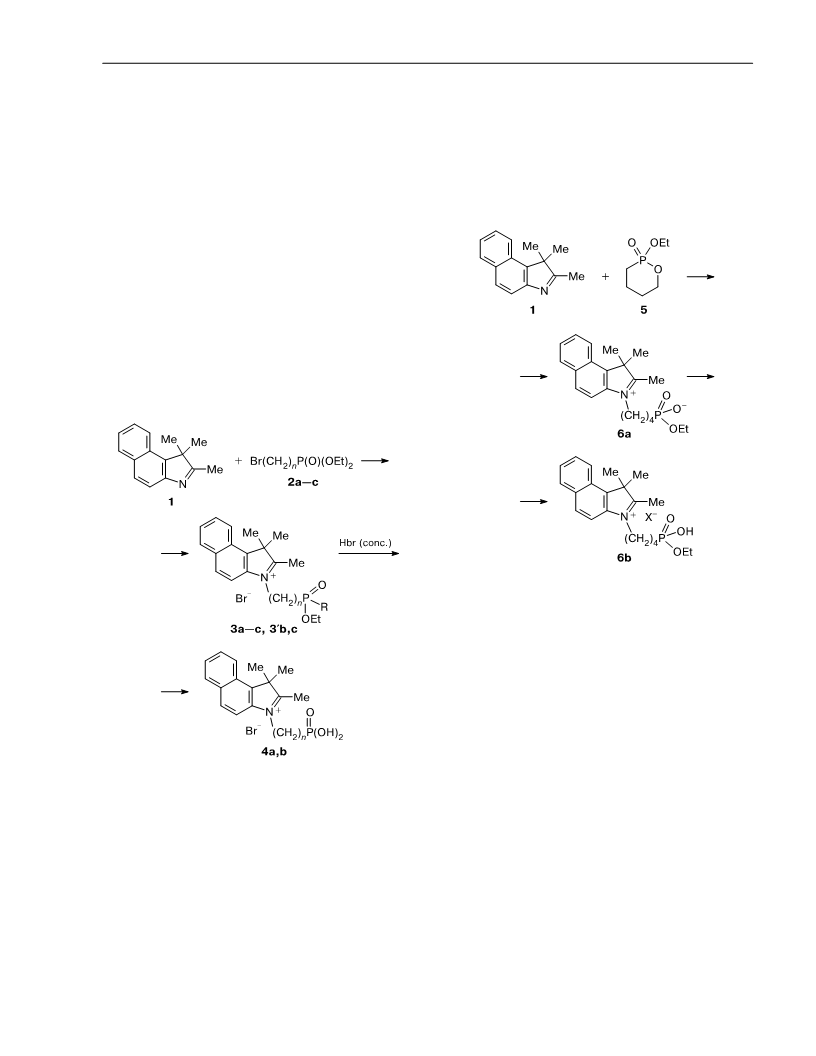
Synthesis of advanced fluorescent probes
Russ.Chem.Bull., Int.Ed., Vol. 65, No. 11, November, 2016 2727
=
CH); 7.2—8.15 (m, 16 H, =CH and arom.). 13C NMR
6.32—6.35, 6.57—6.63, 6.99—7.02, 7.26—7.30, 7.43—7.46 (all
m, 1 H each, HC=CH); 7.68—7.83 (m, 2 H, arom., =CH);
7.94—8.32 (m, 10 H, arom., =CH); 8.59 (t, 2 H, arom., J = 7.7 Hz);
3
(
DMSOꢀd ), δ: 16.5 (d, OCH CH , J = 5.1 Hz); 20.1, 21.3,
6
2
3
CP
1
3
(
1
both s, C(CH ) ); 26.0 (d, CH P, J = 144.1 Hz); 26.2 (d, CH P,
JCP = 136.1 Hz); 27.5, 27.6 (both s, CH ); 53.5 (s, N CH );
3
2
2
CP
2
+
2
2
9.01 (d, 3 H, POH, J = 5.7 Hz); 9.24 (d, 1 H, POH, J
=
2
2
HP
HP
2
13
5
3.9 (s, C(CH ) ); 56.06 (s, C(CH ) ); 61.7 (d, OCH , J
=
= 5.7 Hz). C NMR (CD OD), δ: 19.72 (s, CH ); 20.42
(s, CH ); 21.00 (s, C(CH ) ); 25.53 (d, CH P, J = 142.7 Hz);
2 3 2 2 CP
25.77 (d, CH P, J = 137.6 Hz); 26.24 (s, C(CH ) ); 27.16
2 CP 3 2
3
2
3 2
2
CP
3
2
1
=
5.1 Hz); 94.78, 103.5, 110.3, 122.1, 122.2, 122.3, 124.9, 127.7,
1
1
1
28.3, 130.0, 130.6, 131.7, 133.9 (all s, arom.); 154.3, 154.7,
55.5 (all s, =CH); 175.9 (s, NC=); 183.6 (s, N=C). P NMR
31
(s, CH ); 28.29(s, CH ); 31.61 (s, C(CH ) ); 31.76 (s, C(CH ) );
2 2 3 2 3 2
–
1
+
(
DMSOꢀd ), δ: 31.50, 31.63, 31.68. IR, ν/cm : 1065 (P—O—
56.63 (s, C(CH ) ); 58.07 (s, N CH ); 61.24 (s, C(CH ) ); 63.43
3 2 2 3 2
6
+
+
C H ), 1215 (P=O). Found: m/z 837.4132 [M – Br] .
(s, N CH ); 108.92 (s, arom.); 112.49, 118.82 (both s, =CH);
2
5
2
C H N O P . Calculated: M = 837.4154.
123.01, 124.15 (both s, arom.); 127.28 (s, =CH); 128.15, 128.30,
129.33, 129.65, 130.35, 131.07, 131.07 (all s, arom.); 135.80,
144.62, 145.48 (all s, =CH); 174.43 (s, NC=); 183.86 (s, N=C).
4
9
63
2
6 2
2
ꢀ((1E,3E,5E,7Z)ꢀ1,7ꢀBis[4ꢀ(diethoxyphosphoryl)butyl]ꢀ
3
,3ꢀdimethylꢀ3Hꢀbenzo[e]indolꢀ2(3H)ꢀylidene)heptaꢀ1,3,5ꢀtriꢀ
31
–1
2–
enyl)ꢀ3,3ꢀdimethylꢀ1Hꢀbenzo[e]indolium bromide (7b). The yield
P NMR (CD OD), δ: 28.66. IR, ν/cm : 1088 (PO3) . Found:
3
1
–
was 20%. H NMR (CD OD), δ: 1.23—1.32 (m, 12 H,
m/z 751.3086 [M – Br – 2 H] . C H N O P . Calculated:
3
43 51
2
6 2
OCH CH ); 1.75—1.90 (m, 8 H, CH ); 2.00 (s, 12 H C(CH ) );
M = 751.3071.
2
3
2
3 2
2
.02—2.25, 2.10—2.15 (both m, 4 H, CH P); 3.97—4.07 (m, 4 H,
2ꢀ((1E,3E,5E,7Z)ꢀ1,7ꢀBis{3,3ꢀdimethylꢀ3ꢀ[5ꢀ(dihydroxyꢀ
phosphoryl]pentyl}ꢀ3Hꢀbenzo[e]indolꢀ2(3H)ꢀylidene)heptaꢀ1,3,5ꢀ
trienyl)ꢀ3Hꢀbenzo[e]indolium bromide (7e). The yield was 12%.
2
+
OCH ); 4.21—4.30 (m, 4 H, CH N ); 6.35—6.40 (m, 1 H,
2
2
=
7
CH); 6.57—6.67 (m, 2 H, =CH); 7.47—7.49 (m, 2 H, =CH);
.59—7.65 (m, 4 H, arom.); 7.74—7.77 (m, 2 H, arom.); 7.82—7.84
m, 2 H, =CH); 7.95—8.10, 8.22—8.31 (both m, 6 H, arom.).
1
H NMR (CD OD), δ: 0.70—1.00, 1.02—1.48 (both m, 4 H,
3
(
7 H, CH , C(CH ) ); 1.50—1.78 (m, 10 H, C(CH ) , CH );
2
3 2
3 2
2
1
3
3
C NMR (CD OD), δ: 16.18 (d, OCH CH , J = 5.8 Hz);
1.82 (s, 3 H, C(CH ) ); 1.97—2.07 (m, 4 H, CH P); 4.63 (t, 4 H,
3 2 2
3
2
3
CP
3
+
3
1
6.39 (d, OCH CH , JCP = 5.1 Hz); 21.78, 21.82, (both s,
CH N , J = 7.7 Hz); 6.30—6.35, 6.56—6.67, 6.76—6.83,
2
3
2
1
C(CH ) ); 22.64 (s, CH ); 23.12 (d, CH P, J = 139.8 Hz);
2
7.11—7.20, 7.39—7.48 (all m, 1 H each, HC=CH); 7.55—7.66
(m, 1 H, =CH); 7.68—7.83 (m, 3 H, arom., =CH); 7.95—8.04
(m, 2 H, arom.); 8.07—8.34 (m, 6 H, arom.); 8.58 (t, 2 H, arom.,
3
2
2
2
CP
1
6.95 (d, CH P, J = 151.5 Hz); 29.67, 30.20 (both s, CH );
2 CP 2
0.23, 50.33 (both s, N CH ); 61.70, 62.68 (both d, OCH ,
2 2
+
5
2
3
2
JCP = 7.32 Hz); 62.25 (s, C(CH ) ); 108.02, 110.85, 111.22,
J = 7.7 Hz); 9.01 (d, 3 H, POH, J = 5.7 Hz); 9.24 (d, 1 H,
3 2
HP
2
13
1
1
27.83, 127.86, 128.16, 128.18, 129.90, 129.98, 130.70, 130.78,
35.86, 142.06, (all s, arom.); 147.77, 157.75, 158.74, 159.79 (all
POH, J = 5.7 Hz). C NMR (CD OD), δ: 18.81 (s, CH );
HP 3 2
19.48 (s, CH ); 22.51 (s, CH ); 24.02 (s, CH ); 24.09
2 2 2
31
1
s, =CH); 174.00 (s, NC=); 184.74 (s, N=C). P NMR (CD OD),
δ: 30.29. IR, ν/cm–1: 1055 (P—O—C H ), 1210 (P=O).
Found: m/z 865.4467 [M – Br] . C H N O P . Calculated:
(s, C(CH ) ); 27.74 (d, CH P, J = 141.4 Hz); 27.78 (d, CH P,
3 2 2 CP 2
3
1
JCP = 137.3 Hz); 28.82 (s, C(CH ) ); 29.89 (s, C(CH ) ); 30.67
2
5
3 2
3 2
+
+
(s, C(CH ) ); 30.98 (s, CH ); 31.35 (s, CH ); 42.69 (s, N CH );
3 2 2 2 2
43.40 (s, N CH ); 57.79 (s, C(CH ) ); 62.99 (s, C(CH ) );
51
67
2
6
2
+
M = 865.4469.
ꢀ(5ꢀ(Ethoxy(hydroxy)phosphoryl)pentyl)ꢀ2ꢀ((1E,3E,5E,7Z)ꢀ
ꢀ(3ꢀ(5ꢀ(diethoxyphosphoryl)pentyl)ꢀ1,1ꢀdimethylꢀ1Hꢀbenzoꢀ
e]indolꢀ2(3H)ꢀylidene)heptaꢀ1,3,5ꢀtrienyl)ꢀ1,1ꢀdimethylꢀ1Hꢀ
2 3 2 3 2
3
109.20 (s, arom.); 114.60, 122.81 (both s, =CH); 126.03 (s, arom.);
127.70 (s, =CH); 127.86, 129.63, 130.35, 130.02, 132.59, 132.69,
138.08, 139.65 (all s, arom.); 135.80, 144.73,146.15, 153.82 (all s,
7
[
1
31
benzo[e]indolium bromide (7´c). The yield was 25%. H NMR
=CH); 168.53 (s, NC=); 189.50 (s, N=C). P NMR (CD OD),
3
–
1
2–
(
(
DMSOꢀd ), δ: 0.81—1.0 (br.m, 3 H, OCH CH ); 1.0—2.00
br.m, 26 H); 3.75—4.10 (br.m, 6 H, OCH ); 4.15—4.40 (br.m,
δ: 28.41. IR, ν/cm : 1088 (PO3) . Found: m/z 779.3419. [M –
– Br – 2 H] . C H N O P . Calculated: M = 779.3384.
6
2
3
–
2
45 53
2
6 2
+
4
1
H, CH N ); 6.44—6.41 (br.m, 1 H, =CH); 7.40—8.32 (br.m,
8 H, =CH and arom.). C NMR (DMSOꢀd ), δ: 16.33
2
1
3
The work was carried out using equipment purchased
at the expense of the Moscow State University Developꢀ
ment Program.
6
3
3
(
d, OCH CH , J = 5.8 Hz); 16.50 (d, OCH CH , J
=
CP
2
3
CP
2
3
3
=
2
5.8 Hz); 16.57 (d, OCH CH , J = 6.6 Hz); 22.93 (s, CH );
2 3 CP 2
2.96 (s, CH ); 24.78 (d, CH P, JCP = 144.9 Hz); 26.37
1
2
2
1
(
d, CH P, J = 139.0 Hz); 26.98 (s, CH ); 27.15 (s, CH ); 26.70,
This work was financially supported by the Russian
Science Foundation (Program "Carrying out Fundamenꢀ
tal Scientific Studies and Pioneering Scientific Studies by
Scientific Groups", Project No. 14ꢀ13ꢀ00698).
2
CP
2
2
2
6.78 (both s, C(CH ) ); 27.40, 27.51 (both s, CH ); 44.71, 45.15
3 2 2
+
(
(
(
both s, N CH ); 51.58, 52.86 (both s, C(CH ) ); 61.04, 61.52,
both s, OCH ); 63.70 (d, OCH , JCP = 7.32 Hz); 93.07
s, =CH); 101.96 (s, =CH); 112.03, 122.66, 124.77, 127.63, 128.16,
30.21, 130.32, 131.00, 131.23, 131.65, 133.49, 136.37 (all s,
arom.); 142.83, 144.97, 146.62, 155.37, 156.36 (all s, =CH);
2
3
2
2
2
2
1
Reference
3
1
1
3
71.22 (s, NC=); 183.54 (s, N=C). P NMR (DMSOꢀd ), δ:
6
1.87, 22.69. IR, ν/cm–1: 1078 (P—O—C H ), 1194 (P=O).
1. H. A. Shindy, MiniꢀRev. Org. Chem., 2012, 9, 352.
2. A. Mishra, R. K. Behera, B. K Mishra, G. B. Behera, Chem.
Rev., 2000, 100, 1973.
3. M. Panigrahi, S. Dash, S. Patel, B. K. Mishra, Tetrahedron,
2012, 68, 781.
2
5
+
Found: m/z 865.4453 [M – Br] . C H N O P . Calculated:
M = 865.4469.
ꢀ((1E,3E,5E,7Z)ꢀ7ꢀ[3,3ꢀDimethylꢀ1ꢀ(4ꢀ(dihydroxyphosꢀ
5
1
67
2
6 2
2
phoryl)butyl]ꢀ1Hꢀbenzo[e]indolꢀ2(3H)ꢀylidene)heptaꢀ1,3,5ꢀtriꢀ
enyl)ꢀ3,3ꢀdimethylꢀ1ꢀ(4ꢀ(dihydroxyphosphoryl)butyl)ꢀ3Hꢀbenzoꢀ
4. J. Yin, Y. Kwon, D. Kim, D. Lee, G. Kim, Y. Hu, J.ꢀH. Ryu,
J. Yoon, J. Am. Chem. Soc., 2014, 136, 5351.
5. A. Gorka, R. Nani, M. Schnermann, Org. Biomol. Chem.,
2015, 13, 7584.
1
[
e]indolium bromide (7d). The yield was 13%. H NMR (CD OD),
3
δ: 1.68—1.86 (m, 16 H, C(CH ) , CH ); 1.90—2.01 (m, 4 H,
3
2
2
+
CH ); 2.07—2.14 (m, 4 H, CH P); 4.62—4.68 (m, 4 H, CH N );
2
2
2

 Podrugina
Podrugina
 Temnov
Temnov
 Doroshenko
Doroshenko
 Kuzmin
Kuzmin
 Nekipelova
Nekipelova
 Proskurnina
Proskurnina
 Zefirov
Zefirov