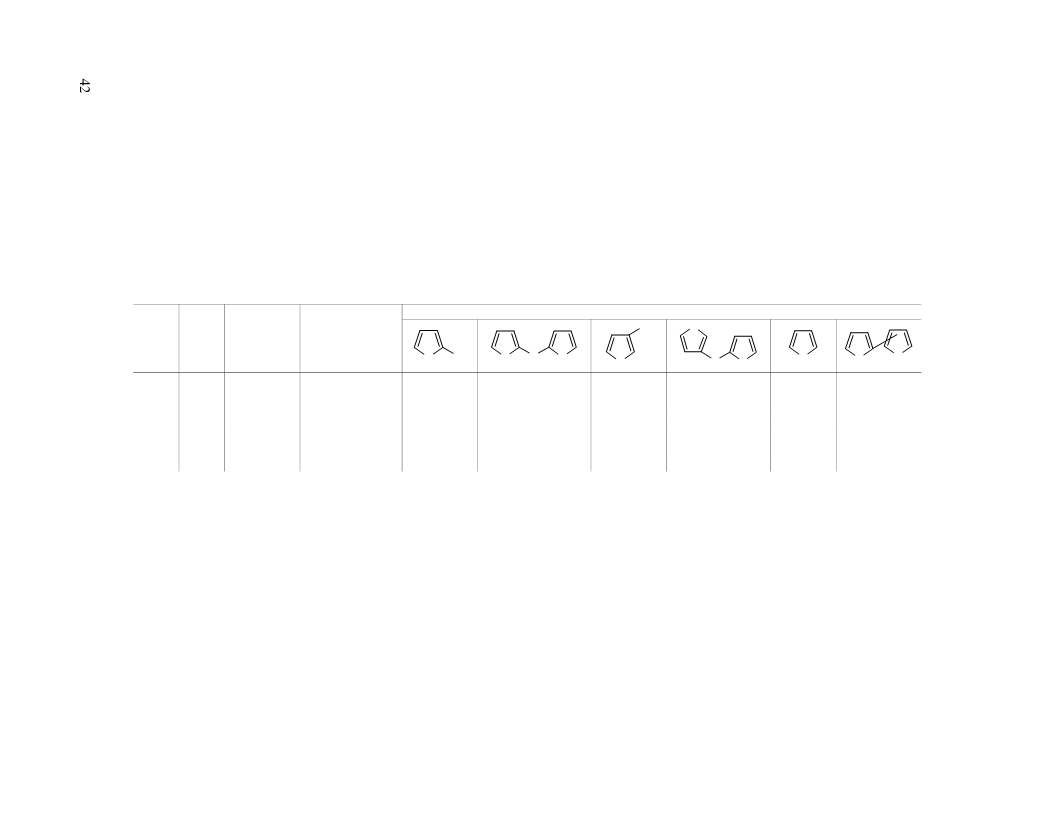
dimethylselenide the selectivity of formation of 2-thiophenethiol can be improved with 100% conversion of the
2-chlorothiophene. However, the yield of the thiol 1 obtained does not exceed 10% (with a 1% yield of sulfide
2) due to the marked reduction of the 2-chlorothiophene to thiophene (yield 46%).
With the aim of increasing the selectivity of formation of the 2-thiophenethiol in the reaction of
2-chlorothiophene with hydrogen sulfide we have introduced methanol into the reaction, since it is a
more powerful hydrogen atom donor than is hydrogen sulfide. This is related to the greater stability of
alkoxyl radicals which are readily converted to carbonyl compounds [3]. For comparison, the effect of
ethanol on the course of the reaction of 2-chlorothiophene with hydrogen sulfide has been studied (Tables 1
and 2).
The reaction was carried out in a hollow quartz tube at a feed velocity of 2-chlorothiophene in the
mixture with the alcohols of 10 ml/h and of hydrogen sulfide of 6 l/h. The methanol was added in amounts of 5,
10, 25, and 50 mole % and ethanol 10 and 25 mole %. Methanol at the 5 mole % level had virtually no effect on
the course of the reaction of 2-chlorothiophene with hydrogen sulfide at 490-530°C and resulted in the principal
formation of the bis(2-thienyl) sulfide (2) with a low conversion of the 2-chlorothiophene. However, at
550-570°C there is a sharp increase in the conversion of the chloro derivative (to 98%) and the 2-thiophenethiol
1 at 570°C becomes the main reaction product (the yield of the thiol 1 was 34% and the sulfide 2 26%). At the
same time, side products of the reaction are formed and these include thiophene (24%).
In the presence of 10 mole % of methanol a marked conversion of the 2-chlorothiophene (25%) is seen
even at 490°C but at this temperature only the sulfide (2) is produced. With an increase in temperature to 590°C
the conversion of the 2-chlorothiophene increases to 100%, increasing the yield of the thiol 1 and the sulfide 2
reaction products; moreover the yield of the thiol now exceeds that of the sulfide at 570-590°C. The optimum
conditions were achieved in the presence of 10 mole % methanol at 570°C when the conversion of the
2-chlorothiophene in reaction with hydrogen sulfide was 98% with a 43% yield of thiol 1 and 31% yield of
sulfide 2. At a temperature above 550°C the conversion of the 2-chlorothiophene to thiophene is significantly
increased (15% at 570°C and 22.6% at 590°C). There appeared in small amounts the side products of the
reaction like 3-thiophenethiol, 2,3'-dithienyl sulfide, and isomeric bithiophenes and dithienothiophenes. Their
overall yield was 7-12%.
In the presence of 25 mole % methanol, the reaction according to Scheme 1 at 490°C occurs with
selective formation of only the one sulfide 2. Moreover, the conversion of the 2-chlorothiophene (19.6%) and
the yield of the sulfide (16.0%) were somewhat lower than in the presence of 10 mole % methanol. However, at
570°C the yield of the thiol 1 and sulfide 2 become approximately the same (38.9 and 40.7% respectively) with
a high conversion of the 2-chlorothiophene (96.0%). The positive effect of using 25 mole % methanol in the
reaction is the lowering at 570°C of the yield of thiophene to 7% and other side products to 5%. Using 50 mole
% methanol inhibits the Scheme 1 reaction at 570-610°C since the yield of the main thiylation products are
decreased and, at 570°C, there is a reduction in the conversion of the chloride. At higher temperatures a
significant amount of side products are observed, as a result of which the yields of the basic thiylation products
1 and 2 are reduced to 26.7 and 18.8% respectively (Table 1).
The presence of ethanol (10 or 25 mole %, 550-590°C) increases the conversion of the
2-chlorothiophene in the Scheme 1 reaction to 100% but it lowers the selectivity since the rate of side process
decomposition of starting materials and final reaction products is increased. The yield of thiol 1 and sulfide 2 at
570°C are only 28 and 39% respectively (Table 2). The high selectivity of the reaction for 2-thiophenethiol in
the presence of methanol (Scheme 1) is evidently related to the higher H-donor property than for hydrogen
sulfide, hindering the formation of 2-thienylthiyl radicals from the thiol 1 and thus slowing the second stage of
the reaction involving formation of sulfide 2 according to Scheme 2.
43

 Deryagina
Deryagina
 Sukhomazova
Sukhomazova
 Levanova
Levanova