
Journal of Pharmaceutical Sciences p. 258 - 271 (1983)
Update date:2022-08-11
Topics:
 Garrett
Garrett
 Seyda
Seyda
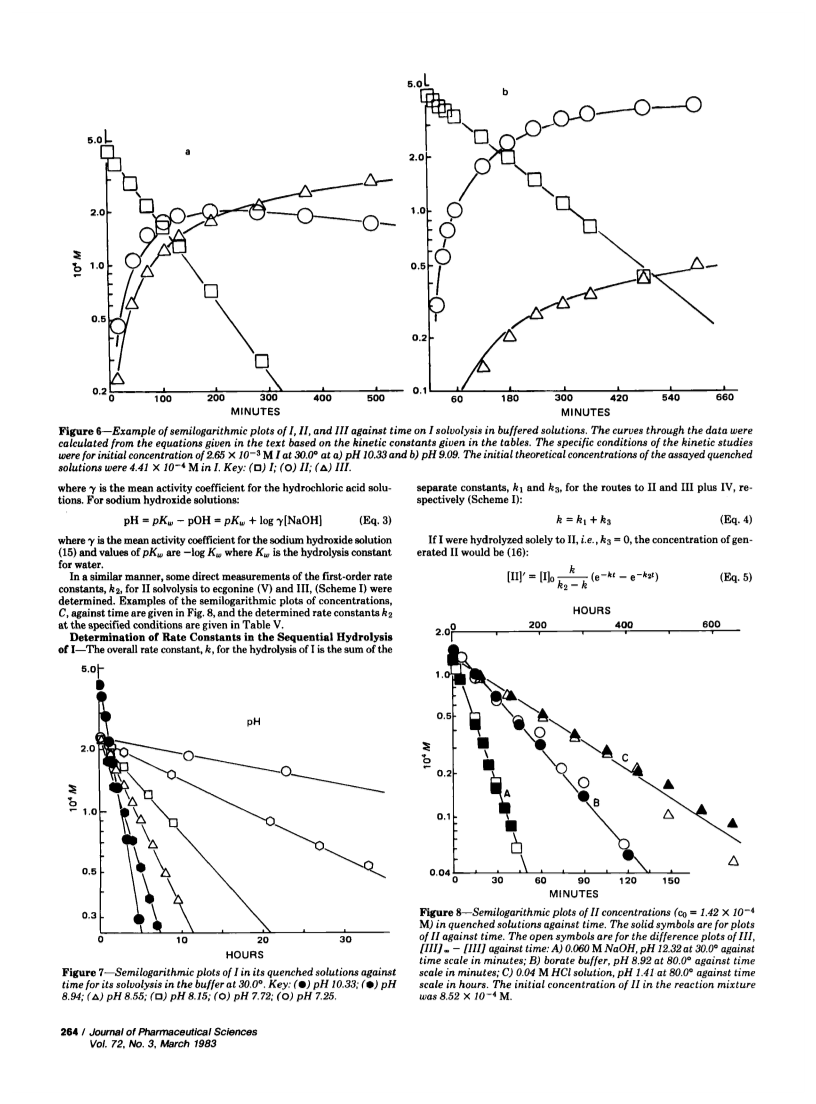
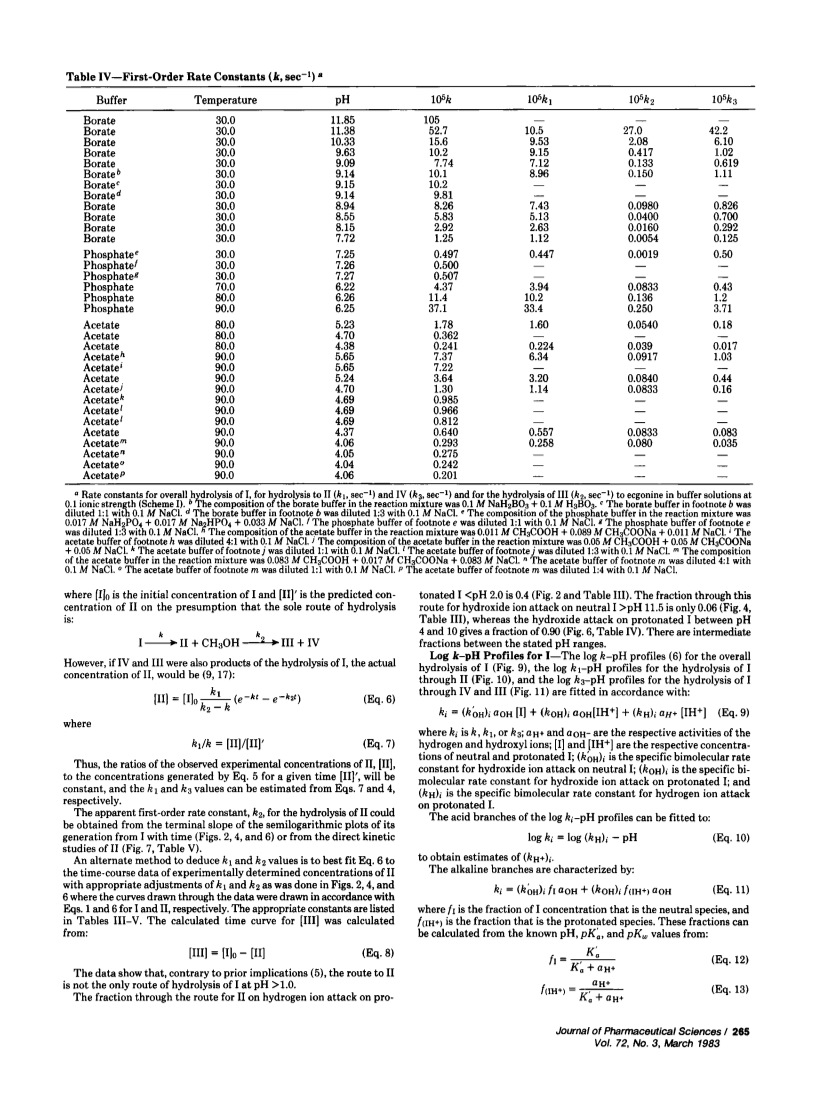
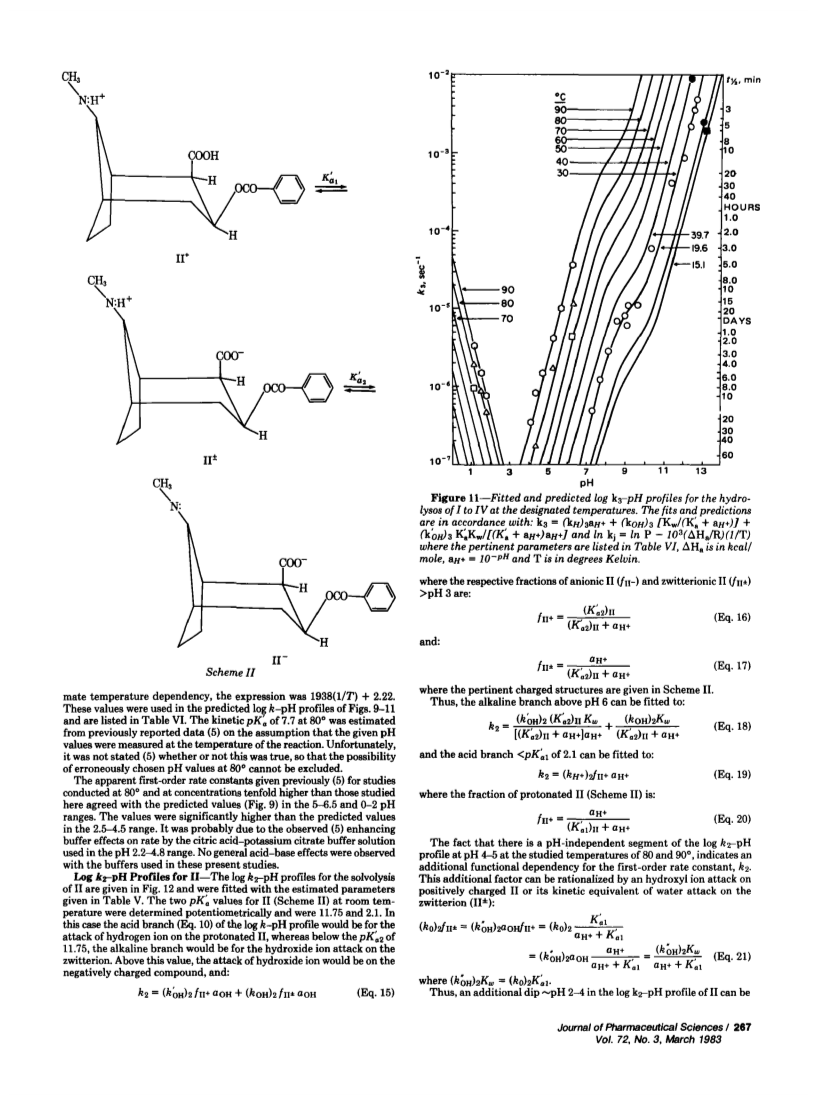
Specific, sensitive, reverse-phase high-performance liquid chromatographic (HPLC) assays of cocaine (I) and its hydrolysis products, benzoylecgonine (II) and benzoic acid (III), have been devised with analytical sensitivities as low as 15 ng/ml of plasma for I using spectrophotometric detection at 232 nm. Cocaine can be separated from its hydrolysis products by extraction at pH 7.5 with haloalkanes. Benzoylecgonine and benzoic acid can be extracted at pH 3.0 with 1-butanol. The evaporated residues were reconstituted in acetonitrile-water for HPLC assay. The assay was used to determine the stabilities of I and II in aqueous solutions, to establish log k-pH profiles at various temperatures, and to evaluate Arrhenius' parameters. Hydrolyses were by specific acid-base catalysis. Cocaine showed hydrogen and hydroxyl ion attack on protonated I with 40 and 90% proceeding through the benzoylecgonine route, respectively, as well as hydroxyl ion attack on neutral cocaine, with only 6% proceeding through the benzoylecgonine route. Cocaine is relatively unstable in the neutral pH range with a half-life of 5 hr in buffer at pH 7.25 and 40°. Similar half-lives were observed in fresh dog plasma at 300 and 30 μg/ml, although one study at 0.5 μg/ml indicated a doubling of the rate.
View More










Doi:10.1002/adsc.201500646
(2015)Doi:10.1016/j.electacta.2014.08.046
(2014)Doi:10.1080/00397911.2019.1665183
(2019)Doi:10.1080/10286020.2013.805207
(2013)Doi:10.1021/jacs.5b01082
(2015)Doi:10.1016/j.jorganchem.2017.04.034
(2017)