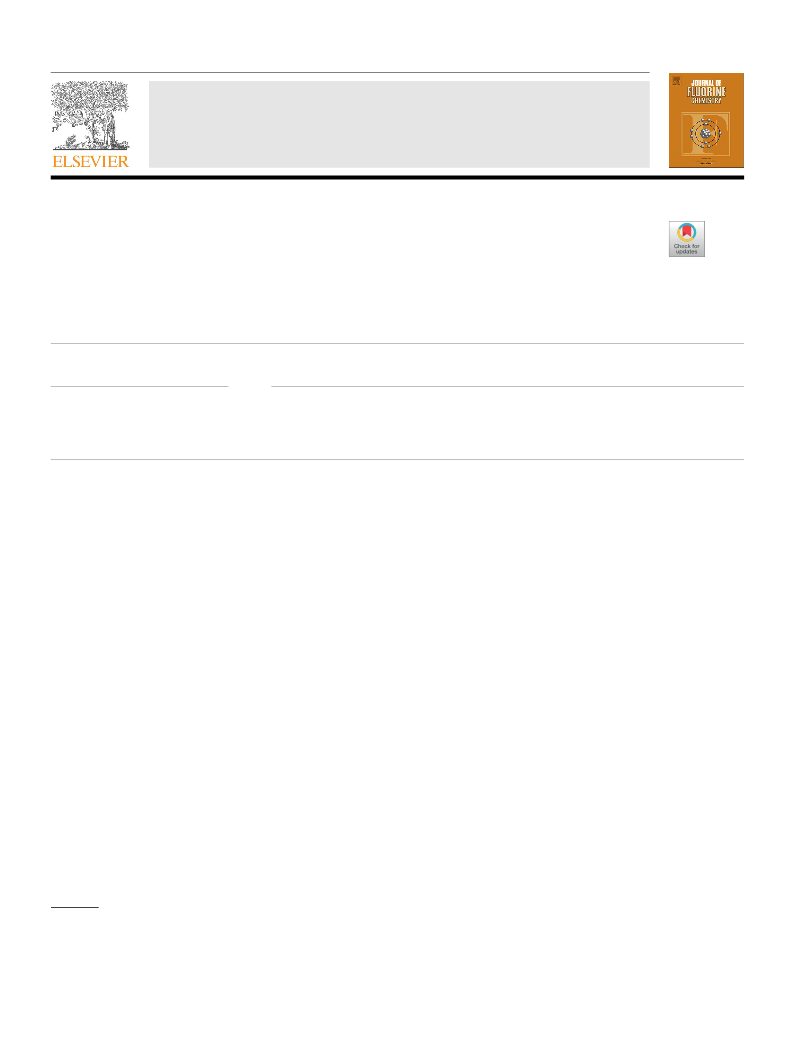
Table 5
4.2.2. 4-Fluorobenzyl chloride (1а)
Experimental results on the synthesis of mono- and difluorobenzyl chlorides 1a-
6a by chlorination of mono- and difluorotoluenes 1–6 with t-BuOCl catalyzed
by FeCl2·4H2O (conditions: 20 °C, 3 h).a.
Yield: 0.88 g (67%) (Method A), 1.69 g (65%) (Method B), colorless
liquid: bp 76–78 °C/20 mm (Lit. data: bp 62 °C/10 mm [18]). The NMR
spectroscopic data agree with those described in ref. [4,8,9]. 1H NMR
3
(400.1 MHz, CDCl3):
δ
4.6 (s, 2H, CH2), 7.1 (t, 2H, JHH
≈
Entry
(Di)fluorotoluene 1-6
Conversion of 1-6, %
Yield of 1a-6а, %
3JHF = 8.5 Hz, H3,5), 7.4 (m, 2H, H2,6). 13C NMR (100.6 MHz, CDCl3): δ
45.5 (s, CH2), 115.7 (d, JCF = 21,7 Hz, C3,5), 130.5 (d, JCF = 8,3 Hz,
2
3
1
2
3
4
5
6
p-Fluorotoluene 1
70
58
59
55
51
61
65
31
36
33
24
36
m-Fluorotoluene 2
o-Fluorotoluene 3
4
1
C2,6), 133.5 (d, JCF = 3.3 Hz, C1), 162.6 (d, JCF = 247.4 Hz, C4). 19F
NMR (376.4 MHz, CDCl3): δ -113.2 (s, 1 F, F4). MS (EI, 70 eV) m/z (%):
144 [M]+ (17), 110 (7), 109 (100), 107 (9), 95 (6), 83 (15).
2,4-Difluorotoluene 4
2,5-Difluorotoluene 5
2,6-Difluorotoluene 6
Difluoroarylmethanes (1b, c). Yield: 0.69 g (70%) (Method A, without
HCONH2) (The isomer ratio 1b:1c
= 2:1), colorless liquid: bp
aMolar ratio [FeCl2·4H2O]:[1–6]:[t-BuOCl] = 1:100:300.
115–117 °C/2 mm.
FeCl2·4H2O catalyst.
4.2.3. 1-Fluoro-2-(4-fluorobenzyl)-4-methylbenzene (1b)
1H NMR (500.2 MHz, CDCl3): δ 2.4 (s, 3H, CH3), 4.0 (s, 2H, CH2),
6.7–7.7 (m, 7HA,B). 13C NMR (125.8 MHz, CDCl3): δ 20.8 (s, CH3), 34.2
4. Experimental
2
2
(s, CH2), 115.1 (d, JCF = 22.9 Hz, CB6), 115.3 (d, JCF = 21.1 Hz,
CA3,5), 128.6 (d, JCF = 7.6 Hz, CB5), 130.2 (d, JCF = 7.4 Hz, CA
,
3
3
2,6
4.1. General
CB3), 131.4 (s, CB2), 133.6 (s, CA1), 135.8 (s, CA1), 159.2 (d,
1H, 13C and 19F NMR spectra were recorded in CDCl3 using a Bruker-
Avance-400 NMR spectrometer (400.1, 100.6 and 376.4 MHz, respec-
tively and a Bruker-Avance-500 NMR spectrometer (500.2, 125.8 and
470.6 MHz, respectively). Chemical shifts (δ) are given in ppm and
referenced relative to TMS. Mass spectra were run on were acquired
with a Shimadzu GCMS-QP2010Plus GC/MS spectrometer (an SPB-5
capillary column, 30 m ×0.25 mm, helium as a carrier gas, temperature
programming from 40 to 300 °C at 8 °C/min, evaporation temperature
280 °C, temperature of the ion source 200 °C, ionization energy 70 eV).
Chromatographic analysis was performed on a Shimadzu GC-9A, GC-
2014 instrument [2 m ×3 mm column, silicone SE-30 (5%) on
Chromaton N-AW-HMDS as the stationary phase, temperature pro-
gramming from 50 to 270 °C at 8 °C/min, helium as the carrier gas
(47 mL/min)].
1JCF = 242.8 Hz, CB1), 161.5 (d, JCF = 244.0 Hz, CA4). 19F NMR
1
(470.6 MHz, CDCl3): δ -117.0 (s, 1 F), -123.2 (s, 1 F), FB and FA4. MS
1
(EI, 70 eV) m/z (%): 218 [M]+ (100), 217 (17), 203 (97), 201 (25), 183
(25), 109 (25).
4.2.4. 4-Fluoro-2-(4-fluorobenzyl)-1-methylbenzene (1c)
1H NMR (500.2 MHz, CDCl3): δ 2.3 (s, 3H, CH3), 3.9 (s, 2H, CH2),
6.7–7.7 (m, 7HA,B). 13C NMR (125.8 MHz, CDCl3): δ 19.0 (s, CH3), 38.7
2
2
(s, CH2), 115.3 (d, JCF = 21.1 Hz, CA3,5), 115.4 (d, JCF = 21.0 Hz,
2
3
CB5), 127.5 (d, JCF = 16.0 Hz, CB3), 128.6 (d, JCF = 7.6 Hz, CB6),
3
130.2 (d, JCF = 7.4 Hz, CA2,6), 131.6 (s, CB1), 132.0 (s, CA1), 135.2 (s,
1
1
CB2), 161.4 (d, JCF = 243.9 Hz, CB4), 161.5 (d, JCF = 244.0 Hz, CA4).
19F NMR (470.6 MHz, CDCl3): δ -116.9 (s, 1 F), -117.5 (s, 1 F), FA and
4
FB4. MS (EI, 70 eV) m/z (%): 218 [M]+ (100), 217 (17), 203 (97), 201
To identify compounds 1a-6a and 1b, c combined samples obtained
in experiments 3–6 were used.
(25), 183 (25), 109 (25).
4.2.5. 3-Fluorobenzyl chloride (2a)
4.2. Preparation of mono- and difluorobenzyl chlorides
4.2.1. General procedures
Yield: 0.46 g (35%) (Method A), 0.81 g (31%) (Method B), colorless
liquid: bp 68–69 °C/14 mm (Lit. data: bp 60 °C/10 mm [18]). 1H NMR
(400.1 MHz, CDCl3): δ 4.6 (s, 2H, CH2), 7.0 (dd, 1H, JHF = 9.0 Hz,
3
Method A. The reactions were carried out in
a
glass tube
3JHH = 8.0 Hz, H4), 7.1 (d, 1H, JHF = 9.0 Hz, H2), 7.2 (d, 1H,
4
3
(V = 10 mL) placed into a stainless steel autoclave (V = 17 mL) at a
controlled heating.
3JHH = 8.0 Hz, H6), 7.3 (t, 1H, JHF = 8.0 Hz, JHH = 8.0 Hz, H5). 13C
2
NMR (100.6 MHz, CDCl3): δ 45.3 (s, CH2), 115.3 (d, JCF = 20.9 Hz,
2
4
The tube under argon was charged with FeCl2·4H2O (0.09 mmol,
0.018 g) and formamide (4.5 mmol, 0.20 g), the mixture was heated for
5 min, and then tetrachloromethane (18 mmol, 2.76 g), methanol
(4.5 mmol, 0.14 g), and fluorotoluene 1–3 (9 mmol, 1.00 g) (or di-
fluorotoluene 4–6 (9 mmol, 1.15 g)) were added. The sealed tube was
placed into an autoclave, and the autoclave was sealed and heated for
6 h at 180 оC. After completion of the reaction, the autoclave was
cooled down to room temperature, the tube was opened, and the re-
action mixture was filtered through a paper filter. The solvent was
distilled off. The target product was separated from the initial mono- or
difluorotoluenes by vacuum distillation.
C4), 115.5 (d, JCF = 21.9 Hz, C2), 124.1 (d, JCF = 3.0 Hz, C6), 130.3
(d, JCF = 8.2 Hz, C5), 139.8 (d, JCF = 7.4 Hz, C1), 162.8 (d,
1JCF = 246.7 Hz, C3). 19F NMR (376.4 MHz, CDCl3): δ -112.5 (s, 1 F, F3).
MS (EI, 70 eV) m/z (%): 144 [M]+ (25), 109 (100), 107 (11), 83 (25),
63 (7), 57 (9).
3
3
4.2.6. 2-Fluorobenzyl chloride (3a)
Yield: 0.40 g (30%) (Method A), 0.94 g (36%) (Method B), colorless
liquid: bp 72–73 °C/20 mm (Lit. data: bp 59 °C/10 mm [18]). 1H NMR
(500.2 MHz, CDCl3): δ 4.7 (s, 2H, CH2), 7.1–7.2 (m, 1H, H3), 7.2 (t, 1H,
3J = 8.0 Hz, H5), 7.3–7.4 (m, 1H, H4), 7.5 (t, 1H, 3J = 8.0 Hz, H6). 13C
3
Method B. The reactions were carried out in a glass reactor
(V = 20 mL) with continuous stirring at room temperature.
The tube under argon was charged with FeCl2·4H2O (0.18 mmol,
0.036 g), synthesized authentic tert-butyl hypochlorite [22] (54 mmol,
4–6 (2.30 g)). The mixture was stirred at r.t. for 3 h. After completion of
the reaction, the reaction mixture was filtered through a paper filter.
The target product was separated from the initial mono- or di-
fluorotoluene by vacuum distillation.
NMR (125.8 MHz, CDCl3): δ 39.5 (d, JCF = 4.6 Hz, CH2), 115.7 (d,
4
2JCF = 21.1 Hz, C3), 124.5 (d, JCF = 3.8 Hz, C5), 124.8 (d,
3
2JCF = 14.3 Hz, C1), 130.6 (d, JCF = 8.2 Hz, C6), 131.0 (d,
1
3JCF = 3.0 Hz, C4), 160.7 (d, JCF = 249.3 Hz, C2). 19F NMR
(470.6 MHz, CDCl3: δ -117.7 (s, 1 F, F2). MS (EI, 70 eV) m/z (%): 144
[M]+ (20), 110 (8), 109 (100), 107 (9), 83 (20), 57 (6).
4.2.7. 2,4-Difluorobenzyl chloride (4a)
Yield: 0.64 g (44%) (Method A), 0.97 g (33%) (Method B), colorless
liquid: bp 52–53 °C/8 mm (Lit. data: bp 60 °C/10 mm [18]). 1H NMR
(400.1 MHz, CDCl3): δ 4.6 (s, 2H, CH2), 6.8 (m, 2H, H5,6), 7.4 (m, 1H,
The structures of the products were proved by NMR and mass
spectrometry and by comparison with authentic samples and reference
data.
3
H3). 13C NMR (100.6 MHz, CDCl3): δ 38.7 (d, JCF = 4.0 Hz, CH2),
4

 Bayguzina, Alfiya R.
Bayguzina, Alfiya R.
 Gallyamova, Leysan I.
Gallyamova, Leysan I.
 Khalilov, Leonard M.
Khalilov, Leonard M.
 Khusnutdinov, Ravil I.
Khusnutdinov, Ravil I.