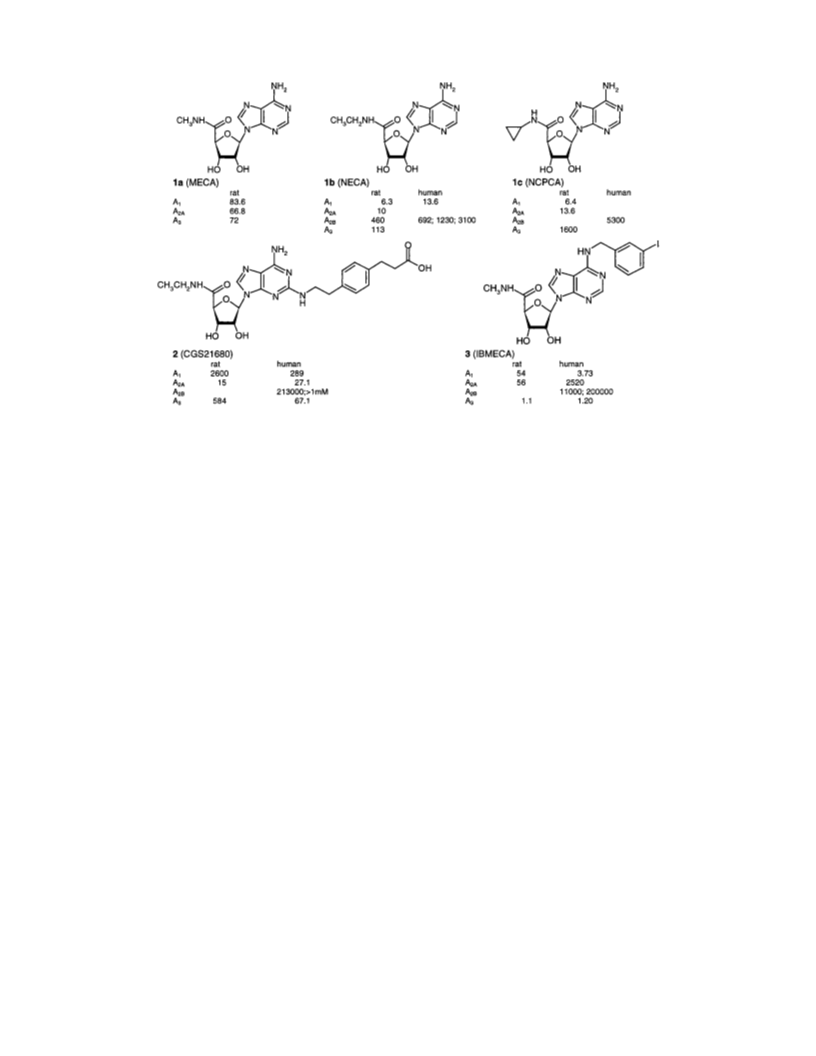
5′-N-Substituted Carboxamidoadenosines
J ournal of Medicinal Chemistry, 1999, Vol. 42, No. 8 1389
1H, H-8), 8.05 (s, 1H, H-2), 7.44 (s(b), 1H, NH), 6.18 (d, 1H, J
) 2 Hz, H-1′), 5.39, 5.37 (m + s, 2H, H-2′, H-3′), 4.72 (s, 1H,
H-4′), 2.57 (m, 3H, CH3), 1.63, 1.41 (2 × s, 2 × 3H, 2 × CH3,
isopropylidene); 13C NMR (CDCl3/CD3OD) 23.44 (CH3).
5′-N -E t h yl-2′,3′-O-isop r op ylid e n e ca r b oxa m id oa d e -
n osin e (6b). Compound 6b was synthesized according to
method A, starting with 2.00 g (6.2 mmol) of compound 5 and
accommodating the amounts of reagents. A solution of ethyl-
amine in water (70 wt %) was used. The compound was
isolated as an oil: yield 1.55 g (72%); Rf 0.50; 1H NMR (CDCl3)
δ 8.31 (s, 1H, H-8), 7.87 (s, 1H, H-2), 6.92 (s, 2H, NH2), 5.98
(s, 1H, H-1′), 5.40 (m, 2H, H-2′, H-3′), 4.71 (s, 1H, H-4′), 3.06
(m, 2H, CH2CH3), 1.63, 1.41 (2 × s, 2 × 3H, 2 × CH3,
isopropylidene), 0.88 (t, 3H, CH2CH3); 13C NMR (CDCl3) 31.85
(CH2CH3), 12.00 (CH2CH3).
NH), 6.68 (s(b), 2H, NH2), 6.15 (d, 1H, J ) 2.5 Hz, H-1′), 5.45
(m, 2H, H-2′, H-3′), 4.76 (s, 1H, H-4′), 3.40 (m, 4H, 2 × CH2,
chloroethyl), 1.63, 1.40 (2 × s, 2 × 3H, 2 × CH3, isopropy-
lidene); 13C NMR δ 43.1, 40.7 (2 × CH2, chloroethyl).
5′-N-(2,2,2-Tr iflu or oet h yl)-2′,3′-O-isop r op ylid en eca r -
boxa m id oa d en osin e (6j). Meth od B: yield 324 mg (81%);
Rf 0.45; 1H NMR data (CDCl3) δ 8.30 (s, 1H, H-8), 7.86 (s, 1H,
H-2), 6.37 (s(b), 2H, NH2), 6.04 (s, 1H, H-1′), 5.25 (m, 2H, H-2′,
H-3′), 4.80 (s, 1H, H-4′), 3.80 (m, 1H, CH2, trifluoroethyl), 1.64,
1.38 (2 × s, 2 × 3H, 2 × CH3, isopropylidene); 13C NMR δ (q,
J C,F ) 280 Hz, CH2CF3), 40.2 (q, J C,F ) 35 Hz, CH2CF3).
5′-N -Me t h oxyl-2′,3′-O-isop r op ylid e n e ca r b oxa m id o-
1
a d en osin e (6k ). Meth od B: yield 170 mg (49%); Rf 0.25; H
NMR (CD3OD) δ 8.20 (s, 1H, H-8), 8.15 (s, 1H, H-2), 6.34 (s,
1H, H-1′), 5.64, 5.45 (2 × m, 2H, H-2′, H-3′), 4.70 (d, 1H, J )
1.7 Hz, H-4′), 3.21 (s, 3H, OCH3), 1.56, 1.39 (2 × s, 2 × 3H, 2
× CH3, isopropylidene).
5′-N-Cyclop r op yl-2′,3′-O-isop r op ylid en eca r boxa m id o-
a d en osin e (6c). Compound 6c was synthesized according to
method A. Five equivalents of cyclopropylamine was added:
5′-N-Meth ylca r boxa m id oa d en osin e (1a ): Yield 60 mg
(43%); Rf 0.06; 1H NMR data (DMSO-d6): δ 8.95 (s, 1H, NH),
8.39 (s, 1H, H-8), 8.23 (s, 1H, H-2), 7.45 (s, 2H, NH2), 5.95 (d,
1H, J 1′,2′ ) 7.7 Hz, H-1′), 4.60 (m, 1H, H-2′), 4.30 (d, 1H, J 4′,3′
) 0.7 Hz, H-4′), 4.15 (m, 1H, H-3′), 2.71 (d, 3H, CH3); mp 241
°C (lit. 240-241 °C20); MS m/z 295 [MH]+.
1
yield 200 mg (58%); Rf 0.55; H NMR (CDCl3) δ 8.32 (s, 1H,
H-8), 7.89 (s, 1H, H-2), 7.25 (s, 1H, NH), 6.40 (s(b), 2H, NH2),
6.08 (d, 1H, J ) 1.7 Hz, H-1′), 5.38 (m, 2H, H-2′, H-3′), 4.71 (s,
1H, H-4′), 2.56 (m, 1H, CH, cyclopropyl), 1.63, 1.39 (2 × s, 2 ×
3H, 2 × CH3, isopropylidene), 0.64, 0.20 (2 × m, 2 × 2H, 2 ×
CH2 cyclopropyl); 13C NMR (CDCl3) δ 21.95 (CH, cyclopropyl),
5.98, 5.75 (2 × CH2, cyclopropyl).
5′-N-(n -P r op yl)ca r boxa m id oa d en osin e (1d ): yield 50
1
mg (85%); Rf 0.15; H NMR data (DMSO-d6): δ 8.99 (t, 1H,
5′-N-(n -P r op yl)-2′,3′-O-isop r op ylid en eca r b oxa m id o-
NH), 8.37 (s, 1H, H-8), 8.19 (s, 1H, H-2), 7.44 (s, 2H, NH2),
5.95 (d, 1H, J 1′,2′ ) 7.7 Hz, H-1′), 5.75, 5.55 (2 × d, 2 × 1H,
2′-OH, 3′-OH), 4.60 (m, 1H, H-2′), 4.31 (d, 1H, J 4′,3′ ) 0.9 Hz,
H-4′), 4.12 (t, 1H, H-3′), 3.43 (m, 2H, CH2CH2CH3), 1.47 (m,
2H, CH2CH2CH3), 1.05 (t, 3H, CH2CH2CH3); 13C NMR (DMSO-
d6) δ 172.7 (C-5′), 157.7 (C-6), 153.5 (C-2), 150.6 (C-4), 143.3
(C-8), 121.7 (C-5), 91.2 (C-1′), 86.8 (C-4′), 75.6, 74.2 (C-3′, C-2′),
42.5 (CH2CH2CH3), 24.3 (CH2CH2CH3), 12.4 (CH2CH2CH3); mp
218 °C (lit.: 220-222 °C20); MS m/z 323 [MH]+.
1
a d en osin e (6d ). Meth od B: yield 66 mg (17%); Rf 0.60; H
NMR (CDCl3) δ 8.30 (s, 1H, H-8), 7.92 (s, 1H, H-2), 7.03 (t,
1H, NH), 6.50 (s(b), 2H, NH2), 6.11 (s, 1H, H-1′), 5.40 (m, 2H,
H-2′, H-3′), 4.73 (s, 1H, H-4′), 3.03 (m, 2H, CH2CH2CH3), 1.63,
1.39 (2 × s, 2 × 3H, 2 × CH3, isopropylidene), 1.25 (m, 2H,
CH2CH2CH3), 0.75 (t, 3H, CH2CH2CH3).
5′-N -Isop r op yl-2′,3′-O-isop r op ylid e n e ca r b oxa m id o-
1
a d en osin e (6e). Meth od B: yield 311 mg (85%); Rf 0.46; H
NMR (CDCl3) δ 8.32 (s, 1H, H-8), 8.01 (s, 1H, H-2), 7.63 (s(b),
2H, NH2), 6.71 (d, 1H, J NH,CH ) 7.0 Hz, NH), 6.20 (d, 1H, J )
1.4 Hz, H-1′), 5.47 (m, 2H, H-2′, H-3′), 4.72 (s, 1H, H-4′), 3.89
(m, 1H, J CH,CH3 ) J CH,NH ) 7.0 Hz, CH(CH3)2), 1.63, 1.40 (2 ×
s, 2 × 3H, 2 × CH3, isopropylidene), 1.00, 0.79 (2 × d, 2 × 3H,
CH(CH3)2); 13C NMR (CDCl3) δ 38.97 (CH(CH3)2), 20.46, 19.88
(2 × CH(CH3)2).
5′-N-Isop r op ylca r b oxa m id oa d en osin e (1e): yield 241
mg (87%); Rf 0.12; 1H NMR (DMSO-d6) δ 8.48 (d, 1H, NH),
8.40 (s, 1H, H-8), 8.19 (s, 1H, H-2), 7.50 (s, 2H, NH2), 5.94 (d,
1H, J 1′,2′ ) 7.5 Hz, H-1′), 5.69, 5.58 (2 × s(b), 2 × 1H, 2′-OH,
3′-OH), 4.60 (dd, 1H, J 2′,1′ ) 7.5 Hz, J 2′,3′ ) 5.0 Hz, H-2′), 4.29
(d, 1H, J 4′,3′ ) 1.5 Hz, H-4′), 4.11 (dd, 1H, J 3′,2′ ) 4.5 Hz, J 3′,4′
) 1.5 Hz, H-3′), 3.98 (m, 1H, CH(CH3)2, 1.12, 1.07 (2 × d, 2 ×
3H, CH(CH3)2; 13C NMR (DMSO-d6) δ 168.8 (C-5′), 155.8 (C-
6), 152.1 (C-2), 149.1 (C-4), 88.1 (C-1′), 84.7 (C-4′), 73.3, 72.3
(C-3′, C-2′), 40.8 (CH(CH3)2), 22.5 (2 × CH(CH3)2); mp 145 °C
(lit.: 137-141 °C20); MS m/z 323 [MH]+.
5′-N-Cyclobu tylca r boxa m id oa d en osin e (1f): yield 173
mg (75%); Rf 0.15; 1H NMR (DMSO-d6) δ 8.86 (d, 1H, NH),
8.43 (s, 1H, H-8), 8.22 (s, 1H, H-2), 7.59 (s, 2H, NH2), 5.95 (d,
1H, J 1′,2′ ) 7.4 Hz, H-1′), 5.60, (s(b), 2H, 2′-OH, 3′-OH), 4.60
(dd, 1H, J 2′,1′ ) 7.4 Hz, J 2′,3′ ) 4.6 Hz, H-2′), 4.28 (d, 1H, J 4′,3′
) 1.5 Hz, H-4′), 4.28 (m, 1H, CH, cyclobutyl), 4.11 (dd, 1H,
J 3′,2′ ) 4.6 Hz, J 3′,4′ ) 1.6 Hz, H-3′), 2.21, 1.95, 1.66 (3 × m, 3
× 2H, 3 × CH2, cyclobutyl); 13C NMR (DMSO-d6) δ 168.8 (C-
5′), 155.7 (C-6), 151.8 (C-2), 149.2 (C-4), 141.5 (C-8), 119.7 (C-
5), 88.3 (C-1′), 84.7 (C-4′), 73.3, 72.5 (C-3′, C-2′), 44.1 (CH2,
cyclobutyl), 30.5 (2 × CH2, cyclobutyl), 15.2 (CH2, cyclobutyl);
mp 235 °C (lit.: 234-235 °C20); MS m/z 335 [MH]+.
5′-N-Cyclob u t yl-2′,3′-O-isop r op ylid en eca r b oxa m id o-
1
a d en osin e (6f). Meth od B: yield 243 mg (64%); Rf 0.50; H
NMR (CDCl3) δ 8.29 (s, 1H, H-8), 8.13 (s, 1H, H-2), 7.21 (d,
1H, J NH,CH ) 8.0 Hz, NH), 6.25 (s, 1H, H-1′), 5.48 (m, 2H, H-2′,
H-3′), 4.70 (s, 1H, H-4′), 4.17 (m, 1H, CH, cyclobutyl), 2.17,
1.95, 1.70, 1.36 (4 × m, 6H, 3 × CH2, cyclobutyl), 1.63, 1.41 (2
× s, 2 × 3H, 2 × CH3, isopropylidene); 13C NMR (CDCl3) δ
43.5 (CH, cyclobutyl), 30.0, 29.6, 14.4 (3 × CH2, cyclobutyl).
5′-N-(ter t-Bu t yl)-2′,3′-O-isop r op ylid en eca r b oxa m id o-
a d en osin e (6g). Meth od B: yield 341 mg (95%); Rf 0.50; 1H
NMR (CDCl3) δ 8.32 (s, 1H, H-8), 8.04 (s, 1H, H-2), 7.63 (s(b),
2H, NH2), 7.11 (s, 1H, NH), 6.25 (d, 1H, J ) 3 Hz, H-1′), 5.55
(m, 2H, H-2′, H-3′), 4.62 (s, 1H, H-4′), 1.61, 1.41 (2 × s, 2 ×
3H, 2 × CH3, isopropylidene), 1.06 (s, 9H, 3 × CH3, tert-butyl);
13C NMR (CDCl3) δ 50.4 (Cq, tert-butyl), 27.9 (s, 3 × CH3, tert-
butyl).
5′-N-Cyclop en tyl-2′,3′-O-isop r op ylid en eca r boxa m id o-
a d en osin e (6h ). Meth od B: yield 383 mg (97%); Rf 0.45; 1H
NMR (CDCl3) δ 8.31 (s, 1H, H-8), 8.13 (s, 1H, H-2), 7.30 (s(b),
2H, NH2), 6.28 (d, 1H, J ) 1.7 Hz, H-1′), 5.51 (m, 2H, H-2′,
H-3′), 4.73 (s, 1H, H-4′), 4.00 (m, 1H, CH, cyclopentyl), 1.62,
1.41 (2 × s, 2 × 3H, 2 × CH3, isopropylidene), 1.81, 1.65, 1.22,
0.85 (4 × m, 8H, 4 × CH2, cyclopentyl); 13C NMR (CDCl3) δ
50.0 (CH, cyclopentyl), 32.3, 31.6 (2 × CH2, cyclopentyl).
5′-N-(2-Ch lor oeth yl)-2′,3′-O-isop r op ylid en eca r boxa m i-
d oa d en osin e (6i). Met h od A. A solution of 2-chloroethyl-
amine hydrochloride (500 mg, 4.3 mmol) in water (200 µL) was
added together with an additional amount of triethylamine
(4.3 mmol, 600 µL). TLC analysis indicated the formation of
one main product, among four other products with Rf values
near that of the main product. This was not a problem for
5′-N-(ter t-Bu tyl)ca r boxa m id oa d en osin e (1g): yield 146
mg (96%); Rf 0.10; 1H NMR (DMSO-d6) δ 8.44 (s, 1H, H-8),
8.16 (s, 1H, H-2), 7.72 (s, 1H, NH), 7.56 (s, 2H, NH2), 5.91 (d,
1H, J 1′,2′ ) 7.0 Hz, H-1′), 4.64 (dd, 1H, J 2′,1′ ) 7.0 Hz, J 2′,3′
)
4.5 Hz, H-2′), 4.23 (d, 1H, J 4′,3′ ) 1.5 Hz, H-4′), 4.09 (dd, 1H,
J 3′,4′ ) 1.5 Hz, J 3′,2′ ) 4.5 Hz, H-3′), 1.27 (s, 9H, 3 × CH3, tert-
butyl); 13C NMR (DMSO-d6) δ 52.4 (Cq, tert-butyl), 28.9 (s, 3
× CH3, tert-butyl); mp 140 °C; MS m/z 337 [MH]+, 359 [MNa]+.
Anal. (C14H20N6O4‚1.1H2O) C, H, N.
5′-N-Cyclop en tylca r boxa m id oa d en osin e (1h ): yield 140
mg (41%); Rf 0.15; 1H NMR (DMSO-d6) δ 8.43 (s, 1H, H-8),
8.14 (s, 1H, H-2), 7.50 (s, 2H, NH2), 5.94 (d, 1H, J 1′,2′ ) 7.0 Hz,
H-1′), 5.65, 5.52 (2 × s(b), 2 × 1H, 2′-OH, 3′-OH), 4.60 (dd,
1H, J 2′,1′ ) 7.0 Hz, J 2′,3′ ) 5.0 Hz, H-2′), 4.30 (d, 1H, J 4′,3′ ) 1.5
Hz, H-4′), 4.13 (dd, 1H, J 3′,4′ ) 1.5 Hz, J 3′,2′ ) 5.0 Hz, H-3′),
4.10 (m, 1H, CH, cyclopentyl), 1.85, 1.64, 1.51, 1.40 (4 × m,
8H, 4 × CH2 cyclopentyl); 13C NMR (DMSO-d6) δ 50.3 (CH,
1
separation: yield 140 mg (38%) as a white powder; H NMR
(CDCl3) δ 8.30 (s, 1H, H-8), 7.91 (s, 1H, H-2), 7.32 (m, 1H,

 De Zwart, Maarten
De Zwart, Maarten
 Kourounakis, Angeliki
Kourounakis, Angeliki
 Kooijman, Huub
Kooijman, Huub
 Spek, Anthony L.
Spek, Anthony L.
 Link, Regina
Link, Regina
 Von Frijtag Drabbe Künzel, Jacobien K.
Von Frijtag Drabbe Künzel, Jacobien K.
 IJzerman, Ad P.
IJzerman, Ad P.