1
20
M.O. Awaleh et al. / Journal of Alloys and Compounds 349 (2003) 114–120
1
3
00–650 8C, is attributed to the departure of three moles of
polyhedra, separated by four remaining Na cations. The
CO gas per two moles of Na Y(CO ) (exp./th.514.9/
[NaYb(CO ) ] layers in Na Yb(CO ) ?2H O transform
2
5
3
4
3
4
`
5
3
4
2
1
2
4.6%); the decomposition reaction can be written:
to [NaYb(CO ) O ] layers, connected by the remaining
3 2 6 `
carbonate groups in Na Yb(CO ) .
5
3 4
Na Y(CO ) → Y O 1 5Na CO 1 3CO
2
5
3
4
2
3
2
3
The second step corresponds to the decomposition of
Na CO and occurs above 850 8C.
References
2
3
The thermal decomposition of Na Yb(CO ) ?2H O
5
3
4
2
occurs in the intervals 190–250 8C, 280–650 8C, and
above 850 8C. The first weight loss is attributed to the
departure of two moles of water per one mole of
[1] Y.B. Khollam, A.S. Deshpande, A.J. Patil, H.S. Potdar, S.B.
Deshpande, S.K. Date, Mater. Chem. Phys. 71 (2001) 235.
[2] J. Cai, J. Liu, Z. Gao, A. Navrotsky, S.L. Suib, Chem. Mater. 13
(
2001) 4595.
Na Yb(CO ) ?2H O (exp./th.56.3/6.4%); X-ray diffrac-
5
3
4
2
[3] B.L. Newalkar, J. Olanrewaju, S. Komarneni, Chem. Mater. 13
(2001) 552.
[4] K.J. Rao, B. Vaidhyanathan, M. Ganguli, P.A. Ramakrishnan, Chem.
Mater. 11 (1999) 882.
tion analysis of the residual shows that the intermediate
phase is Na Yb(CO ) , isostructural with Na Y(CO ) .
5
3
4
5
3 4
However, the crystallinity of this intermediate phase is
poor and the diffraction line width is large. Consequently,
[5] A. Ben Ali, M.O. Awaleh, V. Maisonneuve, M. Leblanc, J. Solid
State Chem. (submitted).
[6] A. Ben Ali, V. Maisonneuve, M. Leblanc, Solid State Sciences
(submitted).
accurate cell parameters of Na Yb(CO ) cannot be given
5
3
4
here. The second and third weight loss steps are similar to
that of Na Y(CO ) . The experimental and theoretical
[7] J.D. Grice, G.Y. Chao, Can. Miner. 35 (1997) 743.
5
3 4
[8] J.D. Grice, Can. Miner. 34 (1996) 649.
second weight loss steps are 11.3 and 11.2%, respectively.
[9] J.D. Grice, R.A. Gault, Can. Miner. 36 (1998) 1293.
[
[
10] H. Schweer, Z. Seidel, Z. Anorg. Allg. Chem. 477 (1981) 196.
11] A. Mochizuki, K. Nagashima, H. Wakita, Bull. Chem. Soc. Jpn. 47
(1974) 755.
6
. Conclusion
[
12] G.M. Sheldrick, in: G.M. Sheldrick, C. Kr u¨ ger, R. Goddard (Eds.),
SHELXS-86 in Crystallographic computing 3, Oxford University
Press, London, 1985, pp. 175–189.
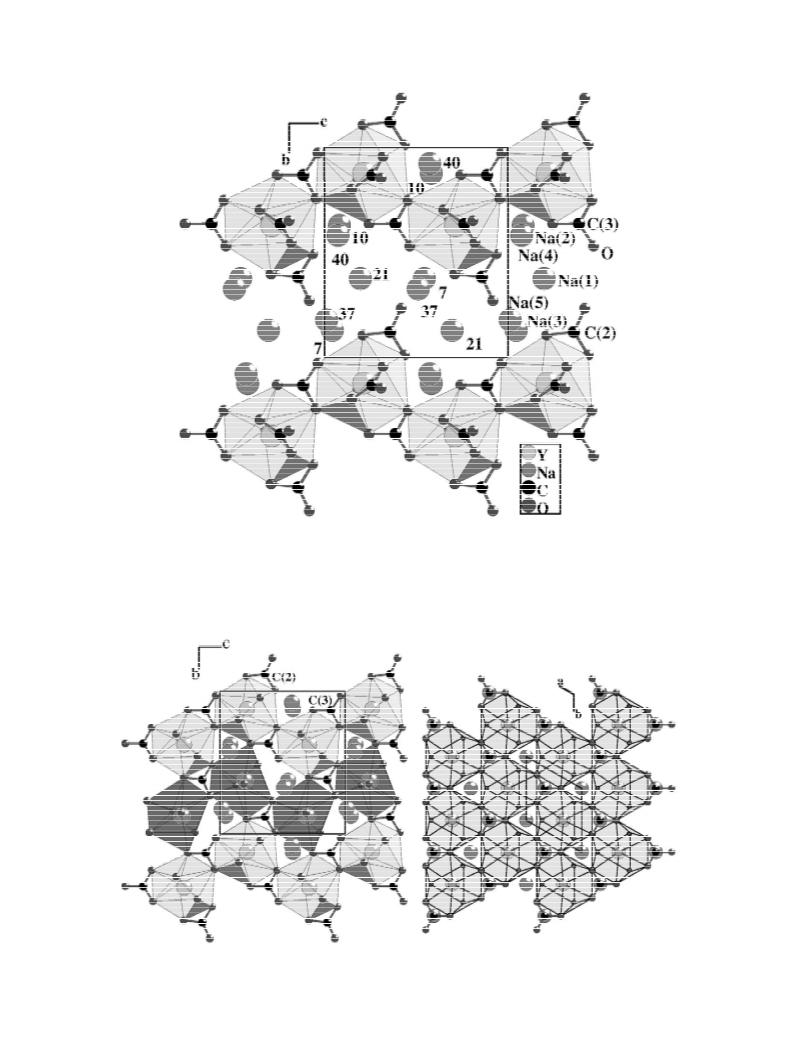
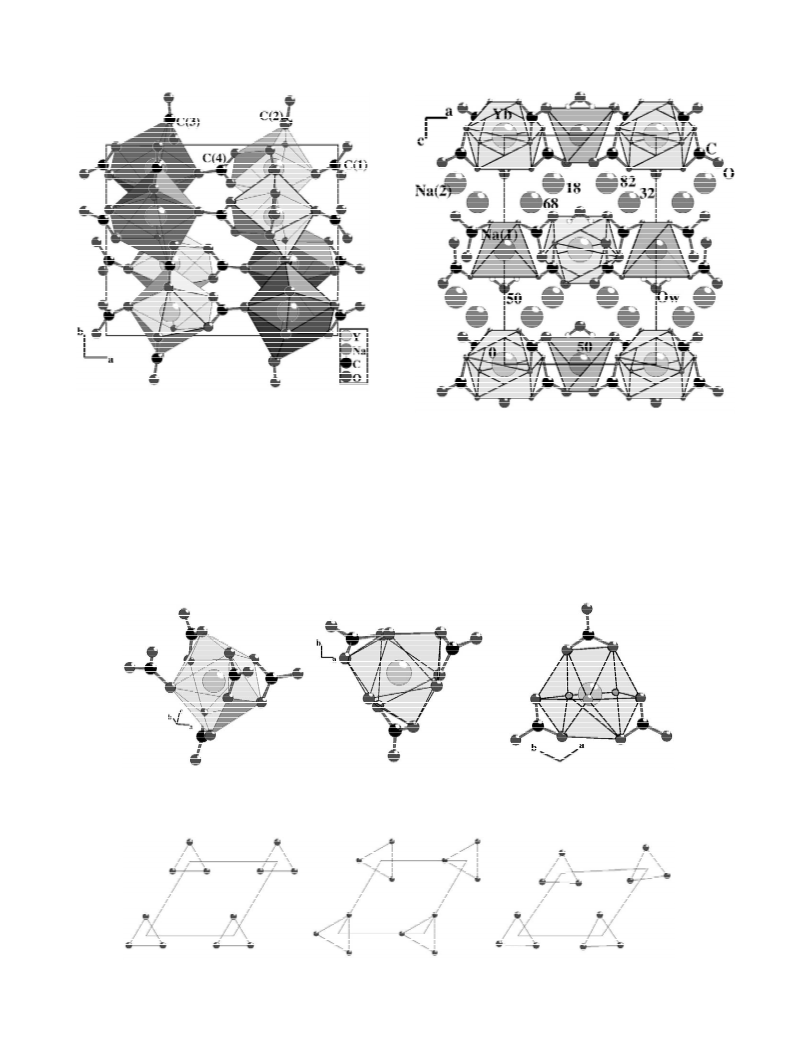
The crystal structures of two carbonates, Na Y(CO )
3 4
5
and Na Yb(CO ) ?2H O, are determined. Both com-
pounds are obtained by microwave-assisted hydrothermal
5
3
4
2
[13] G.M. Sheldrick, SHELXL-97, a Program for Crystal Structure
Determination, 1997, G o¨ ttingen University, Germany.
method in sub-critical conditions. Na Yb(CO ) ?2H O,
[14] G.M. Sheldrick, in: SHELX-76: A Program for Crystal Structure
Determination, Cambridge University Press, Cambridge, 1976.
5
3
4
2
isostructural with Na Sc(CO ) ?2H O, undergoes dehy-
5
3
4
2
[
15] N.E. Brese, M. O’Keeffe, Acta Crystallogr. B47 (1991) 192.
16] J. Rodriguez-Carvajal, FULLPROF, in: Abstracts of the Satellite
Meeting on Powder Diffraction of the XVth Congress of the IUCr,
Toulouse, France, 1990, p. 127.
dration at 190,T ,250 8C and leads to Na Yb(CO )
5
3 4
[
which is isostructural with Na Y(CO ) . In the structure of
5
3 4
2
2
22
Na Y(CO ) , C(2)O and C(3)O3 carbonate groups are
5
3
4
3
stacked in dense ‘flat lying’ layers between which ‘stand-
[17] T.A. Zhdanova, A.A. Vorancov, L.N. Komissarova, J.A. Pajatenko,
Dockl. Akad. Nauk 196 (1971) 1071.
2
2
22
ing on base’ and ‘standing on top’ C(1)O and C(4)O3
3
[
18] N. Mercier, M. Leblanc, J. Durand, Eur. J. Solid State Inorg. Chem.
4 (1997) 241.
carbonate groups are inserted. Dehydration of
3
Na Yb(CO ) ?2H O to give Na Yb(CO ) implies that
5
3
4
2
5
3 4
[19] N. Mercier, F. Taulelle, M. Leblanc, Eur. J. Solid State Inorg.
Chem. 30 (1993) 609.
ytterbium coordination increases, from 8 to 9, simul-
1
taneously with Na(1) coordination. In both structures, it
[20] J.D. Grice, J .V . Velthuizen, R.A. Gault, Can. Miner. 32 (1994) 405.
1
31
is remarkable that Na(1) and Yb form infinite layers of

 Awaleh
Awaleh
 Ben Ali
Ben Ali
 Maisonneuve
Maisonneuve
 Leblanc, Marc
Leblanc, Marc