A R T I C L E S
Mere´nyi et al.
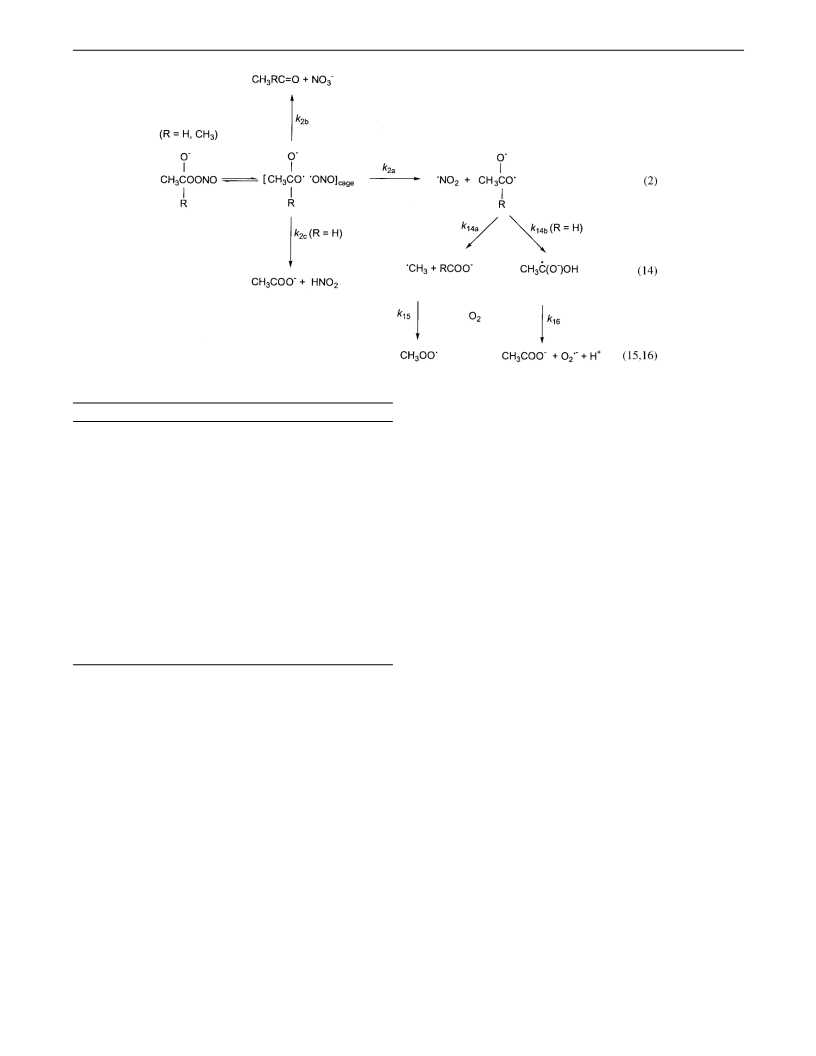
order. From the geminate pair that is formed by homolysis of
the adduct, a small part is released as free radicals (k2a). The
remainder collapses to molecular products in the solvent cage.
Process 2b is the predominant cage collapse with acetaldehyde
and the only cage process with acetone. This is reflected by the
very high yields of NO3- in all carbonyl/ONOO- systems.13-16
Process 2b is probably initiated by N-O coupling to yield a
short-lived R-hydroxyalkyl nitrate, followed by rapid heterolysis
resulting in nitrate and the initial carbonyl compound. Based
on the significantly higher yield of acetate as compared to that
of O2•- in the case of acetaldehyde,4,14 reaction 2c, which is an
in-cage hydrogen atom transfer, has been suggested to occur.4
Once the free radicals have diffused out of the solvent cage,
several processes ensue. Reaction 14a, i.e., â-scission,33 can take
place in the case of acetone and acetaldehyde, whereas reaction
14b, i.e., 1,2-H atom shift,34 occurs only in the case of
acetaldehyde.
oxidation by •NO2 (k ) 4.3 × 106 M-1 s-1).36 Therefore, while
•
0.1 M ferrocyanide is sufficient to reduce all NO2, it falls
somewhat short of capturing CH3OO• quantitatively. The data
in Table 3 reveal that the yield of the radicals escaping the cage
in the acetone system is above 14%. On the other hand, a radical
yield of 15% was obtained when we simulated the yield of
3-
Fe(CN)6 with known rate constants in the presence of 1 M
Fe(CN)6,4 and this yield was unaffected by a further increase
4-
in [Fe(CN)64-]. Thus, the combined Fe(CN)6 and TNM
experiments are consistent with a free radical yield of 14-15%
at low pH.
As mentioned above, the radical yield at high pH was not
measurable. However, the yield of the free radicals should be
equal to the acetate yield (reaction 14a, where R ) CH3), which
we determined to be 13 ( 2% at pH > 14. In conclusion, in
the reaction of ONOO- with acetone, the free radical yields at
low and high pH are similar and amount to 13-15%.
The reaction of ONOO- with acetaldehyde formed 8.7 (
0.4% O2NOO- of the initial ONOO- at low pH, which
approximates the yield of O2•- in this system and is in agreement
We found above that the reaction of ONOO- with acetone
yielded about 4% O2•- of the initial concentration of ONOO-,
where only about 1% was formed relatively fast, at the same
rate as ONOO- disappeared. We suggest that the fast formation
of about 1% O2•- is due to the self-decomposition of CH3OO•
during the lifetime of ONOO-, whereas the other 3% is
attributed to the self-decomposition of CH3OO• formed slowly
via the homolysis of CH3OONO2 (reaction -18), after all
ONOO- has been consumed. Since the self-recombination of
CH3OO• was shown to yield about 29% O2•-, and as the total
•-
with the previously reported yield of O2 determined by use
of TNM.4 The yield of ferricyanide at low pH and in the
presence of 0.1 M ferrocyanide is ca. 20% (Table 3). While
this concentration of ferrocyanide is sufficient to scavenge the
•
excess of NO2, it is not sufficient completely to prevent the
recombination of •NO2 with O2•- and to scavenge all CH3OO•.
We therefore used our suggested model (reactions 2 or 3, 14-
21) to simulate the yield of ferricyanide both at low and high
pH, taking into account 8.7% and 10.9% yields of O2•- at low
and high pH, respectively. We also included in this model the
oxidation of ferrocyanide by CH3OO• and •NO2 and the
reduction of Fe(CN)63- by O2•-, i.e., k ) 6.9 × 103 M-1 s-1 at
I ) 0.2 M.37 The model fits the experimental results (Table 3)
assuming about 16% radical yield and 47% â-scission at low
pH, and about 17% radical yield and 37% â-scission at high
pH. The low pH results are in fair agreement with previous
yields obtained at 37 °C, where the total radical yield was ca.
20%, about half of it being •CH3 radicals formed during
â-scission.14
O2 yield from the acetone-ONOO- reaction is ca. 4%, we
•-
calculate that the yield of CH3OO• formed during ONOO-
decomposition by acetone is 4/0.29 ≈ 14% of the initial
concentration of ONOO-. This implies that about 14% of the
geminate radical pairs formed during homolysis of the peroxidic
adduct escape from the solvent cage, given that the precursor
of CH3OO• is the (CH3)2C(OH)O• radical. Furthermore, since
3% out of the total 4% O2•- was formed via the slow homolysis
of CH3OONO2, it follows that about 75% of the initially formed
CH3OO• recombined with •NO2 and only about 25% underwent
self-recombination. This can be shown35 to imply that k18/k17
≈ 4, whence k18 ≈ 1.5 × 109 M-1 s-1 follows. The rate constant
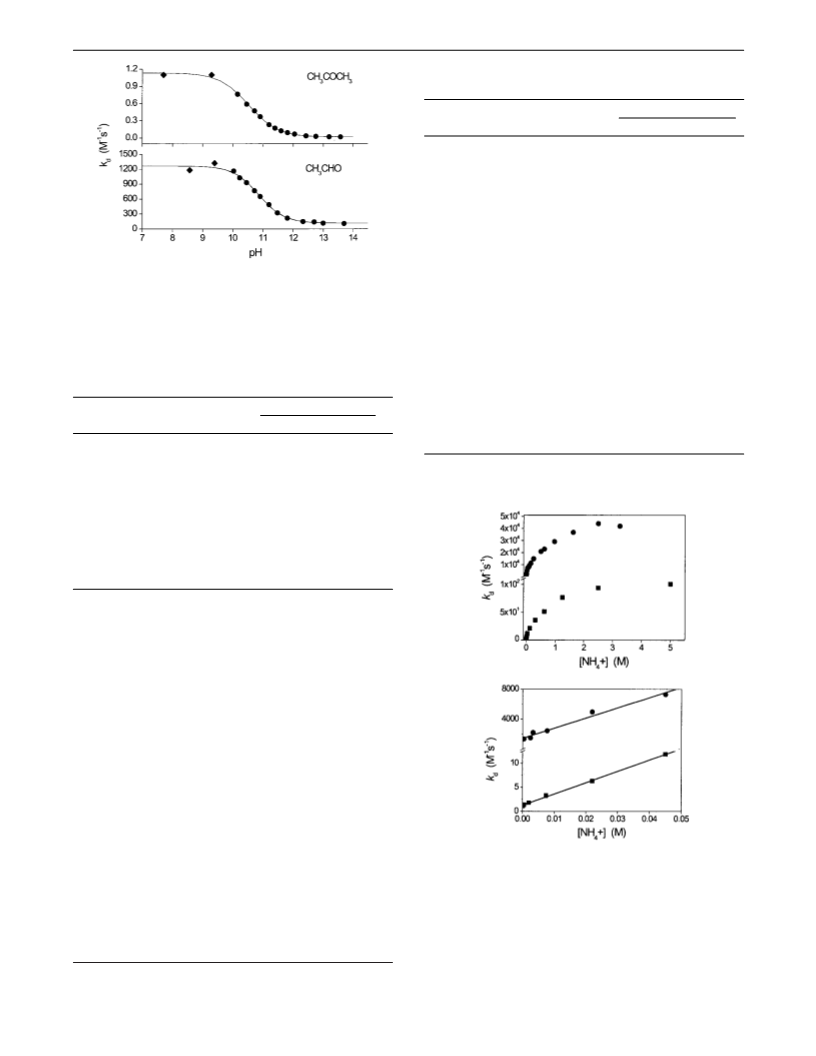
The Properties of the Carbonyl-OONO Adduct. Two
reasonable assumptions can be made for molecules of the size
of the carbonyl-OONO adducts: (i) The rate constant of their
deprotonation by OH- is constant and diffusion-controlled, with
kOH ≈ 3 × 109 M-1 s-1, and this value could hardly be wrong
by more than a factor of 2 or so. (ii) The rate of protonation of
the adduct anions by NH4+ is diffusion-controlled. As the rate
for the slow formation of O2 was measured to be 0.015 s-1
.
•-
Assuming the steady-state approximation for [CH3OO•] and [•-
NO2] formed in reaction -18 and consumed in reactions 17,
18, and 21, we derive the latter rate constant to be 2k17k-18
/
{2k17 + k18(k17/k21)1/2} ≈ (K18)-1(2k172k21)1/2. Thus, (K18)-1
≈
4.9 × 10-11 M and k-18 ≈ 0.07 s-1, where the former value is
close to that of the equilibrium constant of dissociation of
+
constant of proton self-exchange in the reaction NH3 + NH4
HOONO2 into HO2 and NO2, which is 1.4 × 10-11 M.28
•
•
+
h NH4 + NH3 is as high as 109 M-1 s-1 38
,
and the pKa’s of
The rate constant for the oxidation of ferrocyanide by CH3-
the adducts are higher than pKa(NH4+) (vide infra), kHA in the
OO• (k ) 7.1 × 103 M-1 s-1) is much lower than that for
case of NH4 should lie between 109 and 1010 M-1 s-1. We
+
therefore settle for the geometric mean of 3 × 109 M-1 s-1
,
(30) Logager, T.; Sehested, K. J. Phys. Chem. 1993, 97, 10047-10052.
(31) Graetzel, M.; Henglein, A.; Lilie, J.; Beck, G. Ber. Bunsen-Ges. Phys. Chem.
1969, 73, 646-653.
which should be accurate within better than a factor of 2. With
these two assumed rate constants, all the other rate constants
can be unravelled (Table 4). The highest rate constant obtained
is for the reverse of reaction 1, i.e., k-1 > 109 s-1, implying a
lifetime below 1 ns for the carbonyl-OONO adduct. Similar
short lifetimes have been reported for the tetrahedral intermedi-
(32) Schuchman, H.- P.; von Sonntag, C. Z. Naturforsch. 1984, 39B, 217-222.
(33) Walling, C.; Wagner, P. J. K. J. Am. Chem. Soc. 1964, 86, 3368-3375.
(34) Gilbert, B. C.; Holmes, R. G. G.; Laue, H. A. H.; Norman, R. O. C. J.
Chem. Soc., Perkin Trans. 2. 1976, 1047-1052.
(35) During the lifetime of ONOO- one can neglect reactions -18 and 21 in
comparison to 17 and 18. One then obtains for the consumption of CH3-
•
OO• and NO2 the simplified equations -[CH3OO•]/dt ) 2k17[CH3OO•]2
+ k18[CH3OO•][•NO2] and -[•NO2]/dt ) k18[CH3OO•][•NO2]. This system
of coupled differential equations is exactly solvable, and with the initial
condition [CH3OO•]o ) [•NO2]o one obtains ln F ) {r/(1 - r)} ln r, where
F is the fraction of CH3OO• consumed in reaction 17, in the event ≈1/4,
and r is defined as k18/2k17. Thus, r ≈ 2 and k18/k17 ≈ 4 is calculated.
(36) Ottolenghi, M.; Rabani, J. J. Phys. Chem. 1968, 72, 593-598.
(37) Bradic, Z.; Wilkins, R. G. J. Am. Chem. Soc. 1984, 106, 2236-2239.
(38) Meiboom, S.; Loewenstein, A.; Alexander, S. J. Chem. Phys. 1958, 29,
969-970.
9
46 J. AM. CHEM. SOC. VOL. 124, NO. 1, 2002

 Merenyi
Merenyi
 Lind
Lind
 Goldstein
Goldstein