S.-Z. Zheng et al. / Journal of Fluorine Chemistry 145 (2013) 132–135
135
particles on the support is necessary in order to establish a
4.3. Catalyst characterizations
correlation between the morphologies of the Ru particles and
their catalytic behaviors. Concerning the active sites/phase, the
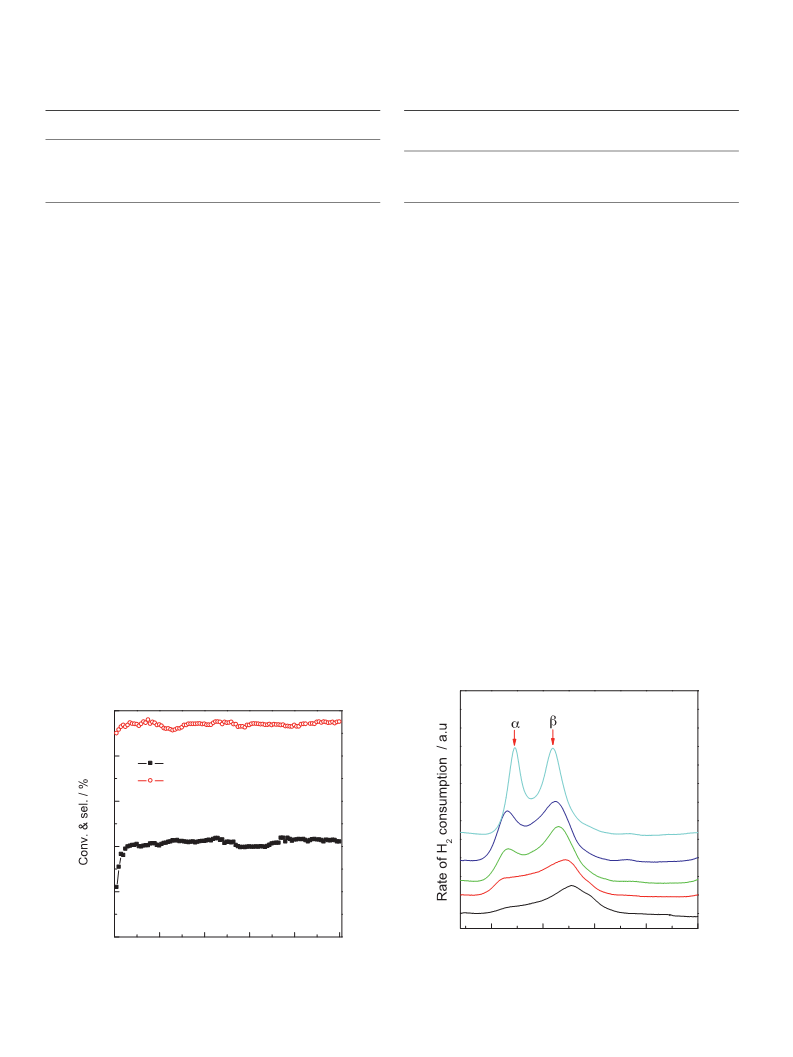
results shown in Table 1 indicate that the TOF value decreases on
the Ru/C catalysts at high Ru content. Note that the ratio of Ru
species interacting with support/total exposed Ru atoms
decreases with increasing Ru loading in the catalyst (71.2% for
1Ru/C, 73.1% for 2Ru/C, 74.4% for 3Ru/C, 66.6% for 4Ru/C, 62.6% for
5Ru/C, based on the TPR profiles in Fig. 5), which is consistent with
the changes in TOFs. Therefore, it seems that the Ru species that
strongly interact with the support might be the catalytically active
components for the reaction.
X-ray diffraction (XRD) patterns were recorded using
a
PANalytic X’Pert PW3040 diffractiometer with Cu K
operating at 40 kV and 40 mA. The patterns were collected in a 2
range from 10 to 908, with a scanning step of 0.158 sÀ1
a radiation
u
.
Transmission electron microscopy (TEM) analysis was per-
formed on a JEM-2100F electron microscopy with a field emissive
gun, operated at 200 kV and with a point resolution of 0.24 nm.
The samples were prepared by dispersing a few milligrams of
powder in ethanol. The dispersion was then immersed for 10 min
in an ultrasonic bath in order to disagglomerate the powder
particles. Finally, one drop was deposited on a Formvar/carbon
copper grid. Various regions of the grid were observed and the
particle sizes were measured from the observation of 150–250
particles.
3. Conclusion
For the Ru/C catalyst, with increasing Ru content, Ru
dispersion increased and the methyl difluoroacetate conversion,
selectivity to difluoroethanol and TOF first increased and then
decreased. Both the highest conversion (39.5%) and the highest
selectivity to difluoroethanol (93.5%) were obtained on 3Ru/C
catalyst. It was also found that the reduction temperature had
great effect on catalytic activity, and the catalyst reduced at
400 8C exhibited the best catalytic performance. Furthermore,
the 3Ru/C catalyst was quite stable during the reaction period
within 100 h.
CO chemisorption experiments were carried out on a Quanta-
chrome CHEMBET-3000 instrument in order to determine the
dispersion of Ru. The sample was placed in a U-shaped quartz
reactor and a high-purity He (99.999%) gas flow of 70 ml minÀ1
was used as the carrier gas. Before CO chemisorption, the samples
were reduced in a H2–N2 mixture (5 vol% H2) stream for 2 h, cooled
down to 30 8C, and then purged with a pure He gas flow for 1 h at
the same temperature. Then pulses of CO were fed into the stream
of carrier gas with a precision analytical syringe.
The reducibility of the catalysts was measured by hydrogen
temperature-programmed reduction (H2-TPR) experiments.
4. Experiment
100 mg of sample was placed in
a quartz tubular reactor
(i.d. = 6 mm). After that the sample was heated from 40 to
600 8C at a rate of 10 8C minÀ1 in a H2–N2 gas mixture (5 vol% H2,
30 ml minÀ1). The hydrogen consumption during the reduction
4.1. Catalyst preparation
Supported Ru/C catalysts were prepared by impregnating the
active carbon support (XC-72, SBET = 223 m2 gÀ1) with an aqueous
solution of RuCl3 for 8 h. Excess solution was removed by mild
evaporation, and then the samples were dried at 110 8C for 12 h.
Before the catalytic test, the catalysts were pre-reduced in ultra-
pure H2 flow (99.999%, 30 ml minÀ1) at different temperatures for
2 h. The catalysts were denoted as xRu/C, where x refers to the
weight percent of Ru (wt.%) in the catalyst.
was determined by
conductivity detector.
a gas chromatograph with a thermal
Acknowledgements
This research is financially supported by National Natural
Science Foundation of China (21173194) and Zhejiang Provincial
Natural Science Foundation of China (Y4100300).
4.2. Catalytic performance test
References
Gas phase hydrogenolysis of methyl difluoroacetate was
performed in a stainless steel tubular reactor (i.d. = 10 mm,
length = 300 mm) at atmospheric pressure. About 500 mg of the
catalyst was loaded in the reactor with a thermal couple placed in
the middle of the catalyst bed to monitor the reaction temperature.
Methyl difluoroacetate was introduced in a trap set maintained at
0 8C before the reactor to achieve a constant methyl difluoroacetate
partial pressure, then methyl difluoroacetate at constant partial
pressure was carried over the catalyst by hydrogen flow
(20 ml minÀ1). The gas line was kept at about 110 8C to avoid
any condensation. The reaction products and reactants were
analyzed on line using a gas chromatography (Shimadzu GC-2014)
equipped with a flame ionization detector (FID) and a DB-Wax
[1] B. Liu, L. Dai, X.L. Yang, et al. Synthetic Communications 2 (2001) 1875–1877.
[2] H. Willenberg, W. Pohlmeyer, W. Rudolph, US Patent 4,694,112[P] (1987).
[3] D.W. Wujciak, R.H. Wade, US Patent 2,824,897[P] (1955).
[4] A. Lecloux, F. Legrand, US Patent 4,542,245 (1985).
[5] P.W. Townsend, C. Huang, G.G. Vernice, US Patent 4,590,310 (1986).
[6] C. Doussain, M. Gubelmann, et al., US Patent 4,950,811 (1990).
[7] G.W. Astrologes, US Patent 4,434,297 (1984).
[8] D.R. Husted, A.H. Albrecht, Journal of the American Chemical Society 21 (1952)
5422–5426.
[9] B.M. Regan, et al., US Patent 3,702,872 (1972).
[10] A. Yamamoto, T. Shibanuma, WO Patent 0,162,694 (2001).
[11] R.N. Haszeldine, R. Rowland, R.P. Sheppard, et al. Journal of Fluorine Chemistry 3
(1985) 291–302.
[12] T. Satokawa, T. Fujii, A. Ohmori, Y. Fujita, US Patent 4,346,250 (1982).
[13] H. Willenberg, W. Pohlmeyer, W. Rudolph, US Patent 4,649,112 (1987).
[14] D.W. Wujciak, US Patent 2,824,897 (1956).
[15] B. Olivier, L. Karol, WO Patent 2,007,093,567.
column (30 m  0.25 mm  0.25
m
m) capillary column.
[16] I. Rossetti, N. Pernicone, L. Forni, Applied Catalysis A 248 (2003) 97–103.

 Zheng, Su-Zhen
Zheng, Su-Zhen
 Cao, Xiao-Yan
Cao, Xiao-Yan
 Zhou, Qiang
Zhou, Qiang
 Wang, Shu-Hua
Wang, Shu-Hua
 Hu, Geng-Shen
Hu, Geng-Shen
 Lu, Ji-Qing
Lu, Ji-Qing
 Luo, Meng-Fei
Luo, Meng-Fei
 Wang, Yue-Juan
Wang, Yue-Juan