D. Bakirtzis et al. / Thermochimica Acta 486 (2009) 11–19
19
as the mass concentration of NaHCO3 increases [21]. It is difficult to
completely quantify the ignition process in this experiment because
the heat flux imposed to the sample is not well defined. This heat
flux consists of radiative, convective and conductive (to the base
heat flux and the critical heat flux decreases.
Secondly, following, the previous observations, we note that the
relative decrease in the time to self ignition as the temperature
increases (see Figs. 11–13) can be explained by noting that the rel-
ative magnitude of the net heat flux decreases as the temperature
(and hence the imposed heat flux) increases.
In this paper, only time to self ignition have been recorded;
which is totally different from the time to piloted ignition. Piloted
ignition occurs in less time than the time to self ignition and at
much lower fuel temperatures (300 ◦C instead of 500–600 ◦C) and
so it could be influenced by the fire retardant in a different way.
to reach ignition conditions due to the thermal decomposition of
the sodium bicarbonate.
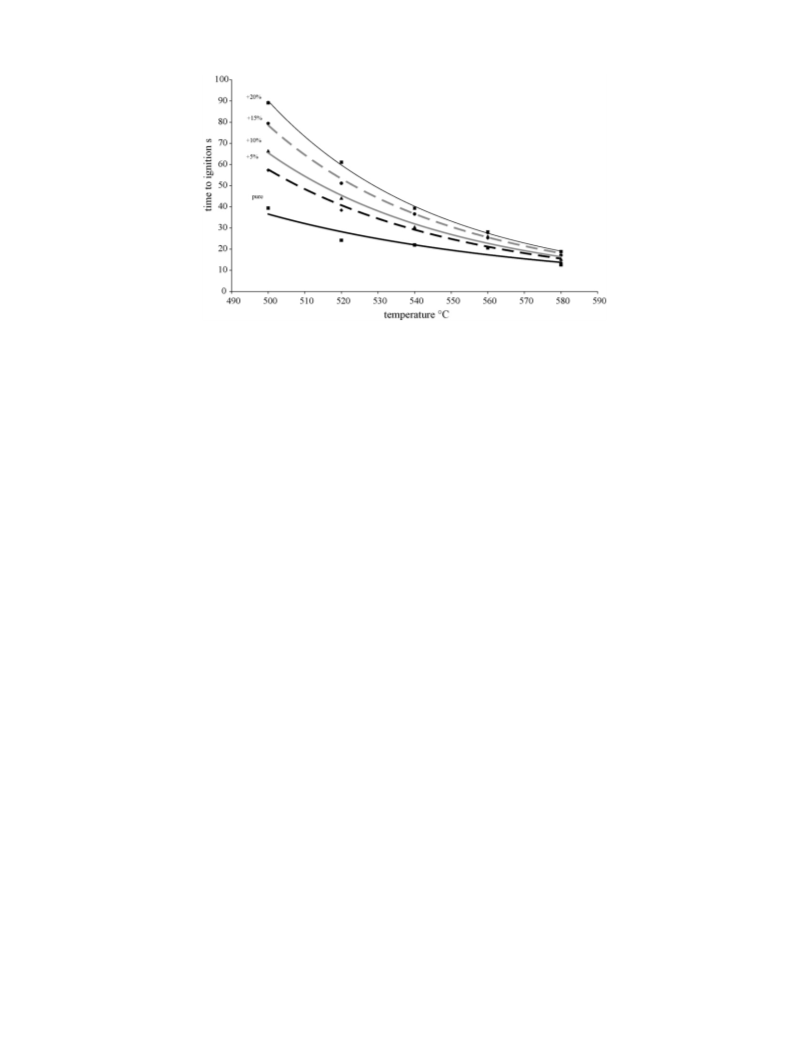
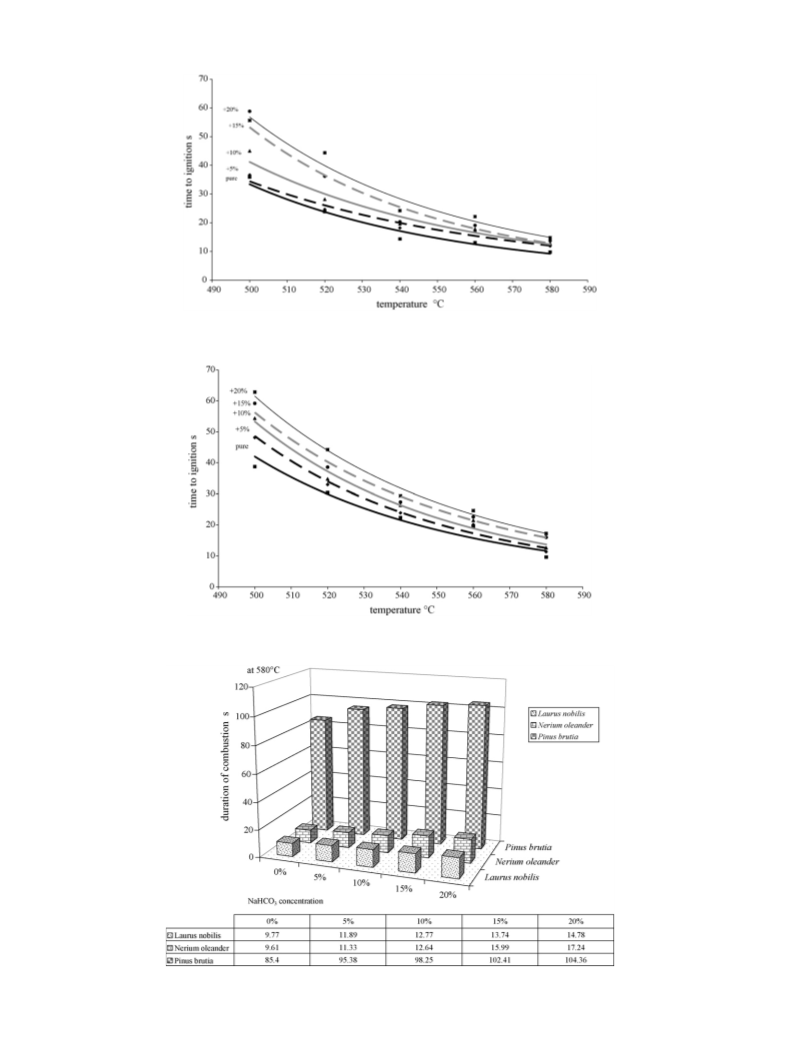
The duration of combustion increased slightly with the addition
of NaHCO3 as Fig. 14 and the associated table show, for a furnace
temperature of 580 ◦C. Similar results were obtained for the other
furnace tested temperatures in this study. The difference in the
duration of combustion was, however, significantly greater for P.
brutia compared to the two other species. Thus, sodium bicarbonate
decreases the threshold of initial temperature of pyrolysis of forest
species (promotion effect). On the other hand NaHCO3 increases the
self ignition delay time and combustion duration of forest species
(inhibition effect). In addition the presence of NaHCO3 increases the
pyrolysis mass residue of forest species, especially when sodium
carbonate applied on N. oleander.
Overall, NaHCO3 exhibits a satisfactory fire retardant perfor-
mance on lignocellulosic materials, in terms of reducing time to
self ignition, mass residue and should be considered important long
term fire retardant additive for the suppression of wild land fires.
3.3.2. Duration of combustion
Acknowledgements
The duration of combustion increased with the addition of
NaHCO3 as Fig. 14 and the associated table show for a furnace
temperature of 580 ◦C. That means also that the heat released
rate decreased if we assume that the total heat released remains
the same for all fire retardant concentrations. Similar results were
obtained for the other furnace temperatures tested in this study.
However, the difference in the time of combustion for the specie
considered is pronounced. The increase of the burning time of the
volatiles is attributed to a heat of pyrolysis of P. brutia higher than
the heat of pyrolysis of the other two species. This higher heat of
pyrolysis decreases the mass burning rate (and so increases the
burning time) because the imposed heat flux is the same at a given
temperature of the furnace. The differences observed in the vari-
ous forest specie studied are due to the composition of the specie
regarding the amount of hemicellulose, cellulose and lignin.
The authors would like to express acknowledgements to Prof.
Euripides Lois of the National Technical University of Athens, for
his assistance and useful discussion.
References
[1] S. Liodakis, D. Vorisis, I. Agioblasitis, Thermochim. Acta 444 (2006) 157–165.
[2] P. Santoni, A. Simeoni, J. Balbi, J. Rossi, T. Marcelli, Fire Saf. J. 41 (2006) 229–235.
[3] Parliamentary Assembly, Recommendation 1761 (2006).
[4] S. Liodakis, D. Bakirtzis, E. Lois, D. Gakis, Fire Saf. J. 37 (2002) 481–494.
[5] P.K. Heda, D. Dollimore, K. Alexander, D. Chen, E. Law, P. Bicknell, Themochim.
Acta 255 (1995) 255–272.
[6] V.I. Babushok, K.L. McNesby, A.W. Miziolek, R.R. Skaggs, Combust. Flame 133
(2003) 201–205.
[7] H.K. Chelliaah, P.C. Wanigarathne, A.M. Lentari, G.S. Fallon, Combust. Flame 134
(2003) 261–272.
[8] A.F. Roberts, Combust. Flame 14 (1970) 261–271.
4. Conclusions
[9] M.Z. Sefain, Z.A. Nagieb, N.A. El-Wakil, M.M. Naoum, Thermochim. Acta 191
(1991) 21–27.
[10] V. Leroy, D. Cancellieri, E. Leoni, Thermochim. Acta 451 (2006) 131–138.
[11] R. Artiaga, S. Naya, A. García, F. Barbadillo, L. García, Thermochim. Acta 428
(2005) 137–139.
[12] G. Steiner, J. Rath, M.G. Wolfinger, G. Staudinger, Thermochim. Acta 398 (2003)
59–71.
[13] Fire Protection Handbook, NFPA, Quincy, Massachusetts, pp. 1.42–1.56.
[14] A.N. Shebani, A.J. van Reenen, M. Meincken, Thermochim. Acta 471 (2008)
43–50.
[15] J.L. Grandmaison, J. Thibault, S. Kaliaguine, Anal. Chem. 59 (1987) 2153–2157.
[16] J. Reina, E. Velo, L. Puigjaner, Thermochim. Acta 320 (1998) 161–167.
[17] N. Grioui, K. Halouani, A. Zoulalian, F. Halouani, Thermochim. Acta 440 (2006)
23–30.
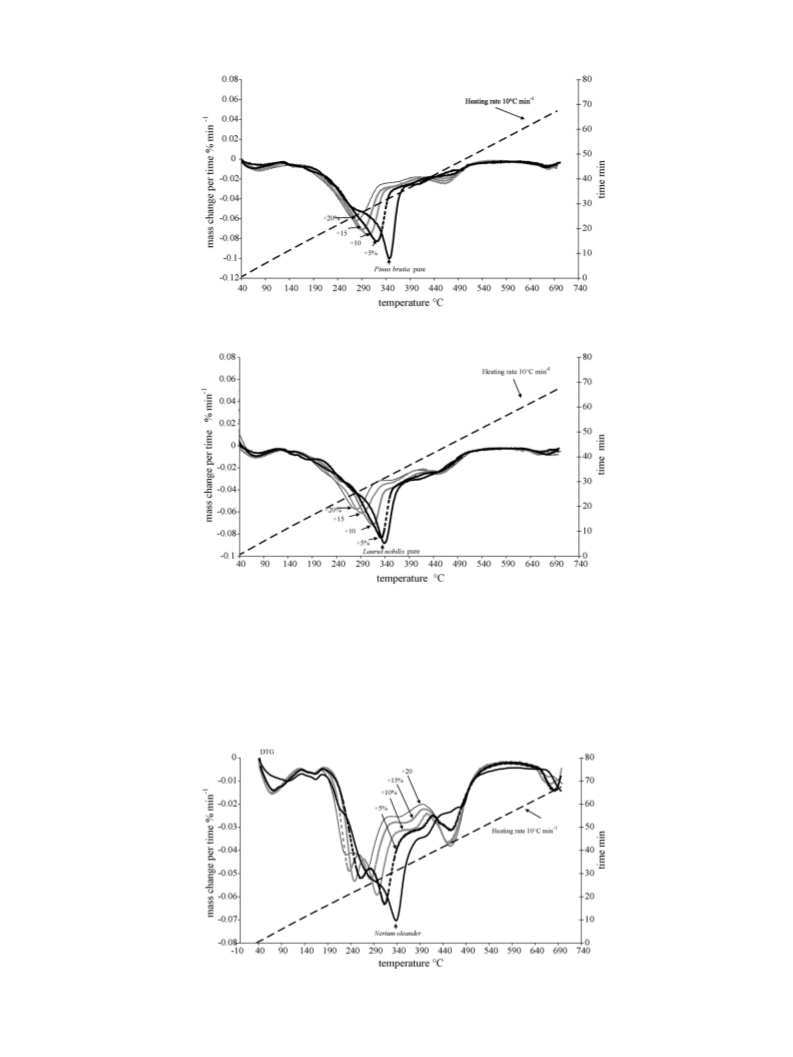
For all three forest species, the thermal degradation profile
was shifted towards lower temperatures. This means that NaHCO3
induced an early emission of the volatile gases by influencing the
kinetics of hemicelluloses and cellulose.
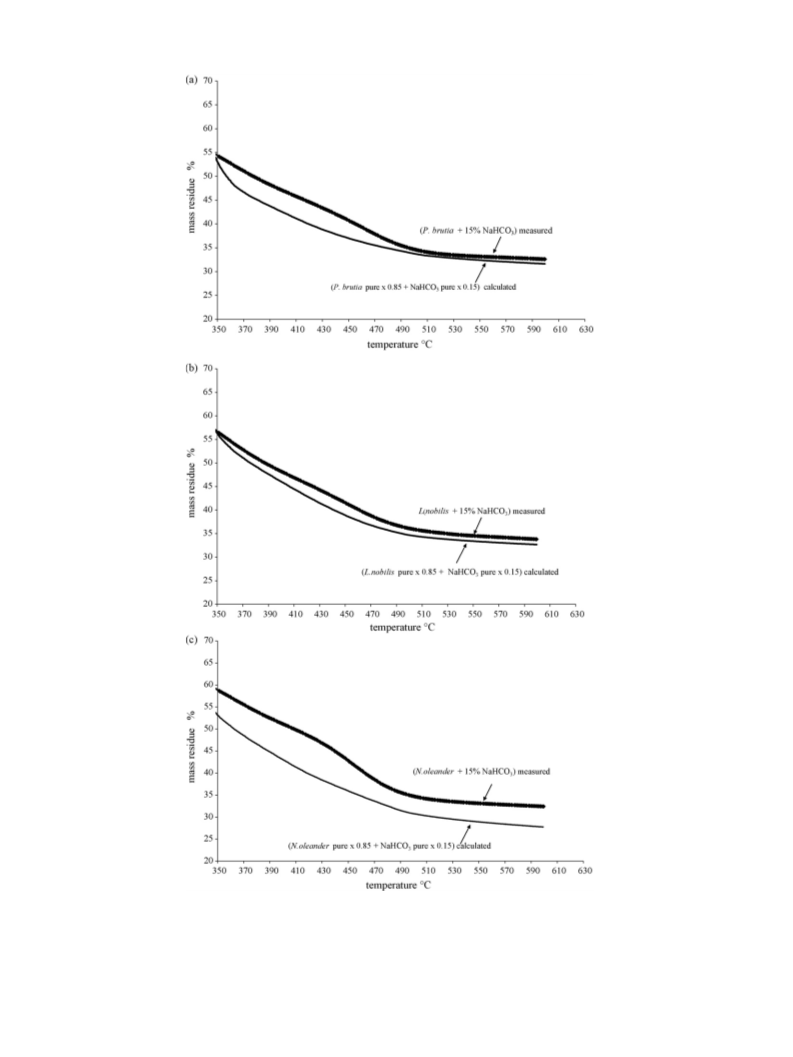
As far it concerns the effect of NaHCO3 on the mass residue, on
P. brutia, L. nobilis was additive and synergistic to N. oleander as
explained.
Time to ignition increased by adding NaHCO3. That was due of
the alteration of the composition of the emitted flammable gases
at the vicinity of the sample, due to the release of water (H2O) and
carbon dioxide (CO2) produced by the decomposition of sodium
bicarbonate. On top, additional energy was needed for the system
[18] E. Hugi, M. Wuersch, J. Wood Sci. 53 (2007) 71–75.
[19] S. Liodakis, D. Bakirtzis, A. Dimitrakopoulos, Thermochim. Acta 390 (2002) 83.
[20] A. Mlaouhi, A.A. Khouja, Renew. Energy 16 (1999) 1118–1121.
[21] D. Bakirtzis, PhD Thesis, National Technical University of Athens, 2005.

 Bakirtzis
Bakirtzis
 Delichatsios
Delichatsios
 Liodakis
Liodakis
 Ahmed
Ahmed