6+257ꢀ3$3(5
Non-acid Synthesis of Nitro Musks
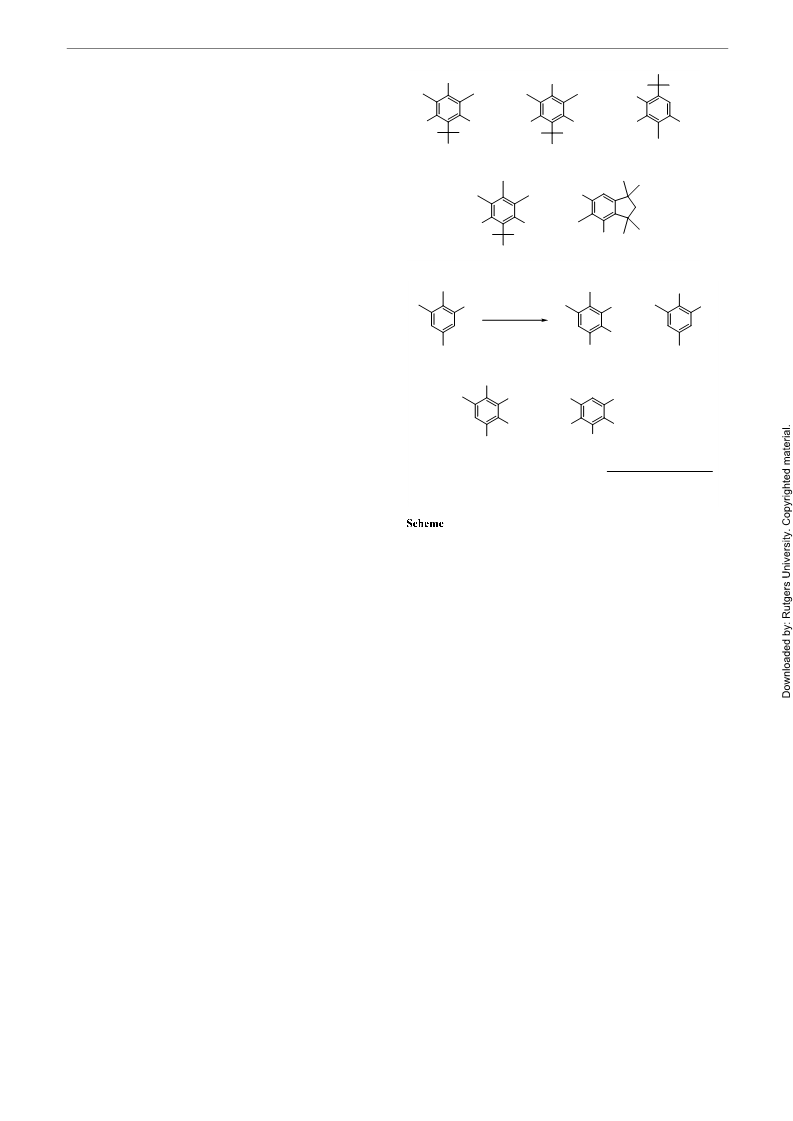
ꢁꢂꢃꢆ
pure musk ketone ꢂꢀas white crystals, 0.59 g (40%).ꢀMp 133–135 °C
3
a
[
lit. 135.5–136 °C].
1
H NMR(CDCl , 200 MHz): d = 2.51 (s, 3H), 2.11 (s, 6H), 1.44 (s,
3
9
H).
+
GC-MS (CI): P/] (%) = 295 (M +1, 100), 279 (9), 266 (25), 250 (2).
–
1
FT-IR (KBr): n = 1713, 1539, 1474, 1372, 1350, 1242, 1125 cm .
0
XVNꢀ$PEUHWWHꢀꢏꢆꢐ
This compound was obtained similarly according to the above pro-
cedure. White solid (46%); mp 81–83 °C [lit. 85 °C].
3
b
1
H NMR(CDCl , 200 MHz): d = 8.06 (s, 1H), 3.92 (s, 3H), 2.40 (s,
3
3
H), 1.43 (s, 9H).
+
GC-MS (CI): P/] (%) = 269 (M +1, 91), 253 (7), 239 (4), 213 (100),
07 (4).
2
FT-IR (KBr): n = 1617, 1541, 1534, 1350, 1271, 1246, 1062, 1052
–
1
cm .
$FNQRZOHGJHPHQW
This work was supported by the Grant-in-Aid for Scientific Rese-
arch of the Ministry of Education, Culture, Science and Sports, Ja-
pan (No. 08101003). The authors would like to thank Mr. Haruyasu
Shiota of Shiono Perfumery Co. Ltd., Osaka, for valuable informa-
tion on commercial nitro musks. N.N. also thanks the Japan Society
for the Promotion of Science (JSPS) for the fellowship (No. 9191).
5HIHUHQFHV
(
1) (a) Bauer, A. %HUꢇꢀ'WVFKꢇꢀ&KHPꢇꢀ*HVꢇ ꢁꢍꢃꢁ, ꢁꢈ, 2832.
(
(
b) Bauer, A. %HUꢇꢀ'WVFKꢇꢀ&KHPꢇꢀ*HVꢇ ꢁꢍꢃꢇ, ꢁꢉ, 1614.
c) Bauer, A. %HUꢇꢀ'WVFKꢇꢀ&KHPꢇꢀ*HVꢇ ꢁꢍꢃꢍ, ꢆꢃ, 1344.
(
2) (a) Bauer, K.; Garbe, D.; Surburg, H. in 8OOPDQQ¶Vꢀ
QF\FORSHGLDꢀRIꢀ,QGXVWULDOꢀ&KHPLVWU\, 5th ed.; VCH:
Weinheim, 1988, Vol. A11, pp.141–225.
b) Ohloff, G. )UDJUDQFHꢀ&KHPLVWU\ꢇꢀ7KHꢀ6FLHQFHꢀRIꢀWKHꢀ6HQVHꢀ
RIꢀ6PHOO; Theimer, E. T., Ed.; Academic: New York, 1982.
c) Wood, T. F. &KHPLVWU\ꢀRIꢀ$URPDWLFꢀ0XVNV; Givaudan
Corp. Publisher: Geneva, 1969.
d) For a recent review on fragrance chemistry, see Fráter, G.;
(
(
separate as a white solid. After 3 h, unchanged nitrogen dioxide was
(
expelled by anhyd N and recovered in a cold trap for reuse. The sol-
2
id product was collected by filtration. The filtrate was washed with
brine, dried (Na SO ), and evaporated under reduced pressure to ob-
tain the second crop. The combined crude musk xylol was recrystal-
(
2
4
Bajgrowicz, J. A.; Kraft, P. 7HWUDKHGURQ ꢁꢃꢃꢍ, ꢂꢈ, 7633.
3) (a) Fuson, R. C.; Mills, J.; Klose, T. G.; Carpenter, M. S. -ꢇꢀ
(
lized from EtOH to yield pure ꢁ, 1.22 g (82%). Mp 107–109 °C
2
UJꢇꢀ&KHPꢇ ꢁꢃꢇꢌ, ꢃꢁ, 587.
a
[
Lit. 1 110 °C].
(
b) Carpenter, M. S.; Easter, W. M.; Wood, T. F. -ꢇꢀ2UJꢇꢀ
1
H NMR(CDCl , 200 MHz): d = 2.17 (s, 6H), 1.46 (s, 9H).
&KHP. ꢁꢃꢈꢁ, ꢃꢊꢋ 586.
(c) Carpenter, M. S.; Easter, W. M. -ꢇꢀ2UJꢇꢀ&KHPꢇ ꢁꢃꢈꢁ, ꢃꢊ,
3
+
GC-MS (CI): P/z (%) = 298 (M +1, 100), 282 (20), 278 (43), 253
6
18.
d) Carpenter, M. S.; Easter, W. M. -ꢇꢀ2UJꢇꢀ&KHPꢇ ꢁꢃꢈꢇ, ꢃꢄ,
7.
e) Carpenter, M. S.; Easter, W. M. -ꢇꢀ2UJꢇꢀ&KHPꢇ ꢁꢃꢈꢇ, ꢃꢄ,
87.
(
16).
(
7
(
–
1
FT-IR (KBr): n = 1545, 1474, 1397, 1354, 1206 cm .
.
\RGDLꢄ1LWUDWLRQꢀRIꢀꢇꢄWHUWꢄ%XW\OꢄꢂꢎꢉꢄGLPHWK\ODFHWRSKHQRQHꢀꢏꢉEꢐꢀ
WRꢀ0XVNꢀ.HWRQHꢀꢏꢂꢐꢑꢀ7\SLFDOꢀ3URFHGXUH
-WHUW-Butyl-2,6-dimethylacetophenone (ꢉE) (1.02 g, 5 mmol) was
(4) Suzuki, H.; Murashima, T.; Shimizu, K. and Tsukamoto, K. -ꢇꢀ
&KHPꢇꢀ6RFꢇꢋꢀ&KHPꢇꢀ&RPPXQꢇ ꢁꢃꢃꢁ, 1049. For a survey of the
N\RGDL-nitration, see; Mori, T.; Suzuki, H. 6\QOHWW ꢁꢃꢃꢈ, 383.
Suzuki, T.; Noyori, R. &KHPWUDFWV ꢁꢃꢃꢌ, ꢃꢌ, 813.
Ridd, J. H. $FWDꢀ&KHPꢇꢀ6FDQG. ꢁꢃꢃꢍ, ꢂꢁ, 11.
4
dissolved in freshly distilled hexane (25 mL) and placed in a three-
necked flask fitted with a gas inlet tube and a vent which permitted
waste gas to escape. The mixture was cooled to –10 °C under stir-
ring and liquid nitrogen dioxide (1 mL, 30 mmol) was introduced in
one portion. To the resulting mixture ozonized oxygen was passed
at a low flow rate. After 1 h, an additional amount of nitrogen diox-
ide (1 mL) was added and ozone was continuously fed for further
Nonoyama, N; Mori, T.; Suzuki, H. =Kꢇꢀ2UJꢇꢀ.KLP.ꢀꢁꢃꢃꢍ,
ꢆꢈ,1591.
(5) Ozone generators (Toshiba WHO-A series; Toshiba Co. Ltd.,
Tokyo) with the output of ozone in 1-40 kg h- scale from air
are available on the market.
1
1
.5 h. Under these conditions, the loss of nitrogen dioxide was not
so significant. Then the mixture was diluted with H O and the or-
2
ganic phase was extracted with CH Cl . The extract was washed
2
2
with brine, dried (Na SO ), and evaporated under reduced pressure
Article Identifier:
2
4
to leave a solid residue, which was recrystallized from EtOH to give
1437-210X,E;1999,0,08,1291,1293,ftx,en;F10799SS.pdf
Synthesis 1999, No. 8, 1291–1293 ISSN 0039-7881 © Thieme Stuttgart · New York

 Suzuki, Hitomi
Suzuki, Hitomi
 Hisatome, Kaori
Hisatome, Kaori
 Nonoyama, Nobuaki
Nonoyama, Nobuaki