Crystal Growth & Design
Article
1
4
H NMR (chloroform-d, 400 MHz): δ 9.20 (d, 2H, J = 5.6 Hz)
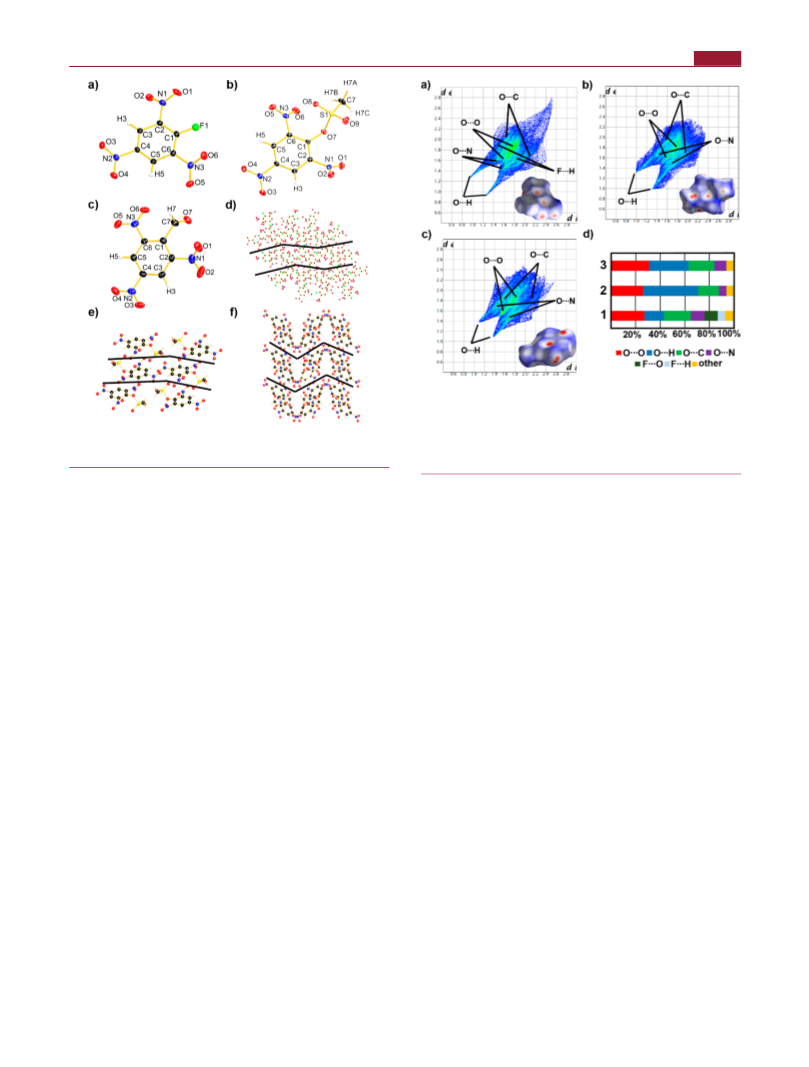
X-ray Measurements. 1-Fluoro-2,4,6-trinitrobenzene and 2,4,6-
trinitrobenzaldehyde were dissolved in ethyl acetate, and single
crystals were obtained after slow solvent evaporation. Single crystals
of 2,4,6-trinitrophenyl methanesulfonate were obtained after slow
solvent evaporation of chloroform. Data collection was performed
with an Oxford Xcalibur3 diffractometer with a CCD area detector,
equipped with a multilayer monochromator, a Photon 2 detector, and
a rotating-anode generator, for data collection using Mo Kα radiation
FH
ppm. 13C{ H} NMR (chloroform-d, 100 MHz): δ 154.2 (d, J
92.0 Hz), 142.4 (s, broad), 139.4 (s, broad), 125.9 (d, J = 0.8 Hz)
1
1
=
FC
3
2
FC
14
19
ppm. N NMR (chloroform-d, 29 MHz): δ −24 (s, NO ) ppm.
F
2
4
NMR (chloroform-d, 377 MHz): δ −113.2 (t, J = 5.6 Hz) ppm. IR
FH
−
1
(
1
(
(
5
°
1
(
2
3
ATR, cm ): ν
541 (vs), 1482 (m), 1421 (m), 1342 (vs), 1319 (s), 1280 (m), 1258
̃
3110 (w), 3089 (w), 3063 (m), 2887 (w), 1620 (s),
m), 1202 (m), 1089 (s), 948 (w), 938 (m), 923 (s), 827 (vw), 776
w), 757 (w), 737 (s), 718 (s), 709 (vs), 672 (m), 649 (s), 551 (m),
19 (m), 481 (w), 462 (w), 406 (w). Raman (1064 nm, 1000 mW, 25
C, cm ): ν
347 (51), 1279 (14), 1187 (4), 1090 (6), 940 (11), 927 (7), 826
21), 817 (12), 741 (3), 522 (2), 377 (5), 354 (8), 333 (20), 313 (7),
05 (26), 149 (8), 105 (48), 90 (40). Anal. Calcd for C H FN O : C,
1.18; H, 0.87; F, 8.22; N, 18.18; O, 41.54. Found: C, 31.42; H, 1.13;
N, 18.23. DTA: 127 °C (mp), 351 °C (dec). IS: 10 J. FS: >360 N.
ESD: 160 mJ.
(
λ= 0.7107 Å). Data collection and reduction were carried out using
34
the CrysAlisPro software. The structures were solved by direct
methods (SIR-2014), refined (SHELXLE) by full-matrix least
squares on F (ShelxL),
35
36
−
1
̃
3063 (9), 2643 (2), 1621 (14), 1548 (36), 1364 (100),
2
37,38
and finally checked using the PLATON
39
40
software integrated in the WinGX software suite. The non-
hydrogen atoms were refined anisotropically, and the hydrogen atoms
were located and freely refined. All Diamond 3 plots are shown with
thermal ellipsoids at the 50% probability level; hydrogen atoms are
shown as small spheres of arbitrary radius.
6 2 3 6
2
,4,6-Trinitrophenyl Methanesulfonate. Potassium picrate
ASSOCIATED CONTENT
Supporting Information
(
10.0 g, 37.4 mmol, 1.0 equiv) was suspended in 200 mL of toluene.
■
Methanesulfonic anhydride (6.5 g, 37.4 mmol, 1.0 equiv) was added
to the yellow suspension in small portions. After perchloric acid (60%
solution in acetic acid, 10 drops) was added, the reaction mixture was
sı
*
stirred for 4 h at 120 °C with a CaCl -filled drying tube on top of the
2
1H, 13C, 19F, and 14N NMR spectra, crystallographic
information, and detonation parameter calculations
reflux condenser. After cooling to room temperature, the yellowish
byproduct potassium mesylate was separated by filtration and the
filter residue was washed with ∼150 mL of toluene. The yellow filtrate
phases were combined, and the solvent was removed in vacuo. The
resulting yellow-orange oil was treated with ∼15 mL of ice-cold ether
to precipitate the solid product. The solvents were removed in vacuo,
and a pale yellow solid was obtained. It was recrystallized from 150
mL of chloroform to afford the product as a white solid (8.6 g, yield
Cambridge Crystallographic Data Centre, 12 Union Road,
Cambridge CB2 1EZ, UK; fax: +44 1223 336033.
7
5%).
1
H NMR (acetone-d , 400 MHz): δ 9.24 (s, 2H), 3.69 (s, 3H)
6
13
1
ppm. C{ H} NMR (acetone-d , 100 MHz): δ 146.3, 145.8, 138.8,
6
1
26.0, 40.4 ppm. 14N NMR (acetone-d , 29 MHz): δ −22 (s, NO )
−1
6
2
ppm. IR (ATR, cm ): ν
543 (m), 1373 (m), 1343 (s), 1243 (m), 1192 (m), 1169 (m), 1087
w), 969 (w), 919 (m), 847 (s), 823 (m), 792 (m), 775 (s), 730 (s),
16 (s), 668 (s), 634 (m), 560 (m), 536 (s), 505 (s). Raman (1064
̃
3095 (w), 2163 (w), 2004 (w), 1614 (w),
Corresponding Author
1
(
7
■
Thomas M. Klapotke − Department of Chemistry, Ludwig
̈
−
1
nm, 1000 mW, 25 °C, cm ): ν
̃
3097 (18), 3035 (15), 2942 (46),
616 (55), 1547 (42), 1363 (100), 1245 (50), 1089 (25), 823 (31),
36 (37), 565 (21), 367 (23), 330 (33), 250 (17). Anal. Calcd for
1
6
C H N O S: C, 27.37; H, 1.64; S, 10.44; N, 13.68; O, 46.87. Found:
7
5
3
9
C, 27.34; H, 1.69; N, 13.60; S, 10.99. DTA: 139 °C (mp), 237 °C
dec). IS: 9 J. FS: >360 N. ESD: 50 mJ.
,4,6-Trinitrobenzaldehyde. Trinitrotoluene (10.0 g, 44.0
Authors
(
Dominik E. Dosch − Department of Chemistry, Ludwig
Marco Reichel − Department of Chemistry, Ludwig
Max Born − Department of Chemistry, Ludwig Maximilian
University of Munich, D-81377 Munich, Germany
Konstantin Karaghiosoff − Department of Chemistry, Ludwig
2
mmol, 1 equiv) and N,N-dimethyl-p-nitrosoaniline (7.0 g, 47.0
mmol, 1.1 equiv) were dissolved in 15 mL of pyridine with 50 mg of
iodine as a catalyst. The mixture was stirred for 7 days at 20−25 °C.
The organic solid was separated by filtration and washed with cold
acetone. The obtained crude product was dried in vacuo and then
dissolved in 180 mL of aqueous HCl (36%) and stirred at 80 °C for 2
h. The product was then separated by filtration and washed acid-free
with water. This way 6.4 g (yield 61%) of pure TNBA could be
obtained as a black solid.
1
H NMR (DMSO-d , 400 MHz): δ 10.55 (s, 1H), 9.16 (s, 2H)
6
ppm. 13C{ H} NMR (DMSO-d , 100 MHz): δ 188.1, 148.1, 147.7,
34.2, 124.7 ppm. N (DMSO-d , 29 MHz): δ −20 (s, NO ) ppm.
IR (ATR, cm ): ν
1
6
14
1
6
2
−1
̃
3096 (w), 2916 (vw), 1714 (m), 1605 (m), 1554
(
s), 1535 (vs), 1452 (w), 1405 (w), 1343 (vs), 1194 (m), 1185 (w),
Author Contributions
1
7
157 (m), 1075 (w), 979 (w), 935 (w), 919 (m), 839 (m), 826 (m),
72 (m), 742 (m), 729 (s), 701 (m), 570 (w), 538 (w), 475 (w), 435
†
D.E.D., M.R., and M.B. contributed equally to this work.
−
1
(m). Raman (1064 nm, 1000 mW, 25 °C, cm ): ν 3104 (8), 2911
̃
(7), 2903 (8), 1713 (16), 1623 (15), 1553 (29), 1382 (32), 1351
(81), 1274 (10), 1197 (12), 981 (8), 938 (7), 842 (13), 826 (18),
Notes
The authors declare no competing financial interest.
3
8
34 (22), 292 (8), 255 (7), 232 (19), 203 (34), 192 (26), 151 (36),
8 (100). Anal. Calcd for C H N O : C, 34.87; H, 1.25; N, 17.43.
ACKNOWLEDGMENTS
Financial support of this work by Ludwig-Maximilian
University (LMU), the Office of Naval Research (ONR)
■
7
3
3
7
Found: C, 34.87; H, 1.38; N, 17.12. DTA: 102 °C (mp), 185 °C
(
dec). IS: 10 J. FS: 360 N. ESD: 120 mJ.
E
Cryst. Growth Des. XXXX, XXX, XXX−XXX

 Dosch, Dominik E.
Dosch, Dominik E.
 Reichel, Marco
Reichel, Marco
 Born, Max
Born, Max
 Klap?tke, Thomas M.
Klap?tke, Thomas M.
 Karaghiosoff, Konstantin
Karaghiosoff, Konstantin