G. Li et al.
MolecularCatalysis455(2018)95–102
3.2. Hydrogenation of LA over Ru-based catalysts
Table 1
Structural properties of nano-TiO2.
The hydrogenation of LA to GVL is carried out in water at 30 °C
under 1.0 MPa (Table 2). A blank test shows that nearly no LA is
transformed when TiO2-0.4 is used as catalyst (entry 1). Ru/TiO2-0.4
exhibits significantly higher catalytic activity (97.4%) with a GVL se-
lectivity of 100% (entry 2). Ru/TiO2-1, Ru/TiO2-2 and Ru/TiO2-3 af-
ford 68.2, 62.8 and 15.5% LA conversion, respectively (entries 3–5),
much lower than that of Ru/TiO2-0.4. The TOF value based on total Ru
atoms of Ru/TiO2-0.4 is as high as 799.8 h−1, which is 1.42, 1.54 and
6.26 times of Ru/TiO2-1, Ru/TiO2-2 and Ru/TiO2-3, respectively. For
comparative purposes, commercial TiO2 supported Ru catalyst (Ru/
TiO2-C) shows a poor activity (19.0%) for LA hydrogenation (entry 6).
Other frequently used heterogeneous Ru-based catalysts inclusive of
Ru/AC, Ru/BC and Ru/MCN achieve obviously slower hydrogenation
rate and relatively lower LA conversion (< 50%) under the same con-
ditions (entries 7–9). Some other literatures have also been reported for
the low-temperature hydrogenation of LA, but a high hydrogen pressure
was generally required [14,20,29]. For example, 4 MPa H2 was required
for the CTH of LA with water over graphene-supported Ru catalysts
(Ru/RGO) [20,29]. Obviously, our method is preferable because only
1 MPa and 30 min was needed at room temperature.
The time-on-stream experiments over Ru/TiO2-0.4 and Ru/TiO2-3
(Fig. 6) indicate that the hydrogenation rate of LA over Ru/TiO2-0.4 is
much higher than that of Ru/C-TiO2-3. For example, > 95% conversion
was observed in 30 and 150 min for Ru/TiO2-0.4 and Ru/TiO2-3, re-
spectively. The catalytic performances of Ru/TiO2-0.4 were further
investigated at lower temperatures (20 and 10 °C) (Table 2, entries 10
and 11). Although an obvious decrease in reaction rate is observed as
decreasing the reaction temperature to 20 and 10 °C, Ru/TiO2-0.4 still
gives 99.1 and 88.5% LA conversion by prolonging time to 1.5 and 3 h,
respectively, indicating Ru/TiO2-0.4 is indeed an excellent low-tem-
perature catalyst for LA hydrogenation.
Samples Stotal
(m2/g)
Pore volume Average pore
TiO2 size TiO2 size (nm)b
(cm3/g)
diameter (nm)
TiO2-0.4 59.4
0.024
0.018
0.015
0.003
7.37
6.16
7.36
6.44
11.1
11.2
13.6
25.2
34.8
40.6
70.3
385.6
6.6
17.7
27.2
TiO2-1
TiO2-2
TiO2-3
48.9
30.8
2.7
211.9
a
Determined by XRD.
Determined by SEM.
b
[31]. The peak width of (101) diffraction peak becomes sharp as in-
creasing the HF amount from 0.4 to 3.0 mL. The average TiO2 crystallite
sizes of the samples (Table 1) are calculated via the Debye-Scherrer
equation based on the main diffraction (101) peak (2θ = 25.28°, Fig.
S1), in which the crystallite size of TiO2-0.4 is around 11.1 nm. UV–vis
analysis (Fig. S2) shows that all TiO2 samples afford similar absorption
at wavelength range 200–400 nm. As the HF amount decreases, the
absorbance increases gradually, indicating the size of TiO2 decreases
[32]. The morphology of TiO2 was observed by SEM. As shown in
Fig. 2, all samples appear to be composed of primary rough particles
and the HF amount has a profound influence in particle size of TiO2. For
example, TiO2-3 exhibits a big particle size of 385 nm along with a wide
size distribution, decreasing the HF amount sharply decreases the
particle size as well as size distribution, and TiO2-0.4 shows particles
with fairly uniform size of 34.8
6.6 nm. In addition, with the de-
crease of the HF amount from 3 to 0.4 mL, the surface areas and pore
volumes are largely increased from 2.7 m2/g and 0.003 cm3/g for TiO2-
3 to 59.4 m2/g and 0.024 cm3/g for TiO2-0.4, respectively (Table 1).
These results indicate that the HF amount plays an important role in
controlling the particle size of TiO2 [33].
Ru-based catalysts are synthesized by a facile impregnation strategy
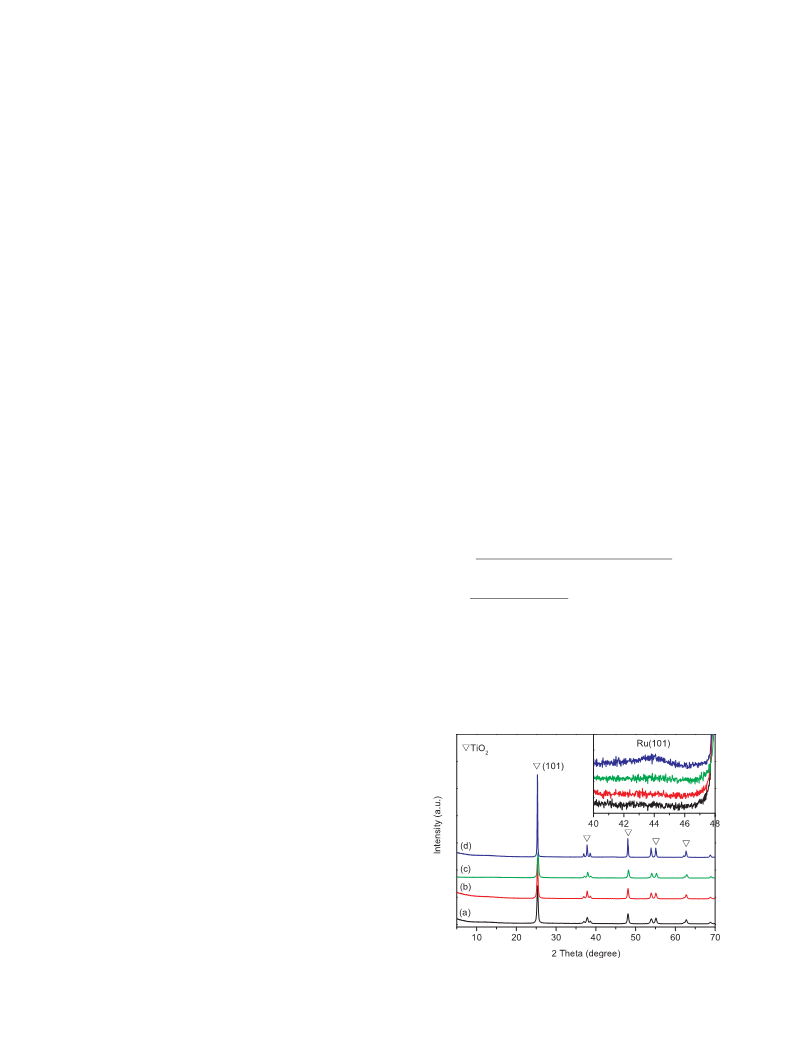
using H2 as the reductant. The XRD pattern (Fig. 1) of Ru/TiO2-0.4
shows nearly no Ru reflection peak at 2θ = 44° compared with Ru/
TiO2-2 and Ru/TiO2-3, indicating the presence of small-sized (smaller
than 3–4 nm in size) and highly dispersed Ru NPs [34]. TEM images
(Fig. 3) reveal that Ru/TiO2-0.4 is comprised of crystalline TiO2 NPs of
around 30 nm in diameter, well agreed with the SEM images. The
HRTEM image (Fig. 3c) shows that Ru NPs with an average size of
1.69 nm are well dispersed in uniform TiO2 NPs without any aggrega-
tion.
In order to further investigate the valence of metal-based species, Ru
XPS analyses are measured (Fig. 4). It is found that the Ru 3d spectra of
Ru/TiO2-0.4 exhibits the lowest binding energy in comparison to those
of other samples, indicating more intimate interaction occurs between
the active Ru sites and TiO2-0.4, thus leading to electron-sufficient Ru
species on TiO2-0.4 [35].
Among the solvents tested (Fig. 7), water is more favorable than
organic solvents for LA hydrogenation. Although the solubility of H2 in
water is much lower than in organic solvents, it was reported that H
spillover can be greatly promoted by water, and H atom of water par-
ticipated in the hydrogenation of the C]O group of LA, which en-
hanced the hydrogenation reaction rate. 37 The reaction slows down at
low catalyst loading but without any losing of the selectivity (Fig. 8).
The effect of hydrogen pressure on catalytic activity is studied (Fig. 9).
Although an obvious decrease in activity is observed as decreasing the
hydrogen pressure, Ru/TiO2-0.4 still gives 33.2% LA conversion at a
low hydrogen pressure of 0.2 MPa.
To further showcase the effectiveness of Ru/TiO2-0.4 catalyst, we
carried out the LA hydrogenation on a one-gram scale (Fig. 10). Direct
LA (1.16 g) hydrogenation with an even lower catalyst loading of
0.0487 mol% Ru at 30 °C for 10 h provides GVL with 85% yield, which
appears to be efficient for industrial application.
H2-TPR profiles of Ru/TiO2 catalysts are shown in Fig. 5. As for Ru/
TiO2-C derived from commercial TiO2, a big peak centered at 199 °C
with a big shoulder at 173/138 °C can be observed. The reduction peaks
of Ru/TiO2-3 downshift to 148 and 189 °C, indicating the existence of
small-sized Ru NPs on surface of TiO2-3. As for Ru/TiO2-2, the reduc-
tion peak is broad and centers at 169 °C. Further decreasing the HF
amount, the reduction peak moves to low-temperature range, and Ru/
TiO2-0.4 affords the lowest reduction temperature (117 and 158 °C),
indicating the smallest size of NPs and easiest reduction property of Ru
species [34]. It is well known that metals supported on titania exhibit
the strong metal-support interaction effect, which leads to the forma-
tion of an electron-rich metal [25,36]. Small-sized TiO2 NPs afford
sufficient contact with metal NPs, thus leading to much stronger metal-
support interaction, which is known to modify the selectivity and ac-
tivity of a catalyst [23].
To further demonstrate the versatility of the Ru/TiO2-0.4 catalyst,
the selective HDO of biomass-derived vanillin to 2-methoxy-4-methyl-
phenol (MMP) in water was performed at 70 °C under 1.0 MPa H2
(Table 3). Similarly, Ru/TiO2-0.4 shows the best activity and affords
53.9% MMP yield for 2 h, this value is 4.6, 6.7 and 8.7 times higher
than that of Ru/TiO2-1, Ru/TiO2-2 and Ru/TiO2-3, respectively. Fur-
thermore, nearly 100% conversion and 100% MMP selectivity could
reach over Ru/TiO2-0.4 catalyst by prolonging reaction time to 4 h,
along which a high specific activity of 51.3 h−1 could be achieved. The
Ru/TiO2-0.4 catalyst even exhibited much higher activity than other
reported metal catalysts [38]. For example, carbon nanotube supported
ruthenium catalysts (Ru/CNT) with decalin/H2O as solvent produced
MMP with a slightly lower yield of 93% under harsh conditions (200 °C
Finally, recycling tests are performed to investigate the durability of
the catalyst (Fig. 11). The Ru/TiO2-0.4 shows a good stability with
maintained activity and selectivity during the tests. The ICP analysis of
97

 Li, Guoqiang
Li, Guoqiang
 Yang, Huanhuan
Yang, Huanhuan
 Cheng, Mei
Cheng, Mei
 Hu, Wei
Hu, Wei
 Tian, Lihong
Tian, Lihong
 Mao, Wuxiang
Mao, Wuxiang
 Nie, Renfeng
Nie, Renfeng