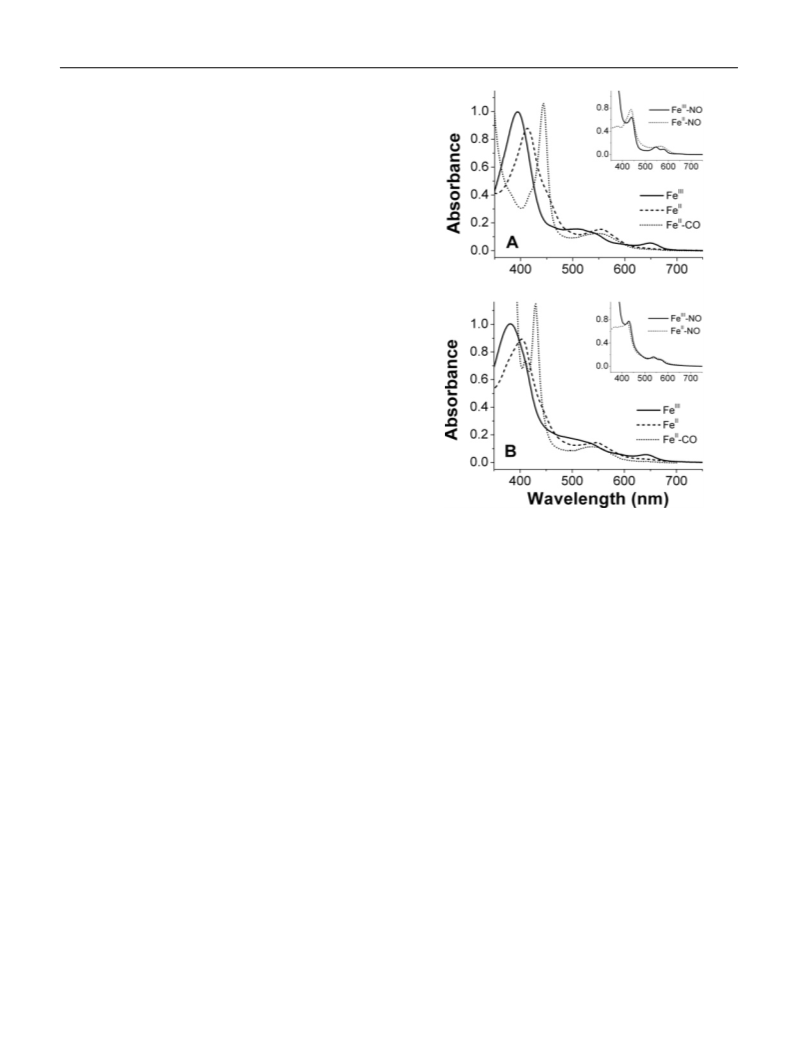
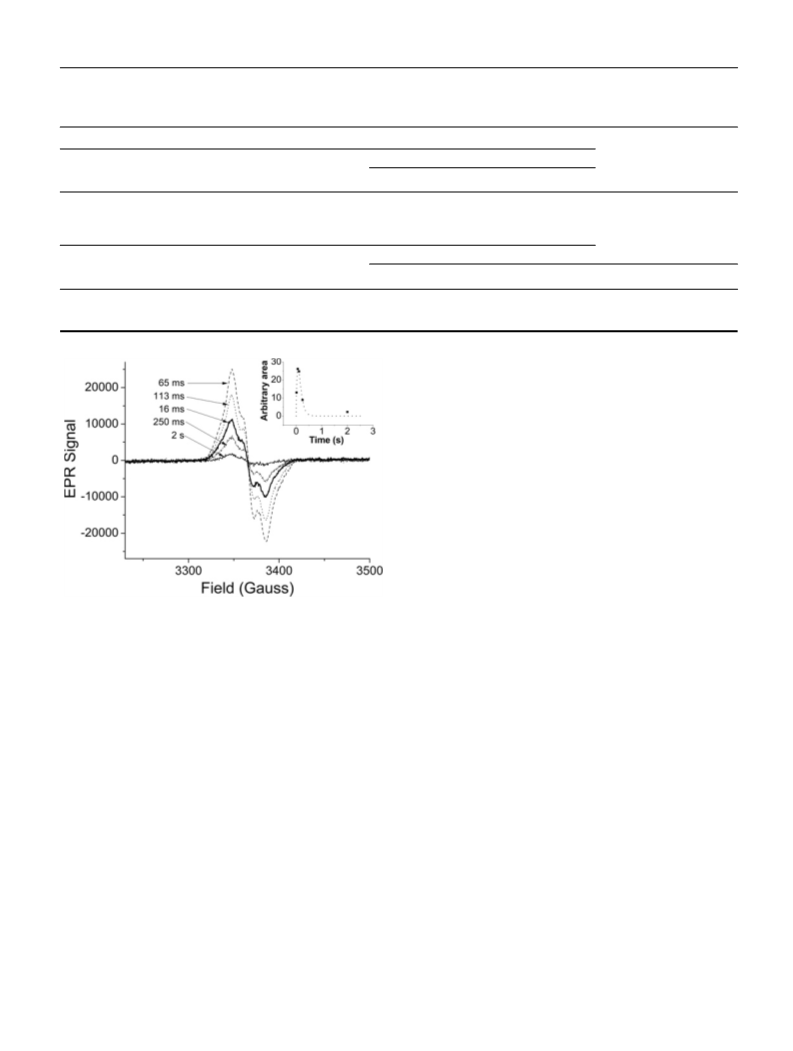
Mesohaem-substituted neuronal NOS
173
18 Yamada, H., Makino, R. and Yamazaki, I. (1975) Effects of 2,4-substituents of
deuteropheme upon redox potentials of horseradish peroxidases. Arch. Biochem.
Biophys. 169, 344–353
haem electronics can affect multiple kinetic parameters in nNOS,
and how these changes combine to alter its catalytic behaviours.
19 Flaherty, M. M., Rish, K. R., Smith, A. and Crumbliss, A. L. (2008) An investigation of
hemopexin redox properties by spectroelectrochemistry: biological relevance for heme
uptake. Biometals 21, 239–248
20 Ignarro, L. J., Ballot, B. and Wood, K. S. (1984) Regulation of soluble guanylate cyclase
activity by porphyrins and metalloporphyrins. J. Biol. Chem. 259, 6201–6207
21 Santolini, J., Meade, A. L. and Stuehr, D. J. (2001) Differences in three kinetic parameters
underpin the unique catalytic profiles of nitric-oxide synthases I, II, and III. J. Biol. Chem.
276, 48887–48898
22 Santolini, J., Adak, S., Curran, C. M. and Stuehr, D. J. (2001) A kinetic simulation model
that describes catalysis and regulation in nitric-oxide synthase. J. Biol. Chem. 276,
1233–1243
23 Adak, S. and Stuehr, D. J. (2001) A proximal tryptophan in NO synthase controls activity
by a novel mechanism. J. Inorg. Biochem. 83, 301–308
AUTHOR CONTRIBUTION
Jesu´s Tejero, Ashis Biswas, Mohammad Mahfuzul Haque and Zhi-Qiang Wang performed
experiments and analysed data. Craig Hemann performed the EPR experiments. Russ
Hille provided the EPR equipment. Cornelius Varnado and Douglas Goodwin contributed
the haem transporter protein and technical advice. Zachary Novince developed the
expression protocol for the mesohaem-containing proteins. Jesu`s Tejero, Ashis Biswas
and Mohammad Mahfuzul drafted the manuscript. Dennis Stuehr designed the research,
analysed data and wrote the final manuscript.
24 Adak, S., Wang, Q. and Stuehr, D. J. (2000) Molecular basis for hyperactivity in
tryptophan 409 mutants of neuronal NO synthase. J. Biol. Chem. 275, 17434–17439
25 Adak, S., Crooks, C., Wang, Q., Crane, B. R., Tainer, J. A., Getzoff, E. D. and Stuehr, D. J.
(1999) Tryptophan 409 controls the activity of neuronal nitric-oxide synthase by
regulating nitric oxide feedback inhibition. J. Biol. Chem. 274, 26907–26911
26 Tejero, J., Biswas, A., Wang, Z. Q., Page, R. C., Haque, M. M., Hemann, C., Zweier, J. L.,
Misra, S. and Stuehr, D. J. (2008) Stabilization and characterization of a heme-oxy
reaction intermediate in inducible nitric-oxide synthase. J. Biol. Chem. 283,
33498–33507
FUNDING
This work was supported by the National Institutes of Health [grant numbers CA53914,
GM51491 (to D.J.S.), GM 075036 (to R.H.)] and American Heart Association postdoctoral
fellowships [grant numbers 0825545D (to A.B.), 0625632B (to J.T.)].
REFERENCES
27 Lang, J., Driscoll, D., Gelinas, S., Rafferty, S. P. and Couture, M. (2009) Trp180 of
endothelial NOS and Trp56 of bacterial saNOS modulate σ bonding of the axial cysteine to
the heme. J. Inorg. Biochem. 103, 1102–1112
1
Pfeiffer, S., Mayer, B. and Hemmens, B. (1999) Nitric oxide: chemical puzzles posed by a
biological messenger. Angew. Chem. Int. Ed. 38, 1714–1731
2
Garcin, E. D., Bruns, C. M., Lloyd, S. J., Hosfield, D. J., Tiso, M., Gachhui, R., Stuehr, D.
J., Tainer, J. A. and Getzoff, E. D. (2004) Structural basis for isozyme-specific regulation
of electron transfer in nitric-oxide synthase. J. Biol. Chem. 279, 37918–37927
Xia, C., Misra, I., Iyanagi, T. and Kim, J. J. (2009) Regulation of interdomain interactions
by calmodulin in inducible nitric-oxide synthase. J. Biol. Chem. 284, 30708–30717
Crane, B. R., Arvai, A. S., Ghosh, D. K., Wu, C., Getzoff, E. D., Stuehr, D. J. and Tainer, J.
A. (1998) Structure of nitric oxide synthase oxygenase dimer with pterin and substrate.
Science 279, 2121–2126
28 Varnado, C. L. and Goodwin, D. C. (2004) System for the expression of recombinant
hemoproteins in Escherichia coli. Protein Expression Purif. 35, 76–83
29 Abu-Soud, H. M., Gachhui, R., Raushel, F. M. and Stuehr, D. J. (1997) The ferrous-dioxy
complex of neuronal nitric oxide synthase. Divergent effects of L-arginine and
tetrahydrobiopterin on its stability. J. Biol. Chem. 272, 17349–17353
30 Adak, S., Ghosh, S., Abu-Soud, H. M. and Stuehr, D. J. (1999) Role of reductase domain
cluster 1 acidic residues in neuronal nitric-oxide synthase. Characterization of the
FMN-free enzyme. J. Biol. Chem. 274, 22313–22320
31 Ilagan, R. P., Tiso, M., Konas, D. W., Hemann, C., Durra, D., Hille, R. and Stuehr, D. J.
(2008) Differences in a conformational equilibrium distinguish catalysis by the endothelial
and neuronal nitric-oxide synthase flavoproteins. J. Biol. Chem. 283, 19603–19615
32 Presta, A., Weber-Main, A. M., Stankovich, M. T. and Stuehr, D. (1998) Comparative
effects of substrates and pterin cofactor on the heme midpoint potential in inducible and
neuronal nitric oxide synthases. J. Am. Chem. Soc. 120, 9460–9465
33 Panda, K., Haque, M. M., Garcin-Hosfield, E. D., Durra, D., Getzoff, E. D. and Stuehr, D. J.
(2006) Surface charge interactions of the FMN module govern catalysis by nitric-oxide
synthase. J. Biol. Chem. 281, 36819–36827
34 Haque, M. M., Panda, K., Tejero, J., Aulak, K. S., Fadlalla, M. A., Mustovich, A. T. and
Stuehr, D. J. (2007) A connecting hinge represses the activity of endothelial nitric oxide
synthase. Proc. Natl. Acad. Sci. U.S.A. 104, 9254–9259
35 Wei, C. C., Wang, Z. Q., Wang, Q., Meade, A. L., Hemann, C., Hille, R. and Stuehr, D. J.
(2001) Rapid kinetic studies link tetrahydrobiopterin radical formation to heme-dioxy
reduction and arginine hydroxylation in inducible nitric-oxide synthase. J. Biol. Chem.
276, 315–319
3
4
5
6
Abu-Soud, H. M., Yoho, L. L. and Stuehr, D. J. (1994) Calmodulin controls neuronal
nitric-oxide synthase by a dual mechanism. Activation of intra- and interdomain electron
transfer. J. Biol. Chem. 269, 32047–32050
Stuehr, D. J., Santolini, J., Wang, Z. Q., Wei, C. C. and Adak, S. (2004) Update on
mechanism and catalytic regulation in the NO synthases. J. Biol. Chem. 279,
36167–36170
7
8
9
Gorren, A. C. F. and Mayer, B. (2007) Nitric-oxide synthase: a cytochrome P450 family
foster child. Biochim. Biophys. Acta 1770, 432–445
Wei, C. C., Crane, B. R. and Stuehr, D. J. (2003) Tetrahydrobiopterin radical enzymology.
Chem. Rev. 103, 2365–2383
Bender, A. T., Kamada, Y., Kleaveland, P. A. and Osawa, Y. (2002) Assembly and activation
of heme-deficient neuronal NO synthase with various porphyrins. J. Inorg. Biochem. 91,
625–634
10 Woodward, J. J., Martin, N. I. and Marletta, M. A. (2007) An Escherichia coli
expression-based method for heme substitution. Nat. Methods 4, 43–45
11 Tamura, M., Asakura, T. and Yonetani, T. (1973) Heme modification studies of myoglobin.
I. Purification and some optical and EPR characteristics of synthesized myoglobins
containing unnatural hemes. Biochim. Biophys. Acta 295, 467–479
12 Modi, S., Primrose, W. U., Lian, L. Y. and Roberts, G. C. (1995) Effect of replacement of
ferriprotoporphyrin IX in the haem domain of cytochrome P-450 BM-3 on substrate
binding and catalytic activity. Biochem. J. 310, 939–943
36 Wei, C. C., Wang, Z. Q. and Stuehr, D. J. (2002) Nitric oxide synthase: use of
stopped-flow spectroscopy and rapid-quench methods in single-turnover conditions to
examine formation and reactions of heme-O2 intermediate in early catalysis. Methods
Enzymol. 354, 320–338
37 Boggs, S., Huang, L. and Stuehr, D. J. (2000) Formation and reactions of the
heme-dioxygen intermediate in the first and second steps of nitric oxide synthesis as
studied by stopped-flow spectroscopy under single-turnover conditions. Biochemistry
39, 2332–2339
13 Antonini, E., Brunori, M., Caputo, A., Chiancone, E., Fanelli, A. R. and Wyman, J. (1964)
Studies on the structure of hemoglobin. III. Physicochemical properties of reconstituted
hemoglobins. Biochim. Biophys. Acta 79, 284–292
14 Ryabova, E. S., Rydberg, P., Kolberg, M., Harbitz, E., Barra, A. L., Ryde, U., Andersson, K.
K. and Nordlander, E. (2005) A comparative reactivity study of microperoxidases based on
hemin, mesohemin and deuterohemin. J. Inorg. Biochem. 99, 852–863
15 Makino, R. and Yamazaki, I. (1972) Effects of 2,4-substituents of deuterohemin upon
peroxidase functions. I. Preparation and some properties of artificial enzymes. J.
Biochem. (Tokyo) 72, 655–664
16 Singh, U. P., Obayashi, E., Takahashi, S., Iizuka, T., Shoun, H. and Shiro, Y. (1998) The
effects of heme modification on reactivity, ligand binding properties and iron-coordination
structures of cytochrome P450nor. Biochim. Biophys. Acta 1384, 103–111
17 Brunori, M., Saggese, U., Rotilio, G. C., Antonini, E. and Wyman, J. (1971) Redox
equilibrium of sperm-whale myoglobin, Aplysia myoglobin, and Chironomus thummi
hemoglobin. Biochemistry 10, 1604–1609
38 Abu-Soud, H. M., Rousseau, D. L. and Stuehr, D. J. (1996) Nitric oxide binding to the
heme of neuronal nitric-oxide synthase links its activity to changes in oxygen tension.
J. Biol. Chem. 271, 32515–32518
39 Salerno, J. C. and Ghosh, D. K. (2009) Space, time and nitric oxide: neuronal nitric oxide
synthase generates signal pulses. FEBS J. 276, 6677–6688
40 Wei, C. C., Wang, Z. Q., Tejero, J., Yang, Y. P., Hemann, C., Hille, R. and Stuehr, D. J.
(2008) Catalytic reduction of a tetrahydrobiopterin radical within nitric-oxide synthase.
J. Biol. Chem. 283, 11734–11742
41 Haque, M. M., Fadlalla, M., Wang, Z. Q., Ray, S. S., Panda, K. and Stuehr, D. J. (2009)
Neutralizing a surface charge on the FMN subdomain increases the activity of neuronal
nitric-oxide synthase by enhancing the oxygen reactivity of the enzyme heme-nitric oxide
complex. J. Biol. Chem. 284, 19237–19247
ꢁ
c
ꢁ
c
The Authors Journal compilation 2011 Biochemical Society

 Tejero, Jesus
Tejero, Jesus
 Biswas, Ashis
Biswas, Ashis
 Haque, Mohammad Mahfuzul
Haque, Mohammad Mahfuzul
 Wang, Zhi-Qiang
Wang, Zhi-Qiang
 Hemann, Craig
Hemann, Craig
 Varnado, Cornelius L.
Varnado, Cornelius L.
 Novince, Zachary
Novince, Zachary
 Hille, Russ
Hille, Russ
 Goodwin, Douglas C.
Goodwin, Douglas C.
 Stuehr, Dennis J.
Stuehr, Dennis J.