Li et al.
FULL PAPER
was concentrated under reduced pressure to give com-
[18] (a) Fukase, H.; Horii, S. J. Org. Chem. 1992, 57, 3651; (b) Fukase,
H.; Horii, S. J. Org. Chem. 1992, 57, 3642; (c) Hayashida, M.; Sa-
kairi, N.; Kuzuhara, H. J. Carbohydr. Chem. 1988, 7, 83; (c) Shing,
T. K. M.; Cheng, H. M. Org. Lett. 2008, 10, 4137.
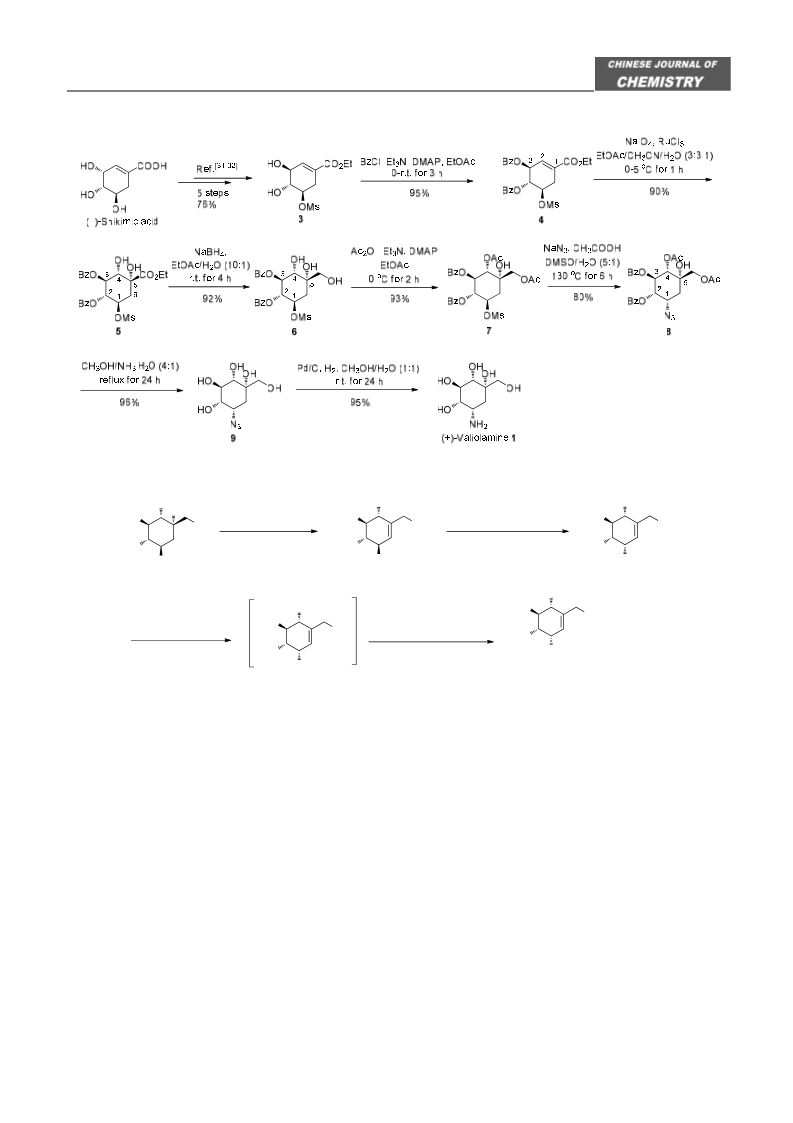
pound 2 (160.0 mg, 0.9133 mmol) in 91% yield as a
2
D
0
[17]
pale-yellow syrup. [α] +88.9 (c 0.35, H O) {ref.
2
23
[
D
2
α] +90 (c 0.43, H O)}. Characterization data are
[19] (a) Chang, Y.-K.; Lee, B.-Y.; Kim, D. J.; Lee, G. S.; Jeon, H. B.; Kim,
K. S. J. Org. Chem. 2005, 70, 3299; (b) Schmidt, R. R.; Kohn, A.
Angew. Chem., Int. Ed. 1987, 26, 482; (c) Kapferer, P.; Sarabia, F.;
Vasella, A. Helv. Chim. Acta 1999, 82, 645; (d) Cumpstey, I.;
Gehrke, S.; Erfan, S.; Cribiu, R. Carbohydr. Res. 2008, 343, 1675;
(e) Yoshikawa, M.; Cha, B. C.; Okaichi, Y.; Takinami, Y.; Yokokawa,
Y.; Kitagawa, I. Chem. Pharm. Bull. 1988, 36, 4236; (f) Li, Q. R.;
Kim, S. I.; Park, S. J.; Yang, H. R.; Baek, A. R.; Kim, I. S.; Jung, Y.
H. Tetrahedron 2013, 69, 10384; (g) Knapp, S.; Naughton, A. B. J.;
Dhar, T. G. M. Tetrahedron Lett. 1992, 33, 1025; (h) Park, T. K.;
Danishefsky, S. J. Tetrahedron Lett. 1994, 35, 2667; (i) Cumpstey, I.
Tetrahedron Lett. 2005, 46, 6257.
[32]
identical with the reported data.
Supporting Information
1
13
H and C NMR spectra of compounds 4-8, 10
and 11.
Acknowledgement
We thank the National Natural Science Foundation
of China (No. 20972048) for the financial support of
this work.
[
[
20] Kumar, B. S.; Mishra, G. P.; Rao, B. V. Tetrahedron 2016, 72, 1838.
21] Lo, H.-J.; Chen, C.-Y.; Zheng, W.-L.; Yeh, S.-M.; Yan, T.-H. Eur. J.
Org. Chem. 2012, 2780.
[
[
22] Chang, Y.-K.; Lo, H.-J.; Yan, T.-H. Org. Lett. 2009, 11, 4278.
23] (a) Ogawa, S.; Ohishi, Y.; Asada, M.; Tomoda, A.; Takahashi, A.;
Ooki, Y.; Mori, M.; Itoh, M.; Korenaga, T. Org. Biomol. Chem. 2004,
2, 884; (b) Ogawa, S.; Kanto, M. J. Nat. Prod. 2007, 70, 493; (c)
Jagdhane, R. C.; Shashidhar, M. S. Tetrahedron 2011, 67, 7963.
References
[
[
1] Mahmud, T. Nat. Prod. Rep. 2003, 20, 137.
2] Arjona, O.; Gómez, A. M.; López, J. C.; Plumet, J. Chem. Rev. 2007,
1
07, 1919.
[
[
3] Chen, X.; Fan, Y.; Zheng, Y.; Shen, Y. Chem. Rev. 2003, 103, 1955.
4] Kameda, Y.; Asano, N.; Yoshikawa, M.; Takeuchi, M.; Yamaguchi,
T.; Matsui, K.; Horii, S.; Fukase, H. J. Antibiot. 1984, 37, 1301.
5] Kameda, Y.; Horii, S. J. Chem. Soc., Chem. Commun. 1972, 746.
6] (a) Takeuchi, M.; Kamata, K.; Yoshida, M.; Kameda, Y.; Matsui, K.
J. Biochem. 1990, 108, 42; (b) Blidi, L. E.; Ahbala, M.; Bolte, J.;
Lemaire, M. Tetrahedron: Asymmetry 2006, 17, 2684.
[24] Mondal, S.; Prathap, A.; Sureshan, K. M. J. Org. Chem. 2013, 78,
7690.
[25] (a) Shing, T. K. M.; Wan, L. H. Angew. Chem., Int. Ed. 1995, 34,
1643; (b) Shing, T. K. M.; Wan, L. H. J. Org. Chem. 1996, 61, 8468.
[26] (a) Kok, S. H. L.; Lee, C.-C.; Shing, T. K. M. J. Org. Chem. 2001,
66, 7184; (b) Shing, T. K. M.; Li, T.-Y.; Kok, S. H. L. J. Org. Chem.
1999, 64, 1941.
[
[
[
[
[
7] Takeuchi, M.; Takai, N.; Asano, N.; Kameda, Y.; Matsui, K. Chem.
Pharm. Bull. 1990, 38, 1970.
8] (a) Ghani, U. Eur. J. Med. Chem. 2015, 103, 133; (b) Fukase, H.;
Horii, S. J. Org. Chem. 1992, 57, 3642.
9] (a) Zhou, T.-C.; Zhong, J.-J. Bioresour. Technol. 2015, 175, 160; (b)
Ogawa, S.; Nose, T.; Ogawa, T.; Toyokuni, T.; lwasawa, Y.; Suami, T.
J. Chem. Soc., Perkin Trans. 1 1985, 2369.
[27] (a) Paulsen, H.; Heiker, F. R. Angew. Chem., Int. Ed. 1980, 19, 904;
(b) Paulsen, H.; Heiker, F. R. Liebigs Ann. Chem. 1981, 2180.
[28] (a) Krishna, P. R.; Reddy, P. S. Synlett 2009, 209; (b) Zhou, B.; Luo,
Z.; Lin, S.; Li, Y. Synlett 2012, 913.
[29] Trost, B. M.; Chupak, L. S.; Luebbers, T. J. Am. Chem. Soc. 1998,
120, 1732.
[30] (a) Enrich, L. B.; Scheuermann, M. L.; Mohadjer, A.; Matthias, K.
R.; Eller, C. F.; Newman, M. S.; Fujinaka, M.; Poon, T. Tetrahedron
Lett. 2008, 49, 2503; (b) Ohira, H.; Torii, N.; Aida, T. M.; Watanabe,
M.; Smith, R. L. Sep. Purif. Technol. 2009, 69, 102; (c) Ghosh, S.;
Chisti, Y.; Banerjee, V. C. Biotech. Adv. 2012, 30, 1425; (d) Nie,
L.-D.; Shi, X.-X. Tetrahedron: Asymmetry 2009, 20, 124; (e) Nie,
L.-D.; Ding, W.; Shi, X.-X.; Quan, N.; Lu, X. Tetrahedron:
Asymmetry 2012, 23, 742; (f) Quan, N.; Nie, L.; Shi, X.; Zhu, R.; Lu,
X. Chin. J. Chem. 2012, 30, 2759.
[
10] Kemeda, Y.; Asano, N.; Yamaguchi, T.; Matsui, K.; Horii, S.; Fukase,
H. J. Antibiot. 1986, 39, 1691.
11] Miyamoto, Y.; Ogawa, S. J. Chem. Soc., Chem. Commun. 1990, 999.
12] (a) Ogawa, S.; Shibata, Y. J. Chem. Soc., Chem. Commun. 1988, 605;
[
[
(
b) Arakawa, K.; Bowers, G. S.; Michels, B.; Trin, V.; Mahmud, T.
Carbohydr. Res. 2003, 338, 2075; (c) Truscheit, E.; Frommer, W.;
Junge, B.; Muller, L.; Schmidt, D. D.; Wingender, W. Angew. Chem.,
Int. Ed. 1981, 20, 744.
[
[
13] (a) Ogawa, S.; Iwasawa, Y.; Toyokuni, T.; Suami, T. Carbohydr. Res.
[31] Quan, N.; Nie, L.-D.; Zhu, R.-H.; Shi, X.-X.; Ding, W.; Lu, X. Eur.
J. Org. Chem. 2013, 6389.
[32] Ding, W.; Yu, J.-P.; Shi, X.-X.; Nie, L.-D.; Li, F.-L. Tetrahedron:
Asymmetry 2015, 26, 1037.
[33] (a) Shing, T. K. M.; Tai, V. W.-F.; Tam, E. K. W. Angew. Chem., Int.
Ed. 1994, 33, 2312; (b) Shing, T. K. M.; Tam, E. K. W.; Tai, V. W.-F.;
Chung, I. H. F.; Jang, Q. Chem. Eur. J. 1996, 2, 50; (c) Plietker, B.;
Niggemann, M. Org. Lett. 2003, 5, 3353.
[34] (a) Dalla, V.; Cotelle, P.; Catteau, J. P. Tetrahedron Lett. 1997, 38,
1577; (b) Dalla, V.; Catteau, J. P.; Pale, P. Tetrahedron Lett. 1999, 40,
5193.
1
985, 141, 29; (b) Namiki, S.; Kangouri, K.; Nagate, T.; Hara, H.;
Sugita, K.; Omura, S. J. Antibiot. 1982, 35, 1234.
14] (a) Li, C.; Hung, Y.-J.; Qamruddin, K.; Aziz, M. F. A.; Stein, H.;
Schmidt, B. Diabetes Res. Clin. Pract. 2011, 92, 57; (b) Aoki, K.;
Ito, Y.; Saito, K.; Shirakawa, J.; Togashi, Y.; Satoh, K.; Muraoka, T.;
Shinoda, K.; Masuda, K.; Kimura, M.; Terauchi, Y. Diabetes Res.
Clin. Pract. 2009, 83, e31; (c) Yan, L.; Xu, H.; Liu, F.; Zhao, J.; Liu,
H. Chin. J. Chem. 2012, 30, 914.
[
[
[
15] Ji, L.; Wu, G.; Ye, W.; Chen, X. Chin. J. Org. Chem. 2016, 36, 2771
(
in Chinese).
16] Sellier, O.; Weghe, P. V.; Nouen, D. L.; Strehler, C.; Eustache, J.
Tetrahedron Lett. 1999, 40, 853.
17] Tatsuta, K.; Mukai, H.; Takahashi, M. J. Antibiot. 2000, 53, 430.
[35] Ivanov, I. V. S.; Romanov, G.; Shevchenko, V. P.; Rozhkova, E. A.;
Maslov, M. A.; Groza, N. V.; Myasoedov, N. F.; Kuhn, H.; My-
agkova, G. I. Tetrahedron 2003, 59, 8091.
(Zhao, X.)
8
www.cjc.wiley-vch.de
© 2017 SIOC, CAS, Shanghai, & WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
Chin. J. Chem. 2017, XX, 1—8

 Li, Fenglei
Li, Fenglei
 Ding, Wei
Ding, Wei
 Quan, Na
Quan, Na
 Wu, Jiajia
Wu, Jiajia
 He, Yungang
He, Yungang
 Zhu, Xingliang
Zhu, Xingliang
 Shi, Xiaoxin
Shi, Xiaoxin
 Zhao, Jianhong
Zhao, Jianhong