Angewandte
Communications
Chemie
pyrimidine substituent to explain the formation of both the
ketone and tertiary alcohol, since derivatives lacking the
[13]
amino group failed to provide the products.
Based on the similarities of the rearrangement and
fragmentation products in the reports by Oka et al. and our
results reported herein, we propose that all of the products
can be explained by the same radical mechanism. CÀN bond
homolysis of the Breslow intermediate initially forms a gemi-
nate radical pair (Scheme 6). The rearrangement product
forms by radical recombination, while the ketone and
pyrimidine products result from disproportionation, that is,
b-hydrogen atom abstraction of the hydroxyl hydrogen by the
[14–16]
carbon radical.
Scheme 4. Ambient-temperature [1,3] rearrangements of N-di(4-fluoro-
[
28]
phenyl)methyl Breslow intermediates.
1
,2,4-triazole 3 f provided rearrangement product 4 f in low
yield as the only isolable product, although interestingly as
a single regioisomer resulting from migration of the N4 sub-
stituent. No significant amount of benzoin was detected in the
Scheme 6. Proposed radical reaction mechanism.
1
[17]
crude reaction mixtures based on TLC and H NMR analysis.
DFT calculations (B3LYP/6-31G*) of the enthalpy of
In both cases the rearrangements occurred at ambient
temperature.
the homolysis reactions of Breslow intermediates 2b–e to
form the benzyl, cinnamyl, and diarylmethyl radicals and
À1
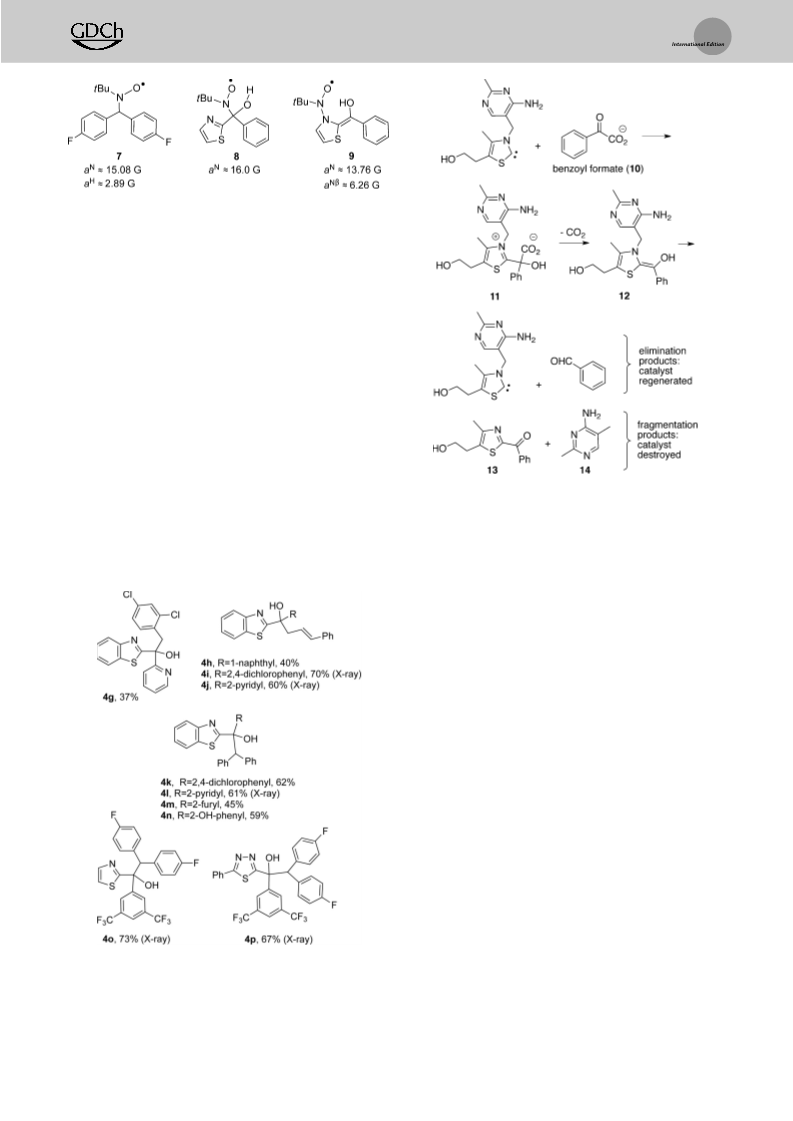
In 1970, Oka et al. reported a similar [1,3] rearrangement
in which thiamine and related compounds underwent reaction
with substituted benzaldehydes in methanol at reflux to
provide rearranged tertiary alcohol products, albeit in very
low yield (Scheme 5; 3-12%, highest yielding example
carbinol radicals 6 were +20.9, 13.0, 8.8, and 6.6 kcalmol ,
[18]
respectively (Scheme 7). The extraordinarily low enthal-
pies of reaction are likely due to the highly delocalized nature
of carbinol radical 6 and its recovery of aromaticity upon
homolysis. The trend in enthalpies for the benzothiazole-
based intermediates correlates with the increasing radical-
stabilizing ability of the R group as the N substituent.
[12]
shown).
The rearrangement products were accompanied
by larger amounts of ketone and pyrimidine products result-
ing from cleavage of the benzylic CÀN bond, as well as
We sought to obtain direct chemical evidence of radical
intermediates, albeit without success. Trapping experiments
with 2,2,6,6-tetramethylpiperidine N-oxide (TEMPO) gave
a “considerable” amount of benzoin. Thus, thiamine served
for a limited period during the reaction as a benzoin catalyst
but ultimately decomposed into rearrangement and fragmen-
tation products. Oka et al. proposed a rather elaborate polar
mechanism that employed the ortho amino group of the
[19]
no isolable addition products.
The absence of isolable
addition products could be due to more rapid recombination
[20,21]
of radicals than escape from the solvent cage.
Deuterium
labeling experiments were problematic because of the
relative unreactivity of the intermediate radicals relative to
[22]
a labeling source. However, EPR experiments gave strong
evidence for a radical pathway. The EPR spectrum of the
reaction of 3e with benzaldehyde in the presence of the
radical trap 2-methyl-2-nitrosopropane (MNP) was consistent
Scheme 5. [1,3] Rearrangement and fragmentation products of
thiamine derivatives reported by Oka et al.
Scheme 7. Calculated enthalpies of homolysis for intermediates
2b–e (DFT calculations at the B3LYP/6-31G* level of theory).
[
12]
3
56
ꢀ 2016 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Angew. Chem. Int. Ed. 2016, 55, 355 –358

 Alwarsh, Sefat
Alwarsh, Sefat
 Xu, Yi
Xu, Yi
 Qian, Steven Y.
Qian, Steven Y.
 McIntosh, Matthias C.
McIntosh, Matthias C.