
Synthetic Communications p. 1 - 7 (2020)
Update date:2022-08-30
Topics:
 Nibin Joy, Muthipeedika
Nibin Joy, Muthipeedika
 Beliaev, Nikolai
Beliaev, Nikolai
 Beryozkina, Tetyana V.
Beryozkina, Tetyana V.
 Bakulev, Vasiliy A.
Bakulev, Vasiliy A.
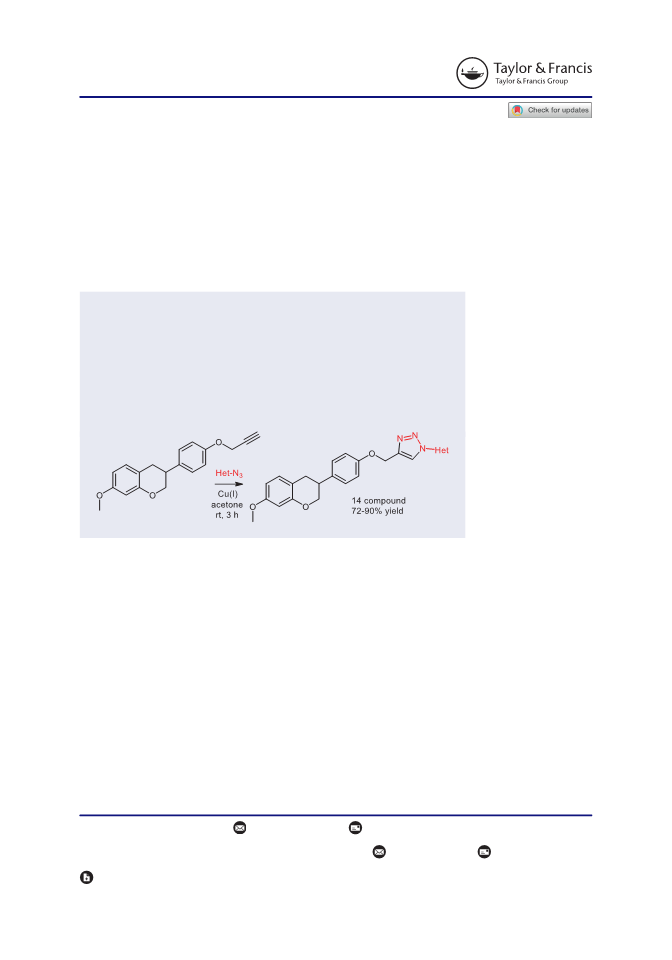
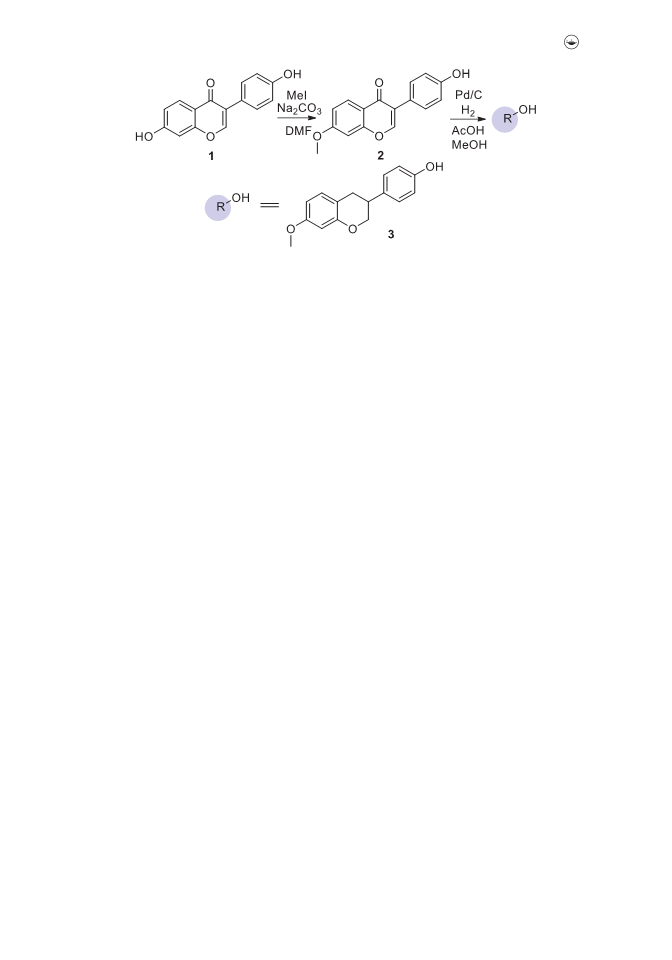
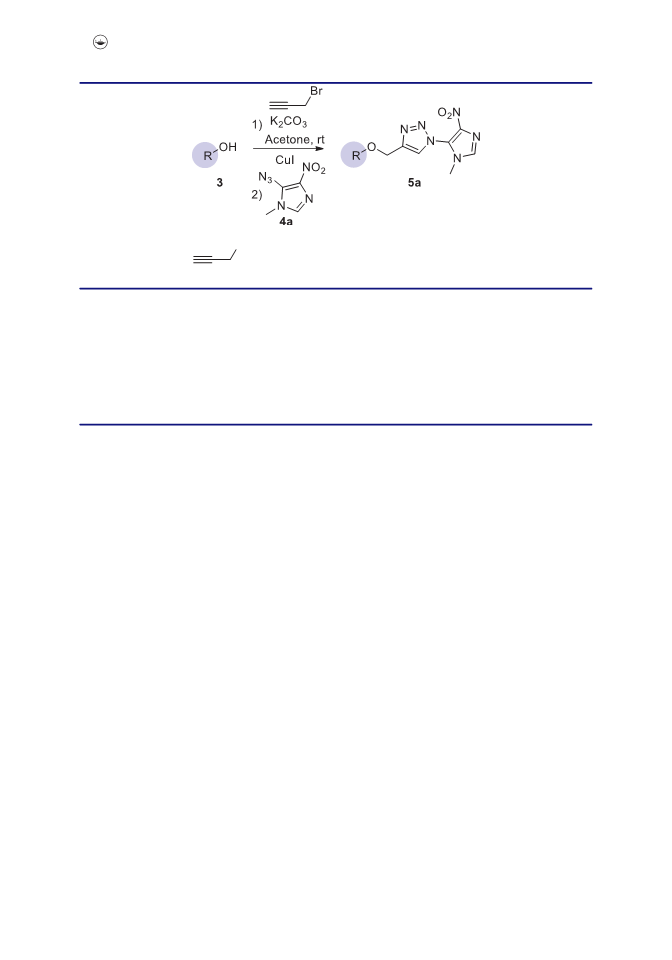
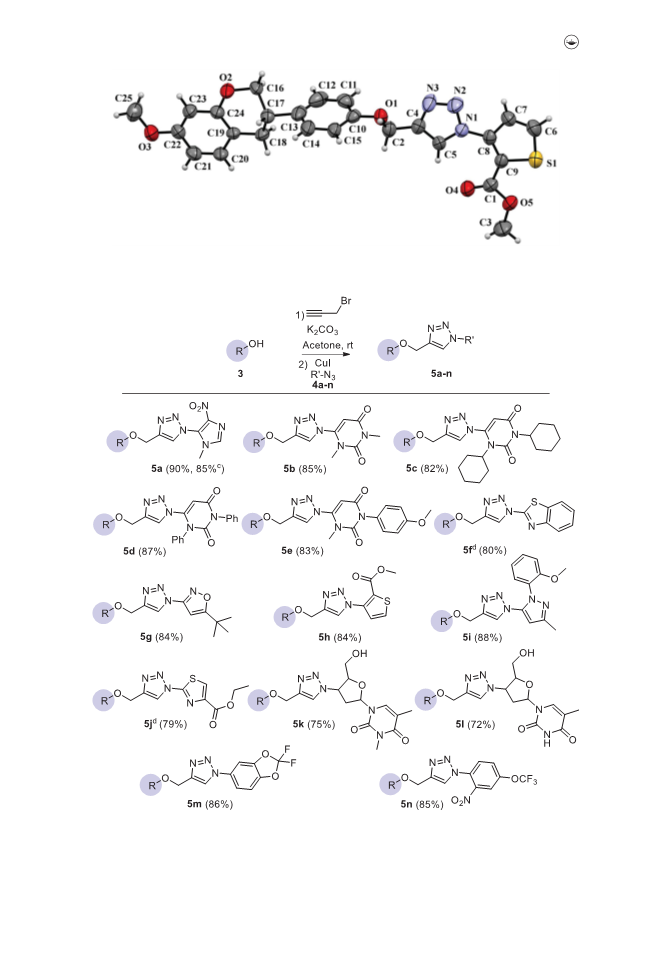
We herein report a convenient methodology for the synthesis of 1-hetero(aryl)-1,2,3-triazoles linked with equol by utilizing copper-catalyzed azide-alkyne cycloaddition reaction under exceptionally mild conditions. The salient features of this developed p
View More



Wuhan Hanye Chemical New Material Co.,Ltd
Contact:+86-27-85308141
Address:LiuDian, Panlongcheng Economic Development Zone, HuangPi district, Wuhan, Hubei 430311 P.R.China
SHAANXI TOP PHARM CHEMICAL CO.LTD
Contact:+86-029-85733403
Address:No.108 ,west sector,south er huan,xi'an,china
Jinan Jiaquan Chemical Co.,Ltd
Contact:+86-531-62318366
Address:Room 502 of Yidonghuayuan No.601 Huaxin Road Licheng District Jinan China
Contact:+86-27-67841589
Address:Add: 999 Gaoxin Road, Donghu New Technology Development Zone, Wuhan City, Hubei, China
Zibo Fuxi'er Chemical Co.,Ltd (Shanxian Fuxi'er Chemical Co.,Ltd)
Contact:+86-533-2091422
Address:Eastern 4 on the 3th Road ,Liangxiang Industrial Park, Zibo city ,Shandong,China
Doi:10.1111/j.1151-2916.1995.tb08388.x
(1995)Doi:10.1002/mrc.4390
(2016)Doi:10.1016/j.bmc.2007.03.060
(2007)Doi:10.1080/00397919508015430
(1995)Doi:10.1002/anie.202000105
(2020)Doi:10.1039/c5cy01349f
(2016)