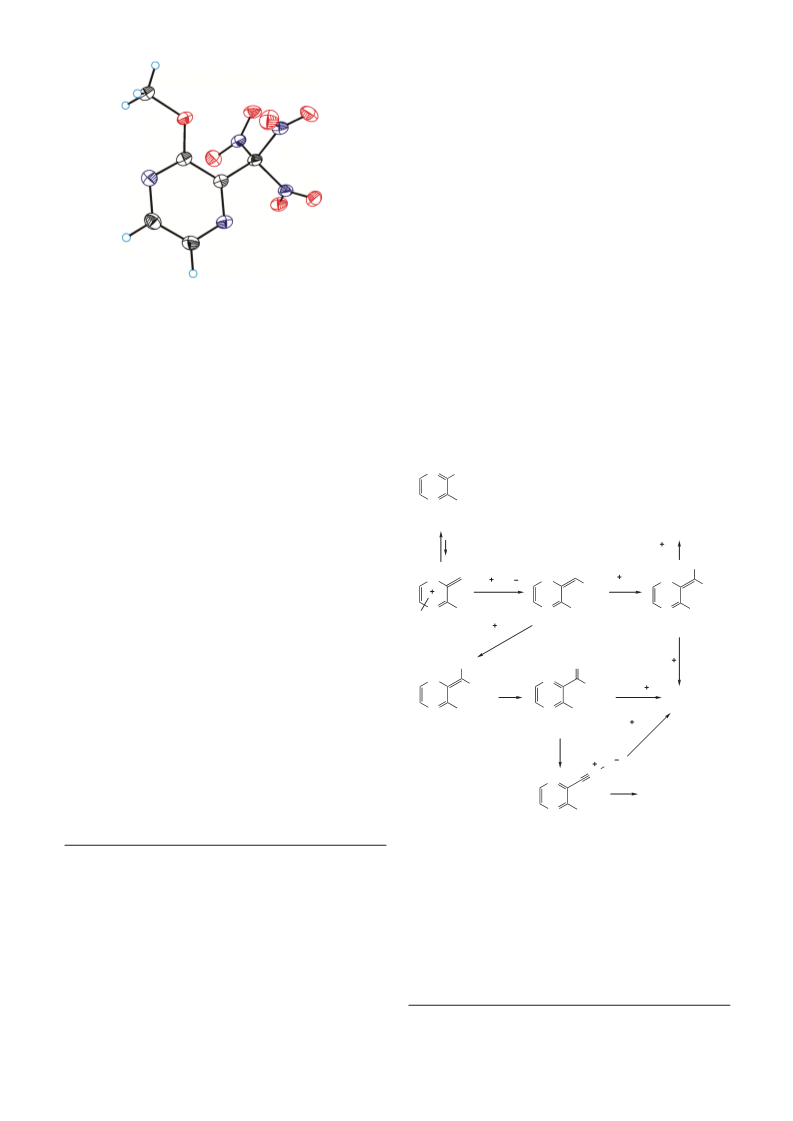
Mendeleev Commun., 2015, 25, 193–195
and coworkers prepared 2,4dimethoxy6trinitromethyl1,3,5
triazine (57%) from 2,4dimethoxy6methyl1,3,5triazine using
7 (a)Y.N. Li, Z.Z. Zhang, Y.S. Zhou, B. Chen and B.Z. Wang, Chin. J.
Explos. Propellants, 2009, 32 (6), 40; (b) L. S. Postnikov, Chem. Nat.
Compd., 2010, 46, 72 (Khim. Prirod. Soedin., 2010, 64); (c) Y.N. Li,
Z.Z. Zhang, Y.P. Ji and Y.I. Wang, Hanneng Cailiao (Chin. J. Energ.
Mater.), 2010, 18 (1), 7; (d) Y. Li, Z. Zhang, Y. Zhou, B. Wang and
Y. Shang, Acta Chim. Sinica, 2011, 69, 701.
17
8
HNO /H SO mixture. More recently, Katritzky et al. obtained
3
2
4
5
methyl2trinitromethylpyridine in 10% yield on the nitration
of 2,5lutidine with nitric acid in trifluoroacetic anhydride. No
previous installation of trinitromethyl group onto pyrazine ring
has been documented.
8
9
A. R. Katritzky, E. F. V. Scriven, S. Majumder, R. G. Akhmedova, A. V.
Vakulenko, N. G. Akhmedov, R. Murugan and K. A. Abboud, Org.
Biomol. Chem., 2005, 3, 538.
(a) A. B. Sheremetev, S. G. Zabusov, T. R. Tukhbatshin, N. V. Palysaeva
and K.Yu. Suponitsky, Chem. Heterocycl. Compd., 2014, 50, 1154 (Khim.
Geterotsikl. Soedin., 2014, 1250); (b) K.Yu. Suponitsky, K.A. Lyssenko,
I. V. Ananyev, A. M. Kozeev and A. B. Sheremetev, Cryst. Growth Des.,
The attachment of trinitromethyl groups to an azine backbone
is a highly desirable process but one of the most difficult to achieve
in practice. Studies on heterocycles bearing trinitromethyl group
have provided extensive information which is relevant to the
1
8
design and development of new high energetic materials. In
this regard, pyrazine derivatives are the benchmark family of
energetic materials whose properties can be tuned through the
chemical modification. The incorporation of trinitromethyl group
into the backbone provides deep insights into the structure–
property relationships.
2
014, 14, 4439.
10 (a) K.Yu. Suponitsky, K.A. Lyssenko, M.Yu.Antipin, N. S.Aleksandrova,
A. B. Sheremetev and T. S. Novikova, Russ. Chem. Bull., Int. Ed., 2009,
5
8, 2129 (Izv. Akad. Nauk, Ser. Khim., 2009, 2065); (b) K.Yu. Suponitsky,
A. E. Masunov and M. Yu. Antipin, Mendeleev Commun., 2009, 19, 311;
c) K.Yu. Suponitsky,Y. Liao andA. E. Masunov, J. Phys. Chem. A, 2009,
13, 10994; (d)A. B. Sheremetev, N. S. Aleksandrova, K.Yu. Suponitsky,
(
1
M.Yu. Antipin and V. A. Tartakovsky, Mendeleev Commun., 2010, 20, 249;
(e)A. B. Sheremetev, I. L.Yudin, N. V. Palysaeva and K.Yu. Suponitsky,
J. Heterocycl. Chem., 2012, 49, 394; (f) K. Yu. Suponitsky and A. E.
Masunov, J. Chem. Phys., 2013, 139, 094310; (g) A. V. Vologzhanina,
A. A. Golovanov, D. M. Gusev, I. S. Odin, R. A. Apreyan and K. Yu.
Suponitsky, Cryst. Growth Des., 2014, 14, 4402.
1 (a) A. B. Sheremetev, I. L.Yudin and K. Yu. Suponitsky, Mendeleev
Commun., 2006, 264; (b) M. Gobel and T. M. Klapotke, Adv. Funct.
Mater., 2009, 19, 347; (c) A. B. Sheremetev, N. S. Aleksandrova, N. V.
Palysaeva, M. I. Struchkova, V. A. Tartakovsky and K. Yu. Suponitsky,
Chem. Eur. J., 2013, 19, 12446.
This work was supported by the Russian Science Foundation
project no. 141301153). We are grateful to Dr. K.Yu. Suponitsky
for helpful discussions.
(
Online Supplementary Materials
Supplementary data associated with this article can be found
in the online version at doi:10.1016/j.mencom.2015.05.011.
1
References
1
2 O. A. Zagulyaeva and I. V. Oleinik, Chem. Heterocycl. Compd., 1995,
1
(a) Industrial and Laboratory Nitrations, ACS Symposium Series 22,
eds. L. F. Albright and C. Hanson, American Chemical Society, Washington,
DC, 1976; (b) K. Schofield, Aromatic Nitration, Cambridge University
Press, Cambridge, 1980; (c) G. A. Olah, R. Malhotra and S. C. Narang,
Nitration: Methods and Mechanisms, VCH, Weinheim, 1989; (d) Nitro
Compounds: Recent Advances in Synthesis and Chemistry, eds. H. Feuer
and A. T. Nielsen, VCH, Weinheim, 1990; (e) N. Ono, The Nitro Group in
Organic Synthesis, WileyVCH, Weinheim, 2001.
J. P. Agrawal and R. D. Hodgson, Organic Chemistry of Explosives,
Wiley, Hoboken, 2007.
V. L. Rusinov and O. N. Chupakhin, Nitroaziny (Nitroazines), Nauka,
Moscow, 1991 (in Russian).
3
1, 715 (Khim. Geterotsikl. Soedin., 1995, 816).
1
3 (a) I. V. Oleinik and O. A. Zagulyaeva, Mendeleev Commun., 1994, 50;
(
(
b) I. V. Oleinik and O. P. Shkurko, Russ. Chem. Bull., 1997, 46, 1351
Izv. Akad. Nauk, Ser. Khim., 1997, 1407).
1
1
4 (a) V. V. Perekalin, E. S. Lipina, V. M. Berestovitskaya and D. A. Efremov,
Nitroalkenes: Conjugated Nitro Compounds, Wiley, Chichester, 1994;
(
b) S. Rajappa, Tetrahedron, 1999, 55, 7065; (c) L. Simkova, F. Liska
and J. Ludvik, Curr. Org. Chem., 2011, 15, 2983.
2
3
4
5 (a) A. I. Titov, Zh. Obshch. Khim., 1948, 18, 534 (in Russian); (b) I. I.
Petrovich, Zh. Obshch. Khim., 1959, 29, 407 (in Russian); (c) S. S. Novikov
and L. I. Khmel’nitskii, Usp. Khim., 1957, 26, 459 (in Russian).
6 L. W. Deady and N. H. Quazi, Aust. J. Chem., 1992, 45, 2083.
7 A. J. Bellamy, N. V. Latypov and P. Goede, J. Chem. Res., M., 2003, 943.
8 (a) O. V. Lebedev, L. V. Epishina, T. S. Novikova, N. N. Makhova, T. I.
Godovikova, S. P. Golova, A. V. Shastin, E. A. Arnautova, T. S. Pivina
1
1
1
(a) V. A. Tartakovskii, O. P. Shitov, I. L. Yudin and V. A. Myasnikov,
USSR Patent SU 1703645, 1991; (b) I. L. Yudin, A. B. Sheremetev,
O. P. Shitov and V. A. Tartakovskii, Mendeleev Commun., 1995, 196;
(
c) B. B. Averkiev, M. Yu. Antipin, I. L. Yudin and A. B. Sheremetev,
and L. I. Khmel’nitskii, in International Annual Conference of ICT,
J. Mol. Struct., 2002, 606, 139; (d) I. L. Yudin, A. B. Sheremetev, B. B.
Averkiev and M. Yu. Antipin, J. Heterocycl. Chem., 2005, 42, 691.
(a) S. P. Philbin, R. W. Millar and R. G. Coombes, Propellants Explos.
Pyrotech., 2000, 25, 302; (b) A. J. Bellamy and P. Golding, Cent. Eur. J.
Energ. Mater., 2007, 4 (3), 33; (c) A. J. Bellamy and P. Golding, Cent.
Eur. J. Energ. Mater., 2008, 5 (2), 3.
th
29
(Energetic Materials), Karlsruhe, 1998, pp. 56.1–56.12; (b) A. V.
Shastin, T. I. Godovikova and B. L. Korsunskii, Russ. Chem. Rev., 2003,
2, 279 (Usp. Khim., 2003, 72, 311); (c) Q. Wu, W. Zhu and H. Xiao,
Struct. Chem., 2013, 24, 1725.
5
6
7
R. Murugan, E. F. V. Scriven, G. F. Hillstrom and P. K. Ghoshal, PCT Int.
Appl. WO 02090328, 2002.
Received: 24th February 2015; Com. 15/4572
–
195 –

 Yudin, Igor L.
Yudin, Igor L.
 Palysaeva, Nadezhda V.
Palysaeva, Nadezhda V.
 Averkiev, Boris B.
Averkiev, Boris B.
 Sheremetev, Aleksei B.
Sheremetev, Aleksei B.