Q. Liu, D.J. Burton / Journal of Fluorine Chemistry 130 (2009) 922–925
925
Et2O The mixture was cooled to À100 to À110 8C with liquid
nitrogen/pentane slush bath. Then, 145 ml (365 mmol) of a 2.5 M n-
BuLi solution was gradually added to the reaction mixture,
maintaining the internal temperature at <À100 8C. After complete
addition of the n-BuLi solution, the reaction mixture was stirred at
À90 to À100 8C for 1 h. Then, a solution of I2 [110 g I2 (443 mmol) in
200 ml THF] was gradually added to the reaction mixture via a
syringe, maintaining the temperature at À95 to À100 8C. After
complete addition of the I2 solution, the reaction mixturewas stirred
at À95 to À100 8C for 1 h, then slowly warmed to À30 8C over 3 h.
Then, the reaction mixture was quenched with dilute HCl at À30 8C
until pH <6. The solution was washed with saturated aqueous
Na2S2O3 until the color of the solution changed from dark brown to
light yellow. The organic layer was extracted with Et2O (3Â 100 ml),
washed with NaHCO3 solution (5Â 50 ml), water (5Â 100 ml), then
dried over MgSO4. After removal of the MgSO4 by gravity filtration,
most of the solvent was removed by rotary evaporation. Removal of
the remaining solvent by flash distillation at ꢀ20 8C/1 mm Hg
yielded 94.2 g (89%) of a colorless liquid, 6, GLPC = 95%. The 19F and
1H NMR were identical to previously prepared material [8].
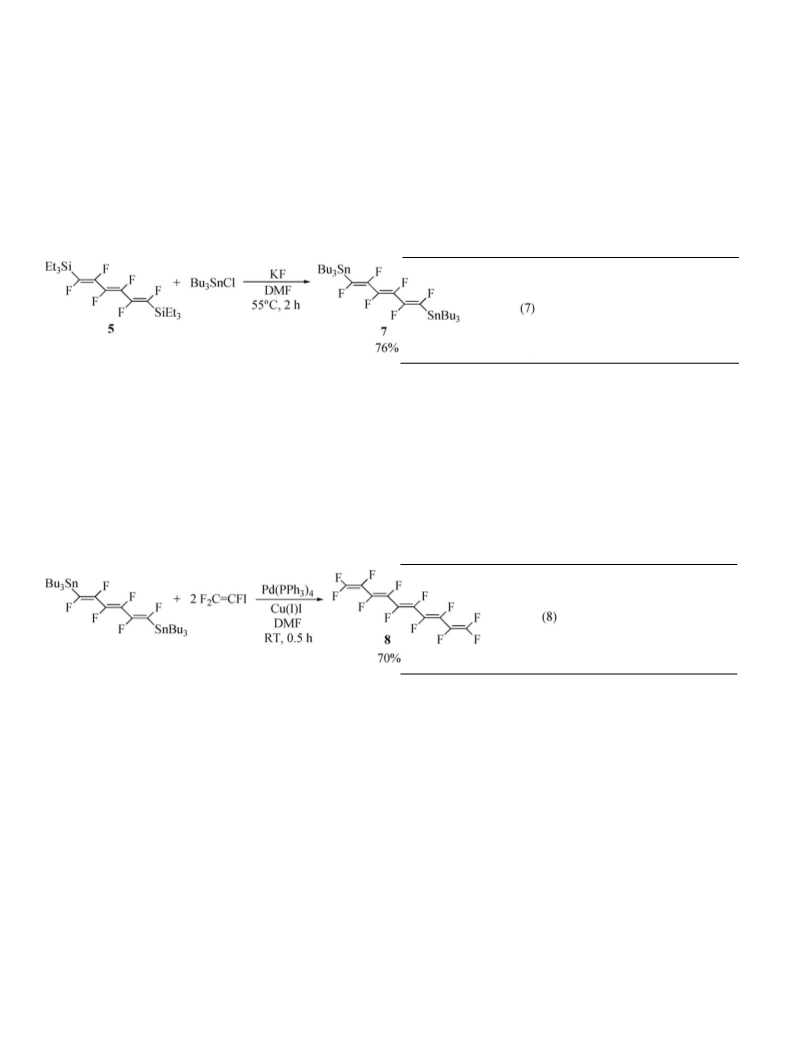
(1.08 mmol) of 5, 0.74 g (2.2 mmol) of tributyltin chloride,
0.16 g (2.6 mmol) KF and 2 ml DMF. The reaction mixture was
stirred at 55 8C for 2 h. The dark reaction mixture was poured onto
a silica gel column and eluted with pentane. Removal of solvent at
RT/1 mm Hg gave 0.63 g (76%) of a colorless liquid, 7. 19F NMR:
d
3
3
À139.8 (dt, JFF = 124 Hz, JFF = 22 Hz, 2F), À151.7 (m, 2F), À163.4
(dm, JFF = 124 Hz, 2F). 1H NMR:
d 1.6 (m), 1.34 (m), 1.2 (tm,
3
3JHH = 8 Hz), 0.9 (t, 3JHH = 7 Hz). 13C NMR:
d 28.7 (m), 27.1 (m), 13.6
(m), 10.3 (n)-butyl group.
3.7. Preparation of (3E,5E,7E)-dodecafluoro-1,3,5,7,9-
decapentaene, 8
A one-neck 50 ml flask, equipped with a stir bar and attached
to a nitrogen inlet tee, was charged with 0.44 g (0.57 mmol) of 7,
0.17 g (0.9 mmol) iodotrifluoroethene, 2 ml DMF, 0.03 g (3 mol%)
Pd(PPh3)4 and 0.05 g (0.3 mmol) Cu(I)I. The reaction mixture was
stirred at RT for 0.5 h. Flash distillation of the reaction mixture at
RT/0.1 mm Hg gave a liquid, which on simple distillation gave
0.20 g (70%) of a colorless liquid 8, GLPC = 96%. 19F NMR:
(m), À103.5 (m), À148.4 (m), À151.8 (m), À155.2 (m), À186.3
(m). On storage in the freezer, this product underwent decom-
position.
d
À90.7
3.4. Preparation of (E)-octafluoro-1,3,5-hexatriene, 4
A one-neck 100 ml round bottom flask equipped with a Teflon-
coated stir bar and attached to a nitrogen inlet tee was charged
with 12.02 g (19 mmol) of 3, 6.86 g (32.7 mmol, GLPC purity = 99%)
iodotrifluoroethene, 30 ml DMF, 0.83 g (4.3 mmol), Cu(I)I and 1.1 g
(3 mol%) Pd(PPh3)4. The reaction mixture was stirred at RT for 6 h;
followed by flash distillation to give a crude liquid product.
Fractional distillation of the crude product via a 8 cm Vigreux
column yielded 2.32 g (61%) of a colorless liquid 4, bp 67–68 8C/
4. Conclusion
(E)-(1,2-Difluoro-1,2-ethenediyl)bis[tributylstannane] 3, read-
ily undergoes a Pd(PPh3)4/CuI-catalyzed coupling reaction with
iodotrifluoroethene to give (E)-octafluoro-1,3,5-hexatriene in
good isolated yield and high isomeric purity, (1Z,3E,5Z)-
(1,2,3,4,5,6-hexafluoro-1,3,5-hexenetriyl)bis[tributylstannane], 7,
was prepared from (1Z,3E,5Z)-(1,2,3,4,5,6-hexafluoro-1,3,5-hexe-
netriyl)bis[triethylsilane], which was prepared via a P(PPh3)4/
CuI-catalyzed cross-coupling reaction of 3 with (E)-1,2-difluoro-1-
iodo-2-triethylsilylethene. Pd(PPh3)4/CuI-catalyzed cross-coupling
of 7 with iodotrifluoroethene gave (3E,5E,7E)-dodecafluoro-
1,3,5,7,9-decapentaene, 8.
760 mm Hg, GLPC purity = 96%. 19F NMR:
d
À91.5 (dd, 2JFF = 40 Hz,
3JFF = 32 Hz), À104.6 (ddm, 3JFF = 115 Hz, 2JFF = 40 Hz), À150.9 (m),
3
3
À185.2 (ddm, JFF = 115 Hz, JFF = 32 Hz). GC–MS, m/z (relative
intensity): 224 (M+, 37), 155 (100). HRMS: calculated for C6F8:
223.9872, observed 223.9856 [7].
3.5. Preparation of (1Z,3E,5Z)-(1,2,3,4,5,6-hexafluoro-1,3,5-
hexeneyl)bis(triethylsilane), 5
Acknowledgement
A one-neck 50 ml flask, equipped with a stir bar, and attached toa
nitrogen inlet tee was charged with 4.49 g (7 mmol) of 3, 3.7 g
(12.2 mmol) of 6, 8 ml THF and 8 ml DMF, 0.1 g (0.5 mmol, 4 mol%)
Cu(I)I and 0.25 g (3 mol%) Pd(PPh3)4. The reaction mixture was
stirred at RT for 2 h. The dark reaction mixture was poured onto a
silica gel column and eluted with pentane, Rf = 0.6. Removal of
We thank the National Science Foundation for financial support
of this work.
References
solvent at RT/1 mm Hg gave 2.28 g (90%) of a colorless liquid, 5. 19
F
[1] D.J. Burton, S.W. Hansen, P.A. Morken, K.J. MacNeil, C.R. Davis, L. Xue, J. Fluorine
Chem. 129 (2008), 435–442 and references therein.
[2] A. Karpfen, J. Phys. Chem. A 103 (1999) 2817–2821.
[3] E.M. Torres, T.L. Fonseca, S.C. Esteves, O.A.V. Amaral, M.A. Castro, Chem. Phys. Lett.
403 (2005) 268–274.
3
3
NMR:
d
À148.9 (dt, JFF = 133 Hz, JFF = 23 Hz), À150.8 (m, 2F),
À161.8 (ddd, 3JFF = 133 Hz, 4JFF = 14 Hz, 5JFF = 6 Hz); 1H NMR:
d 1.02
(t, JHH = 7.8 Hz), 0.80 (q, JHH = 7.8 Hz); 13C NMR:
d 162.5 (ddd),
3
3
1JCF = 286 Hz, 2JCF = 72 Hz, 3JCF = 2 Hz), 150.5 (m), 104.4 (m), 6.98 (s),
2.21(s). FTIR: 2959 (vs), 2939(m), 2914(s), 1458(w), 1285 (m), 1181
(vs), 1006 (s). GC–MS, m/z (relative intensity) 416 (M+, 2.7), 105
(40.6), 77 (100). HRMS: calculated for C18H30Si2F6: 416.1790,
observed 416.1806.
[4] V. Dedek, Z. Chvatal, J. Fluorine Chem. 31 (1986) 363–379.
[5] N. Jing, D.M. Lemal, J. Org. Chem. 59 (1994) 1844–1848.
[6] Q. Liu, D.J. Burton, Org. Lett. 4 (2002) 1483–1485.
[7] Literature data [5] reported for (E)-1,3,5-hexatriene: d À91.2 (m), À104.4 (dm,
J = 115 Hz), À150.8 (m), À185.0 (dm, J = 115 Hz).
[8] S.A. Fontana, C.R. Davis, Y.-B. He, D.J. Burton, Tetrahedron 52 (1996) 37–
44.
[9] P. Martinet, R. Sauvetre, J.-F. Normant, J. Organomet. Chem. 367 (1989) 1–
10.
3.6. Preparation of (1Z,3E,5Z)-(1,2,3,4,5,6-hexafluoro-1,3,5-
[10] L. Xue, L. Lu, S. Pedersen, Q. Liu, R. Narske, D.J. Burton, J. Org. Chem. 62 (1997)
1064–1071.
hexenyltriyl)bis[tributylstannane], 7
[11] D.R. Coulson, Inorg. Synth. 13 (1972) 121–124.
[12] C. Lim, D.J. Burton, C.A. Wesolowski, J. Fluorine Chem. 119 (2003) 21–26.
[13] D.J. Burton, V. Jairaj, J. Fluorine Chem. 125 (2004) 673–680.
A one-neck 50 ml flask, equipped with a stir bar, condenser, and
attached to
a nitrogen inlet tee, was charged with 0.45 g

 Liu, Qibo
Liu, Qibo
 Burton, Donald J.
Burton, Donald J.