Chung et al.
Decyl Glucoside Synthesized by Direct Glucosidation of D-Glucose Over Zeolite Catalysts and Its Estrogenicity
1
00
80
60
2
. EXPERIMENTAL DETAILS
FAU (zeolite Y, Si/Al = 3) was purchased from Zeobuilder
Co. The MOR (Mordenite, Si/Al = 10) zeolite was also
purchased from Tosoh Co. The cations of zeolites were
exchanged with H ion. We denote the H ion exchanged
zeolites following the zeolite code names, H-FAU and
H-MOR.
+
+
4
2
0
0
0
DGs were prepared from D-glucose (Sigma, 99%) with
H-FAU(3)
H-BEA(13)
H-MFI(25)
H-MOR(10)
1
-decanol (Aldrich, 99%) by direct glucosidation over the
zeolite catalysts. D-glucose (2.5 g) and 1-decanol (50 mL)
were introduced as the reactants. The reactants were put
into the reactor with the zeolite catalyst. The reaction was
0
1
2
3
4
5
ꢀ
carried out directly in the reactor with stirring at 130 C.
Process time (h)
The compositions of the products were measured by GC
equipped HP-1 capillary column and detected by an FID
analyzer. The conversion was determined as the percentage
of D-glucose consumed. The DG yields were determined
as the percentage of the amount produced with respect to
the sum of total products.
Surface tensions were measured using a ring and plate
method tensiometer (Kruss K100, Germany) and maxmum
bubble pressure tensiometer (Kruss BP2, Germany). Criti-
cal micelle concentration (CMC) was determined from the
value of surface tension. Contact angles (CA) were mea-
sured using pendant drop tensiometer (Kruss DSA 100,
Germany).
Figure 1. Variation of conversion of D-glucose with process time on
various zeolite catalysts at 130 C.
ꢀ
The catalytic activities of the zeolite catalysts are listed
in Table I. Conversion of D-glucose was obtained above
7
0% on the H-FAU zeolite catalyst. The H-FAU cata-
lyst showed a high yield of DGs. The selectivity of DG
exceeded 70% on the H-FAU zeolite. These results mean
that the weak acid sites of the zeolites can induce higher
catalytic activity in the reaction.
3
.2. Physical Properties of DGPs and Nonylphenol
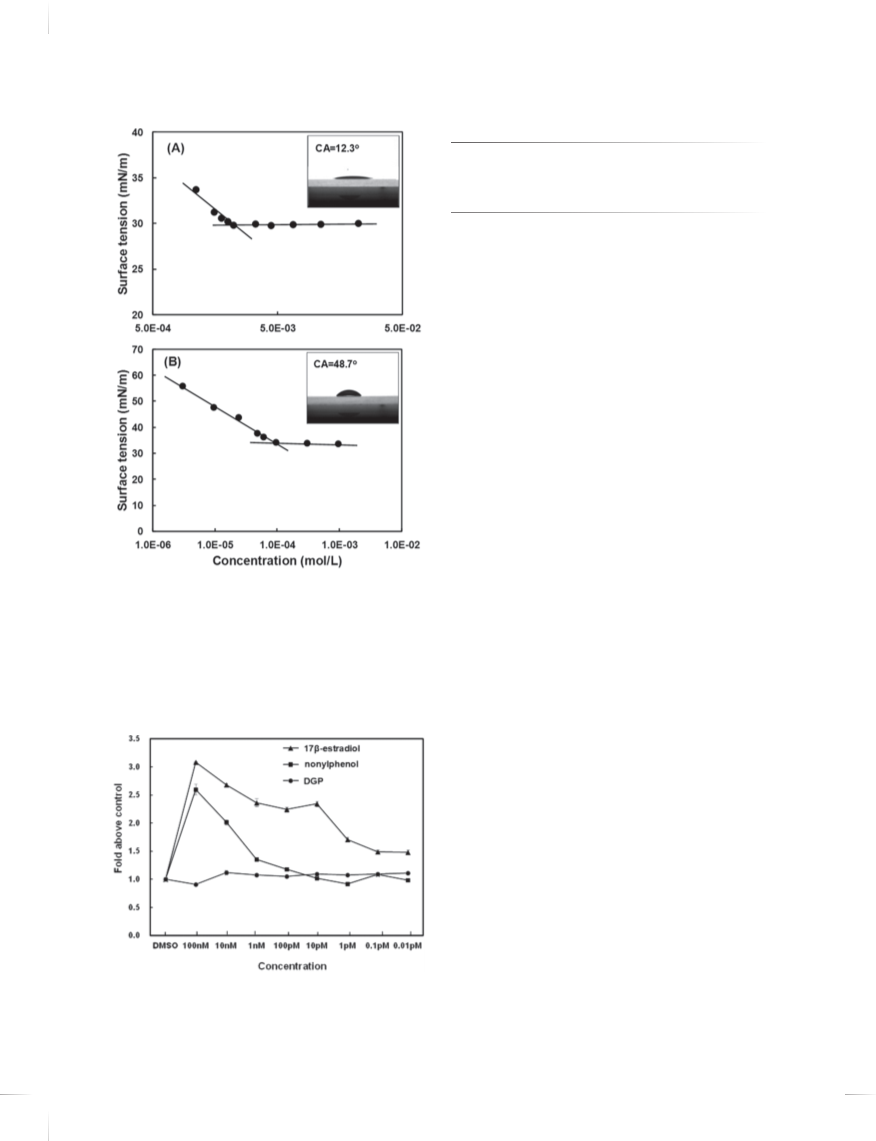
Figure 2 shows the surface tensions and contact angles
of DGP and nonylphenol. The CMCs of DGP were
MCF-7 (Michigan Cancer Foundation-7) cell line as a
IP: 127.0.0.1 On: Mon, 21 Jan 2019 02:07:08
human breast cancer cell line was induced in the esti-
Copyright: American Scientific Publishers
determined from the values of concentration at surface
mation of estrogenicity. The cell line was culti vD a et el idv e or ne d by Ingenta
tension unchanging despite of increasing of their concen-
Dulbecco’s modified Eagle’s medium (DMEN). Chemi-
cal stock solution dissolved in dimethylsulfoxide (DMSO)
was diluted again to 0.1% solvent concentration with
DMSO. The cell line was evaluated the MTT (2-(4,5-
dimethylthiazol-2-yl)-2,5-diphenyltetra zolium bromide,
Sigma) assay after cultivation in an incubator for 144 h.
After DMSO injection to the cultivation medium, the sam-
ple was estimated the absorbance at 540 nm by spec-
trophotometer. The control was consisted of cultivation
medium only. 17ꢁ-estradiol (99%, Sigma) and nonylphe-
nol (99%, Sigma) were induced as positive controls and
decyl-ꢂ-glucopyranoside (99%, Sigma-Aldrich) was used
as the object controls.
tration. From the results, the CMC of DGP was deter-
−3
mined as 2ꢄ2 × 10 mol/L and surface tension at CMC
was 30.1 mN/m. The CMC of nonylphenol was measured
−4
as 1ꢄ2 × 10 mol/L, the surface tension at CMC was
3
4.4 mN/m, respectively. The surface tension at CMC were
similar between two materials. The contact angle of DGP
was lower than those of nonylphenol and water (CA =
ꢀ
7
4.8 ꢃ. This indicates that the DGP is more hydrophilic
than nonylphenol and has a high wettability.
3
.3. Estrogenicities of DGP and Nonylphenol
To test the estrogenic effect of DGP, we performed the
E-assay on MCF-7 cells using 17ꢁ-estradiol and nonylphe-
nol as positive controls. Compared to DMSO-treated cells,
17ꢁ-estradiol and nonylphenol promoted MCF-7 cell pro-
liferation most potently by 3.08 and 2.60 fold, respectively,
3
. RESULTS AND DISCUSSION
.1. Catalytic Activities of the Zeolite Catalysts
In this glucosidation reaction, two DGs isomers i.e., the
ꢂ, ꢁꢃ-decyl glucopyranoside (DGP) and (ꢂ, ꢁꢃ-decyl glu-
3
(
Table I. Catalytic activities of the zeolite catalysts in the glucosidation.
cofuranoside (DGF) isomers, were produced mainly as an
anomeric mixture. A small amount of short-chain alco-
hols and their derivatives were also formed as byproducts
through the decomposition of 1-decanol.
Figure 1 presents the variation of D-glucose conversion
with process time over various zeolite catalysts. The con-
version was increased with increasing process time to 5 h.
Yield (%)
Selectivity (%)
Catalyst
Conversion (%)
DGP
DGF
DGP
DGF
H-FAU (3)
83ꢄ1
77ꢄ5
75ꢄ8
63ꢄ0
55ꢄ1
38ꢄ3
35ꢄ2
15ꢄ4
20ꢄ2
32ꢄ9
27ꢄ3
40ꢄ0
73ꢄ2
53ꢄ8
56ꢄ3
27ꢄ8
26ꢄ8
46ꢄ2
43ꢄ7
72ꢄ2
H-BEA (23)
H-MFI (25)
H-MOR (10)
J. Nanosci. Nanotechnol. 19, 1172–1175, 2019
1173

 Chung, Kyong-Hwan
Chung, Kyong-Hwan
 Kim, Hangun
Kim, Hangun
 Park, Young-Kwon
Park, Young-Kwon
 Kim, Byung-Hoon
Kim, Byung-Hoon
 Kim, Sun-Jae
Kim, Sun-Jae
 Jung, Sang-Chul
Jung, Sang-Chul