1
Mp 75–76 ЊC (no decomposition). H NMR (CD3OD, 298
the Tables and Figures, the latter showing 20 (295 K) or 50%
(153 K) probability amplitude displacement ellipsoids for the
non-hydrogen atoms, hydrogen atoms having arbitrary radii of
0.1 Å.CIF depositions have been made with the Cambridge
Crystallographic Data Base.†
K): δ 3.90 (s, 8H, C2H4), 8.90 (br s, 2H, NH). 13C NMR
(CD3OD, 298 K): δ 47.1 (s, C2H4), 174.6 (s, SCN2). MS ES: m/z
(%) 203.2 ([M Ϫ I5]ϩ, 11), 171.3 ([M Ϫ (S ϩ I5)]ϩ, 10), 102.1
([imidazolidine-2-thione ]ϩ, 100). IR (Nujol) ν/cmϪ1: 3343s br,
1629w br, 1511s sh, 1378w sh, 1310m sh, 1275m sh, 1191m sh,
1038w, 909m sh.
Crystal/refinement data
5
2 = C6H10N4S, M = 170.2. Monoclinic, space group P21/c (C 2h
,
1-[4,5-dihydro-1H-imidazolidin-2-yl]imidazolidine-2-thione, [2],
and its hydrobromide salt, [2ؒHBr]
No. 14), a = 7.9079(8), b = 12.480(2), c = 7.7913(7) Å,
β = 92.89(1)Њ. V = 768.0 Å3. Dc (Z = 4) = 1.472 g cmϪ3. µMo
=
3.6 cmϪ1; specimen: cuboidal section, ca. 0.12 (no absorption
correction). 2θmax = 50Њ, N = 1249, N0 = 762 (I > 2σ(I )); R =
0.049, Rw = 0.052.
Following the procedure of Poos et al. utilising mercuric
oxide,3,7 a suspension of imidazolidine-2-thione (4.0 g, 39.2
mmol) and mercuric oxide (8.00 g, 36.9 mmol) was heated in
xylene (40 cm3) under reflux (4 h) and filtered while hot. The
resultant dark insolubles were further extracted with chloro-
form and combined with the xylene residues. Removal of the
solvent in vacuo yielded a yellow solid (ca. 0.75 g, 25% yield by
imidazolidine-2-thione), which was recrystallised from ethanol
as rather fine tan needles. Recrystallisation of these needles
from an equimolar amount of aqueous hydrobromic acid
yielded colourless prisms of the monohydrobromide salt,
2ؒHBr.
2ؒHBr = C6H11BrN4S, M = 251.3. Orthorhombic, space
16
2h
group Pnma (D , No. 62), a = 15.295(3), b = 6.895(7), c =
9.063(2) Å, V = 959.7 Å3. Dc (Z = 4) = 1.745 g cmϪ3. µMo = 45
cmϪ1; specimen: 0.59 × 0.58 × 0.37 mm; ‘T’min,max = 0.16, 0.36.
2θmax = 65Њ, Nt = 1521, N0 (I > 3σ(I )) = 1039; R = 0.033, Rw =
0.049.
3 = (C6H9N2S)I3, M = 521.9. Orthorhombic, space group
16
2h
Pbca (D , No. 61), a = 13.420(1), b = 12.950(1), c = 14.439(1) Å,
V = 2509 Å3. Dc (Z = 8) = 2.477 g cmϪ3. µMo = 76 cmϪ1
;
specimen: 0.18 × 0.15 × 0.13 mm; ‘T’min,max = 0.54. 2θmax = 65Њ,
N = 34480, N0 = 17029 (Rint = 0.041), N0 = 12932; R = 0.037, Rw
= 0.044.
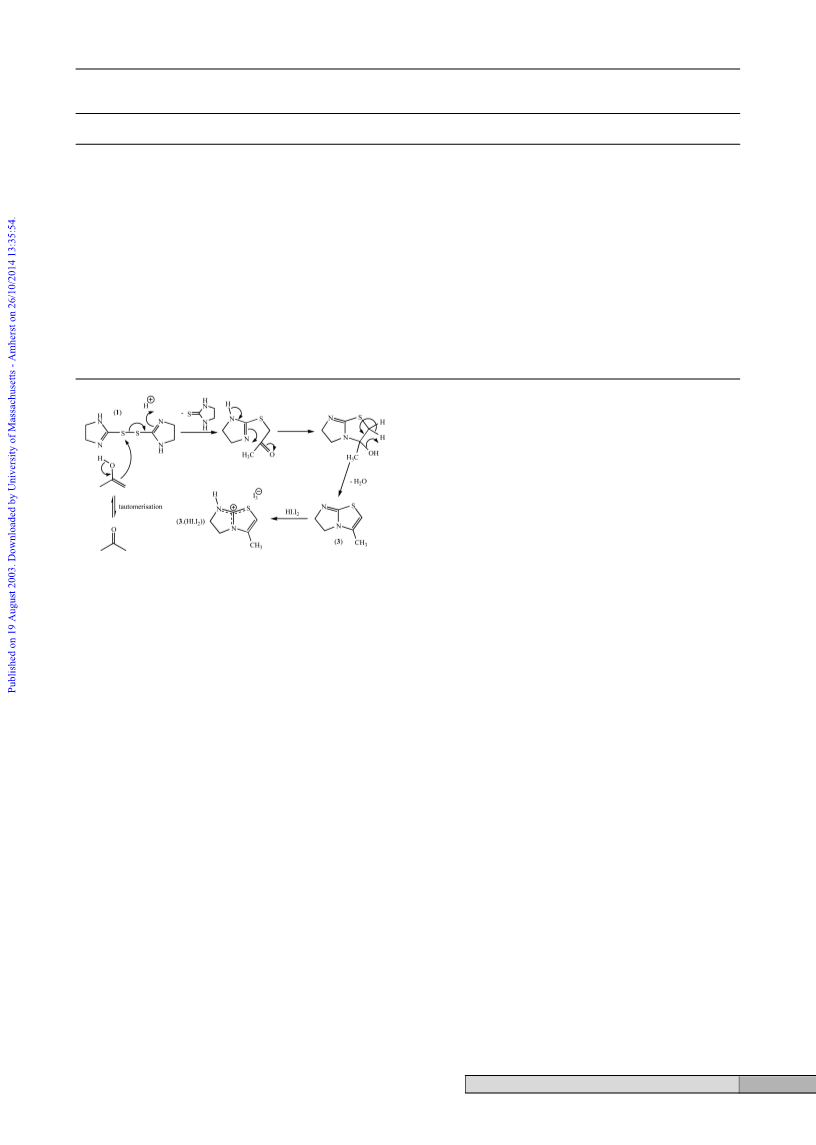
3-Methyl-5,6-dihydro-7H-imidazolidin[2,1-b]thiazolium
triiodide, [3ؒ(HIؒI2)]
Deep magenta 2,2Ј-bis(4,5-dihydro-1H-imidazolidine)disulfide
hydroperiodide, 1ؒ(HIؒ2I2), (0.3 g, 0.36 mmol) was dissolved in
acetone (ca. 10 cm3) rendering a deep orange solution that
yielded dark plate-like crystals upon standing at room temper-
ature. These were collected by filtration and washed with cold
Acknowledgements
The partial support of this work by the Postgraduate Education
and Research Program in Chemistry (PERCH), Thailand, is
gratefully acknowledged.
1
hexane (3 × 5 cm3) (0.09 g, 48%). Mp 70–72 ЊC. H NMR
(CD3OD, 298 K): δ 2.30 (d, 3H, 3-CH3, 4JHH 1.34 Hz), 4.44 (m,
† CCDC reference numbers 187057, 187058 and 211802. See http://
www.rsc.org/suppdata/ob/b3/b306647a/ for crystallographic data in .cif
or other electronic format.
4
4H, C H ), 6.47 (d, 1H, SCH᎐C, JHH 1.35 Hz). 13C NMR
᎐
2
4
(CD3OD, 298 K): δ 13.0 (s, 3-CH3), 44.9 (s, CH2), 49.9 (s, CH2),
103.8 (s, SC᎐C), 133.7 (s, 3-C), 172.3 (s, ϩCN S). MS APCI: m/z
᎐
2
(%) 141.7 ([M Ϫ I3]ϩ, 100). IR (Nujol) ν/cmϪ1: 3493w br, 3400m
sh, 1591w sh, 1558s sh, 1294m, 1200w sh, 1166w sh. Calc. for
C6H9N2S1I3: C; 13.81, H; 1.74, N; 5.37, S; 6.14. Found (best): C;
14.10, H; 1.84, N; 6.61, S; 7.47%. [Microanalyses for 3ؒ(HIؒI2)
were routinely high in both N and S content, thereby suggesting
that recrystallised samples were consistently contaminated by
imidazolidine-2-thione.2I2 even after repeated recrystallisation
from acetone. This tentatively provides further evidence for the
reaction mechanism proposed in Fig. 6, which eliminates imid-
azolidine-2-thione].
References
1 M. Jaffé and B. Kühn, Chem. Ber., 1894, 27, 1663.
2 H. E. Heldal and J. Sletten, Acta Chem. Scand., 1996, 50, 596.
3 G. I. Poos, J. Kleis and C. K. Cain, J. Org. Chem., 1959, 24, 645.
4 R. Hull and T. P. Seden, Synth. Commun., 1980, 10, 489.
5 B. T. Johnson and C. O. Edens, J. Am. Chem. Soc., 1942, 64, 2706.
6 F. H. Herbstein and W. Schwotzer, J. Am. Chem. Soc., 1984, 106,
2367.
7 H. Z. Lecher and K. Gubernator, J. Am. Chem. Soc., 1953, 75, 1087.
8 A. Saczewski, A. Bulakowska and M. Gdaniec, J. Heterocycl.
Chem., 2002, 39, 911.
9 D. C. Kim, K. H. Yoo, K. J. Shin, S. W. Park and D. J. Kim,
J. Heterocycl. Chem., 1997, 34, 57.
Structure determinations
10 A. M. Demchenko, V. A. Chumakov, A. N. Krasovsky, E. B.
Rusanov, A. N. Chernega, V. V. Pirozhenko and M. O. Lozinsky, Zh.
Obshch. Khim., 1997, 67, 1886.
11 Unit cell data of two products both resulting from EtOH and
MeOH recrystallisation of 1ؒ(HIؒ2I2): (all T = 123 K) a; 11.0604(13)
Å, b; 11.1829(12) Å, c; 9.5618(12) Å, α; 90Њ, β; 110.03(1)Њ, γ; 90Њ and
a; 10.2839(12) Å, b; 9.1691(7) Å, c; 6.7637(7) Å, α; 105.03(1)Њ, β;
81.09(2)Њ, γ; 96.42(1)Њ. Data from reference 4 (T = 298 K) for
imidazolidine-2-thione, I2 co-crystallate of 2ؒ(HIؒI2), a; 11.102(7) Å,
b; 11.193(7) Å, c; 9.551(6) Å, α; 90Њ, β; 110.00(10)Њ, γ; 90Њ. Data from
For 2 and 2ؒHBr unique single-counter four-circle diffract-
ometer data sets were measured at ca 295 K, yielding N unique
reflections, N0 as defined below being considered ‘observed’ and
used in the full matrix least squares refinement, 2ؒHBr data
being subject to analytical absorption correction. Anisotropic
displacement parameter forms were refined for the non-hydro-
gen atoms, (x, y, z, Uiso)H being refined (3 excepted). For
3ؒ(HIؒI2), a full sphere of CCD area-detector diffractometer
data was measured (Bruker AXS instrument, ω-scans; mono-
chromatic Mo Kα radiation, λ = 0.71073 Å (all structures);
T ca. 153 K), Nt(otal) reflections merging to N unique after
‘empirical’/multiscan absorption correction (proprietary soft-
ware). Conventional residuals on |F|, R, Rw (weights: (σ2(F) ϩ
0.0004 F 2)Ϫ1) are cited at convergence; neutral atom complex
scattering factors were employed within the context of the Xtal
3.7 program system.14 Pertinent results are given below and in
reference
4 (T = 298 K) for bis(diiodine) co-crystallate of
imidazolidine-2-thione, a; 10.276(7) Å, b; 9.170(6) Å, c; 6.792(5) Å,
α; 105.10(10)Њ, β; 81.00(10)Њ, γ; 96.40(10)Њ.
12 R. C. Moser, L. J. Powers and Z. S. Ariyan, Diamond Shamrock
Corp., USA, Cam. Pat. Appl. No. 269, 924, 1982.
13 D. E. Kuhlia, Pfizer Inc., USA, U.S. Pat. Appl. No. 73-399053
19730920, 1975.
14 S. R. Hall, D. J. du Boulay and R. Olthof-Hazekamp (eds.), ‘The
Xtal 3.7 System’, University of Western Australia, 2001.
O r g . B i o m o l . C h e m . , 2 0 0 3 , 1, 3 2 1 7 – 3 2 2 2
3222

 Thanyasirikul, Yupa
Thanyasirikul, Yupa
 Pakawatchai, Chaveng
Pakawatchai, Chaveng
 Cole, Marcus L
Cole, Marcus L
 Junk, Peter C
Junk, Peter C
 Skelton, Brian W
Skelton, Brian W
 White, Allan H
White, Allan H