7
2
M. Jose John et al. / Thermochimica Acta 534 (2012) 71–76
Na C O to Na CO in the temperature range 783–803 K. Mechan-
hand, isoconversional methods do not compute a frequency factor
nor determine reaction models which are needed for a complete
and accurate kinetic analysis. In solid state kinetics, mechanistic
interpretations usually involve identifying a reasonable reaction
model [31] because information about individual reaction steps is
often difficult to obtain. A model can describe a particular reac-
tion type and translate that mathematically into a rate equation.
Many models have been proposed in solid-state kinetics and these
models have been developed based on certain mechanistic assump-
tions. Solid-state kinetic reactions can be mechanistically classified
as nucleation, geometrical contraction, diffusion and reaction order
models [10]. The TG data were subjected to weighted least squares
analysis by all kinetic models given in [10].
2
2
4
2
3
ical addition of foreign substances into inorganic salts is known to
give rise to two different types of effects on the thermal behavior
of salts: (i) by the formation of a stable compound between the salt
and the additive [20] and (ii) by a catalytic action of the additive
[
21–24]. Algird [9,25] observed that electron donors when present
along with Ag C O accelerates the thermal decomposition rate
2
2
4
and electron acceptors retards.
CuO is a black solid having molar mass of 79.545 with an ionic
structure. It melts above 1474 K with some loss of oxygen. CuO
has application as a p-type semiconductor, because it has a narrow
band gap of 1.2 eV [26]. The effectiveness of the reagent depends on
the method of preparation, a problem that is typical for other het-
erogeneous reagents where surface area, among other variables, is
significant [27]. TiO is a white solid having a molar mass of 79.870.
2
3
. Results and discussion
It melts at 2143 K and boils at 3245 K. Titanium dioxide is the most
widely used white pigment because of its brightness and very high
refractive index (ꢀ = 2.4). It is also used as a semi-conductor [28].
The effects of semiconducting oxides such as CuO and TiO2 on
the thermal decomposition kinetics of Na C O were examined
2
2
4
using mechanical mixtures of compositions 0.5, 1, 2, 5 and 10 wt%
of the oxide. The experimental mass loss data obtained from TG
were transformed into ˛ versus t data as reported earlier [32], in
the range ˛ = 0.05–0.95 with an interval of 0.05, at all tempera-
tures studied. The ˛ versus t curves for the thermal decomposition
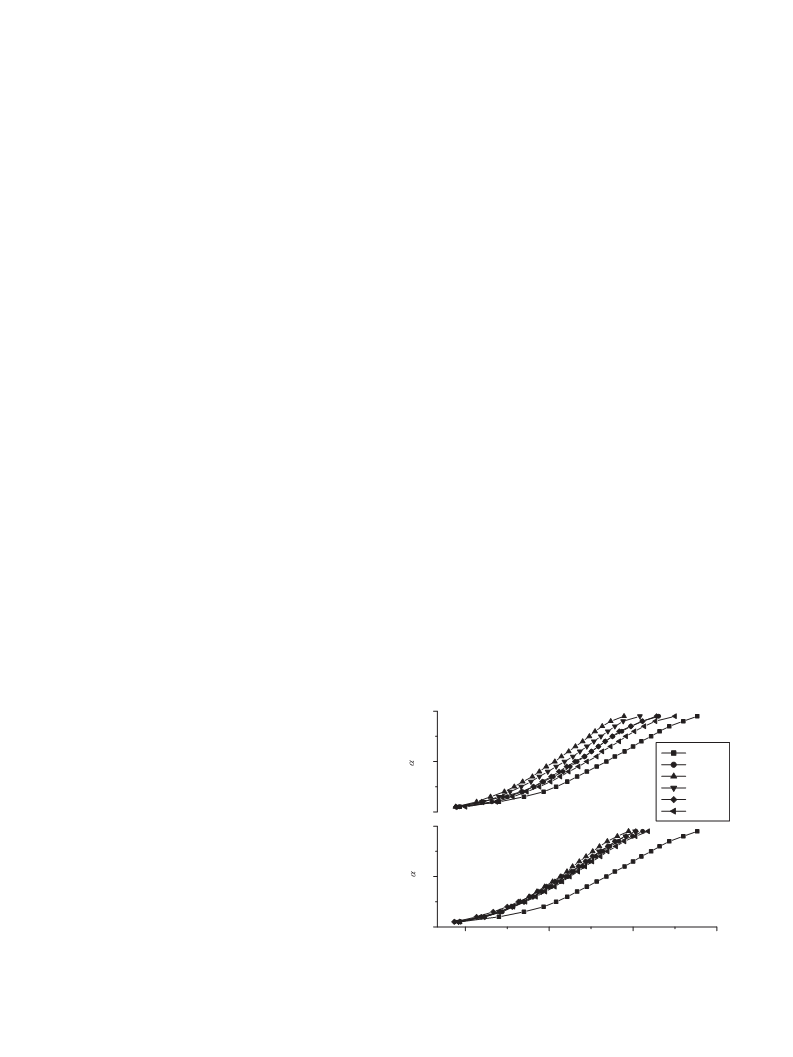
of all oxide mixed samples are shown in Figs. 1 and 2. The effect
2
. Experimental
2.1. Materials
All the chemicals used in the present study were of AnalaR grade
samples of E Merck. Mechanical mixtures of sodium oxalate and
metal oxides, CuO and TiO , were prepared by mixing the oxalate
and oxide of same particle size, viz., 106–125 m. Different samples
with oxide concentrations 0.5, 1, 2, 5 and 10 wt% were prepared by
mixing 2 g sodium oxalate thoroughly, in an agate motar, with the
required quantity of the oxide.
of pre-compressed oxide (CuO and TiO ) additives on the ther-
2
2
mal decomposition of Na C O was also examined at 783 K; the
2
2
4
˛
versus t plots is shown in Fig. 3.
Weighted least squares analysis of the thermal decompo-
sition of oxide mixed samples of Na C O showed that, as
2
2
4
with untreated sodium oxalate [11], there are two stages in
the decomposition; an acceleratory stage up to ˛ = 0.5 fol-
lowed by the decay stage, which are respectively described by
the Prout–Tompkins {ln[˛/(1 − ˛)] = kt} and contracting cylinder
Pre-compressed oxides (CuO and TiO ) were prepared by sub-
2
jecting to compression to 0.5 g each of CuO and TiO2 of particle
3
size 106–125 m in a hydraulic press at pressures of 2 × 10 ,
3
3
−2
4
× 10 and 6 × 10 kg cm . The pellets of metal oxides were taken
1/2
model [1 − (1 − ˛) = kt] rate laws with separate rate constants,
out, powdered in an agate mortar and fixed the particle size in
the range 106–125 m. Required quantity of the pre-compressed
oxide was thoroughly mixed with sodium oxalate (particle size:
k
1
and k . The rate constants obtained for both stages of thermal
2
decomposition are shown in Table 1. Both oxides cause an increase
in the rates of both stages of decomposition up to an oxide concen-
tration of 1 wt% and then decreases (see Fig. 4 and Table 1). The
effect of mixing pre-compressed oxides on the thermal decom-
position of Na C O was also examined at 783 K (Table 2). The
1
2
2
06–125 m), in an agate mortar, to get 1 wt% mixture.
.2. Methods
2
2
4
dependence of the rate constant of both acceleratory and decay
stages of the thermal decomposition of pre-compressed oxide
mixed sodium oxalate on the concentration of pre-compressed
.2.1. Thermogravimetric analysis
Thermogravimetric measurements in static air were carried out
on a custom-made thermobalance fabricated in this laboratory
15,29]. A major problem [30] of the isothermal experiment is that
[
a sample requires some time to reach the experimental temper-
ature. During this period of non-isothermal heating, the sample
undergoes some transformations that are likely to affect the suc-
ceeding kinetics. The situation is especially aggravated by the fact
that under isothermal conditions, a typical solid-state process has
its maximum reaction rate at the beginning of the transformation.
So we fabricated a thermobalance particularly for isothermal stud-
ies, in which loading of the sample is possible at any time after
the furnace has attained the desired reaction temperature. The
operational characteristics of the thermobalance are, balance sen-
1
0
.0
.5
CuO
0
wt %
0.5 wt %
1 wt %
2
5
wt %
wt %
0
1
.0
.0
10 wt %
TiO2
−5
sitivity: ± 1 × 10 g, temperature accuracy: ± 0.5 K, sample mass:
−
2
5
× 10 g, atmosphere: static air and crucible: platinum. Thermal
decomposition of sodium oxalate was found to be very slow below
83 K and very fast above 803 K. The decomposition was thus stud-
0.5
7
ied in the range 783–803 K. The loss in mass of sodium oxalate was
measured as a function of time (t) at five different temperatures (T),
viz., 783, 788, 793, 798 and 803 K.
0
.0
1
5
30
45
60
t / min
2.2.2. Kinetic analysis
Historically model-fitting methods were widely used because of
Fig. 1. ˛ versus t plots for the thermal decomposition of oxide mixed sodium oxalate
their ability to directly determine the kinetic triplet. On the other
samples at different concentrations at 783 K.

 Jose John
Jose John
 Muraleedharan
Muraleedharan
 Kannan
Kannan
 Ganga Devi
Ganga Devi