
Chemistry - A European Journal p. 6259 - 6263 (2014)
Update date:2022-08-17
Topics:
 Durie, Alastair J.
Durie, Alastair J.
 Fujiwara, Tomoya
Fujiwara, Tomoya
 Cormanich, Rodrigo
Cormanich, Rodrigo
 Buehl, Michael
Buehl, Michael
 Slawin, Alexandra M. Z.
Slawin, Alexandra M. Z.
 O'Hagan, David
O'Hagan, David
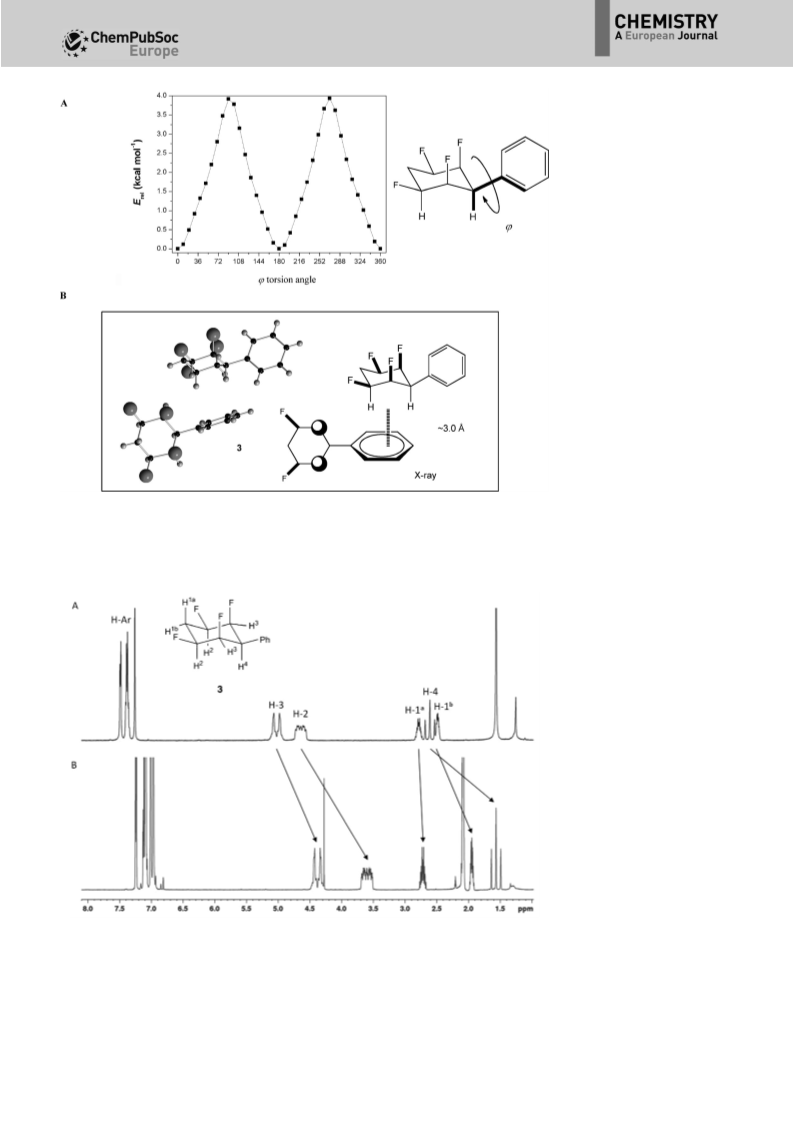
A stereocontrolled synthesis of all-cis-1,2,4,5- tetrafluoro-3- phenylcyclohexane is developed as the first functionalised example of this polar cyclohexane motif. The dipolar nature of the ring, arising due to two 1,3-diaxial C-F bonds, is revealed in the solid-state (X-ray) structure. The orthogonal conformation of the aryl and cyclohexyl rings in all-cis-1,2,4,5- tetrafluoro-3-phenylcyclohexane, and in an ortho-nitro derivative, result in intramolecular 1hJHF and 2hJCFNMR couplings relayed through hydrogen bonding. The aryl group of all-cis-1,2,4,5-tetrafluoro-3-phenylcyclohexane is elaborated in different ways to demonstrate the versatility of this compound for delivering the motif to a range of molecular building blocks. A stereocontrolled synthesis of all-cis-1,2,4,5-tetrafluoro-3-phenylcyclohexane is developed as the first functionalised example of this polar cyclohexane motif. The dipolar nature of the cyclohexane ring is explored and the aryl ring is elaborated in different ways to demonstrate its versatility as a molecular building block (see figure).
View More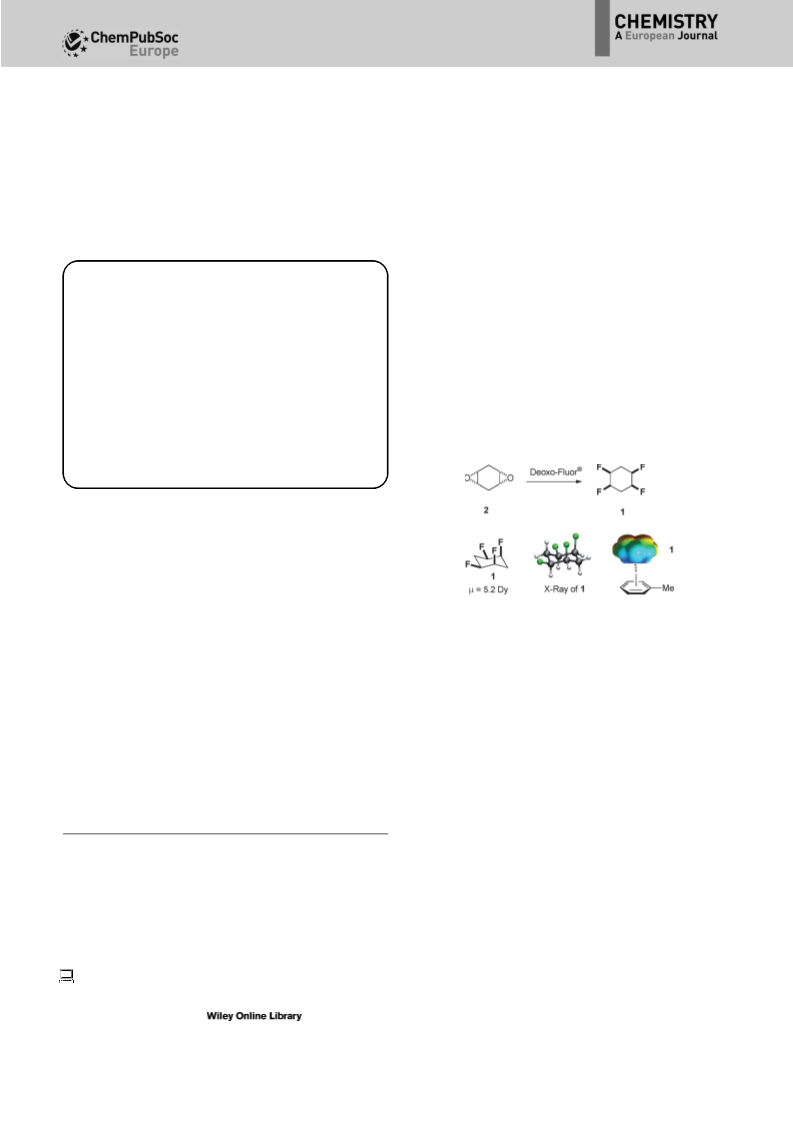




shijiazhuang shuanglian chemical industry co.,ltd
Contact:0311-82190302
Address:Luquan Intersection , Shijiazhuang--Taiyuan Expressway,Shijiazhuang City
Contact:+33-5-34012600
Address:28 ZA des Pignès
Shanghai PotentPharm Science and Technology Co.,Ltd
Contact:86-021-51969655
Address:Unit B, Building 18, No.300, Chuantu Rd,Pudong District, Shanghai 201202, China
Yibin Hope Pharmaceutical Co., Ltd.
Contact:+86-831-5109851 86-831-5109836
Address:Luolong Industrial Zone,Nanxi, Yibin City, Sichuan, China. | Email: wishmarketing@gmail.com | Website: http://www.hopepharma.cc/
Shanghai Goyic Pharmaceutical&Chemical Co,. Ltd
Contact:+86-021-60275964
Address:No. 528 ruiqing road
Doi:10.1055/s-0035-1561433
(2016)Doi:10.1002/cphc.202000267
(2020)Doi:10.1111/febs.12791
(2014)Doi:10.1039/b707292a
(2007)Doi:10.1007/s11164-013-1059-6
(2014)Doi:10.1016/j.tetlet.2009.08.028
(2009)