
International Journal of Chemical Kinetics p. 253 - 260 (1999)
Update date:2022-08-10
Topics:
 Khan, M. Niyaz
Khan, M. Niyaz
 Bakar, Baki B.
Bakar, Baki B.
 Yin, Fenny W. N.
Yin, Fenny W. N.
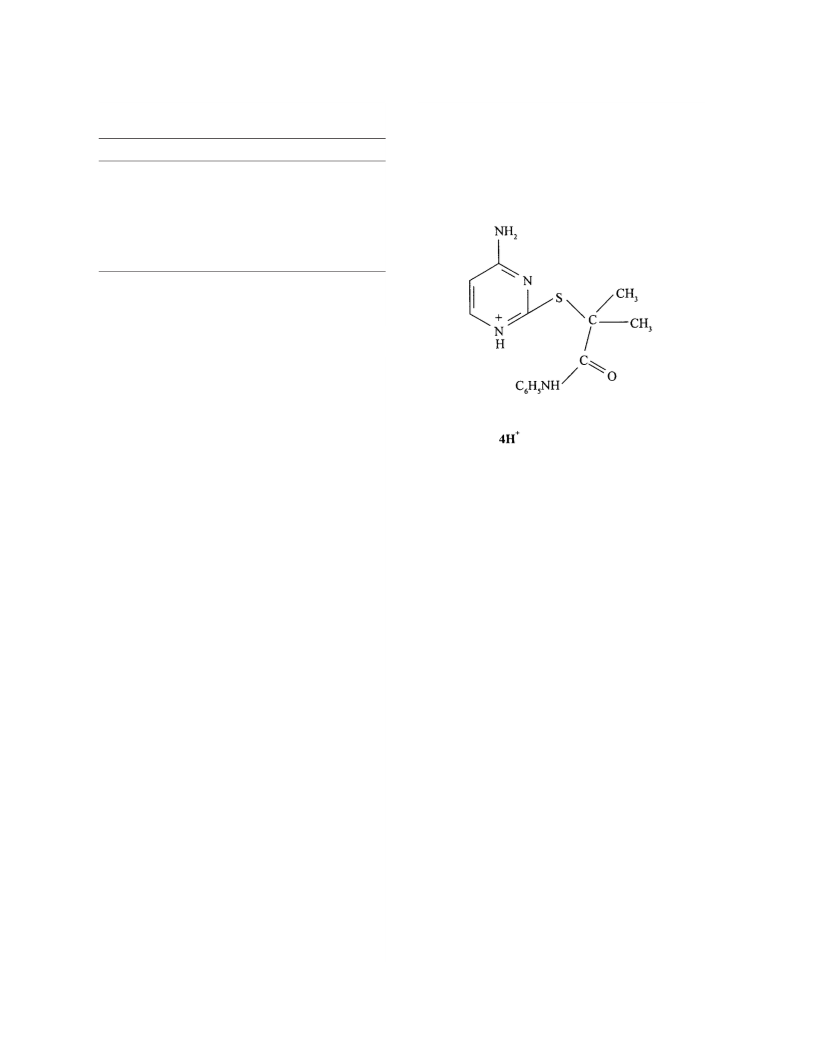
Pseudo-first-order rate constants (kobs) for hydrolysis of a sulfonylurea herbicide, azimsulfuron, AZIM, {N-[[(4,6-dimethoxy-2-pyrimidinyl)amino]carbony]-1-methyl-4- (2-methyl-2H-tetrazol-5-yl)-1H-pyrazole-5-sulfonamide} (AZS) follow an empirical relationship: kobs = α1+α2[-OH]+α3 [-OH]2 within the [NaOH] range of 0.1-2.0 M at different temperatures ranging from 40 to 55 °C. The contribution of α3[-OH]2 term is small compared with α2[-OH] term and this turns out to be zero at 60 °C. Pseudo-first-order rate constants (kobs) for hydrolysis of AZS within the [H+] range from 2.5×10-6 to 1.4 M follow the relationship: kobs = (α1Ka+β1[H*]+β2 [H+]2)/([H+]+Ka) where pKa = 4.37 at 50 °C. The value of β1 is nearly 25 times larger than that of α1. The rate of alkaline hydrolysis of AZIM is weakly sensitive to ionic strength.
View More






ShiJiaZhuang Dowell Chemical Co.,Ltd.
website:http://www.dowechem.com
Contact:+86-13463963265,+86-311-89805679
Address:Xiyangling village, high tech Zone, Shijiazhuang,Hebei, China
Zhangjiagang Jianing Import & Export Co.,Ltd.
Contact:086-512-55379012 13913607595
Address:NO.1 Guotai North Road Zhangjiagang Economic Development Zone,215600,Jiangsu,China
Contact:+33-5-34012600
Address:28 ZA des Pignès
website:http://www.debyesci.com
Contact:+85221376140
Address:Rm. 19C, Lockhart Ctr., 301-307 Lockhart Rd., Wan Chai
Xinjiang Fufeng Biotechnologies Co., Ltd.
Contact:+86-539-7287111
Address:GANQUANPU INDUSTRIAL PARK, ECONOMIC AND TECHNOLOGICAL DEVELOPMENT AREA (TOUTUNHE DISTRICT) OF URUMQI
Doi:10.1039/c5cc02989a
(2015)Doi:10.1016/j.ica.2016.07.001
(2016)Doi:10.1039/c1nj20400a
(2011)Doi:10.1016/S0040-4039(00)86026-8
(1983)Doi:10.1016/0022-328X(89)85030-2
(1989)Doi:10.1016/S0960-894X(02)00332-3
(2002)