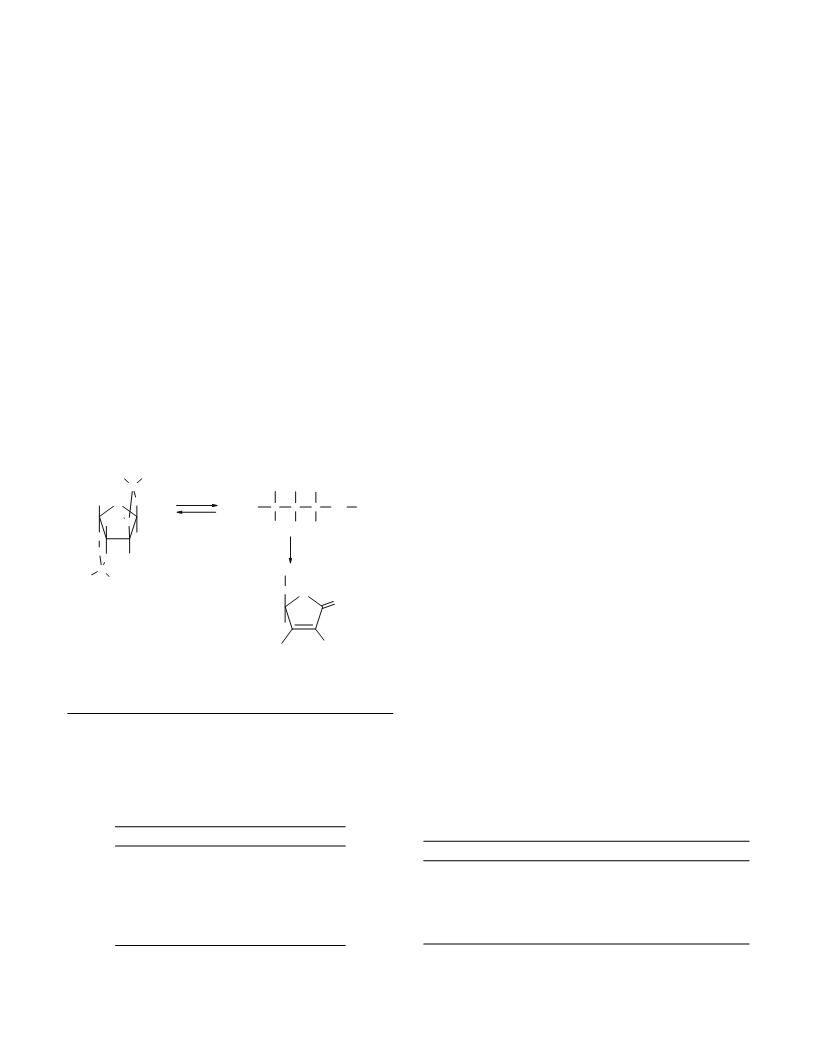Pharmaceutical Chemistry Journal
Vol. 35, No. 4, 2001
SYNTHESIS OF ASCORBIC ACID FROM DIACETONE-
2
-KETO-L-GULONIC ACID
M. A. Nadtochii and T. A. Melent’eva1
1
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 35, No. 4, pp. 54 – 56, April, 2001.
Original article submitted June 27, 2000.
The commercial production of ascorbic acid (vitamin C)
is based on the method proposed in 1934 by Reichstein and
Grussner [1]. This process, with insignificant modifications,
is still used for the synthesis of ascorbic acid (I) throughout
the world. In the final stage of this process,
diacetone-2-keto-L-gulonic acid hydrate (II) is converted
into I via 2-keto-L-gulonic acid (III) in the presence of an
acid catalyst. The mechanism of this conversion is unknown
and the optimum conditions were selected empirically.
The conversion of diacetoneketogulonic acid II into
ascorbic acid I includes two main stages. First, the protective
isopropylidene groups are detached with the formation of
ketogulonic acid III and two acetone molecules. The com-
plete hydrolysis of acid II requires two water molecules. One
of these is taken from the initial hydrate II and another is lib-
erated upon the cyclization of acid III into ascorbic acid.
We have developed a method for obtaining I from II in
toluene in the presence of concentrated hydrochloric acid as
a catalyst. Important factors determining the yield of I are the
hydrogen chloride concentration and the process duration.
An increase in the concentration of HCl leads to an increase
in the yield of I, which reaches a maximum for [HCl] =
H C
CH3
3
C
OH H OH
H O
H
O
O
2
HOCH2
C
C
C
CO COOH
III
·
H O
2
H
O
2Me CO
2
H
OH H
–
2
H C
COOH
5.75 ´ 10 mole/liter (Table 1) and then decreases, proba-
bly because of decomposition of I in the strongly acidic me-
dium. Table 2 illustrates the dependence of the yield of I on
the duration of reaction mixture heating. Here, maximum
yield is reached after heating for 5.5 h, and then the yield de-
creases somewhat.
2
O
O
H
C
CH OH
2
H C
3
CH3
HOCH
O
II
O
H
Since the conversion of II into III is reversible, removal
of acetone from the reaction zone favors a shift of the equi-
librium toward the formation of III. As a result, the yield of I
may reach up to 94%. Table 3 illustrates the dependence of
the ascorbic acid yield on the reaction duration upon acetone
removal. As seen, a maximum is reached after heating for
6 h, after which the yield of I decreases. This pattern is anal-
ogous to that observed without acetone removal, which is ev-
OH
HO
I
(
Compound III probably has a lactol structure)
1
State Research Institute of Vitamins, State Unitary Enterprise, Moscow,
Russia.
TABLE 1. Dependence of the Ascorbic Acid
Yield on the HCl Concentration (Temperature,
7
0°C; Process Duration, 5 h)
TABLE 2. Dependence of the Ascorbic Acid Yield on the Duration
of Heating at 70°C (HCl Concentration, 0.05 mole/liter)
–
2
HCl] ´ 10 , mole/liter
[
Yield, %
Process duration, h
Yield, %
4
4
5
5
5
6
.00
73
.85
77.5
78.5
81
2.0
3.5
5.0
5.5
6.5
53
.125
.425
.75
70
72.5
75
83
.125
77.5
72.5
231
0
091-150X/01/3504-0231$25.00 © 2001 Plenum Publishing Corporation

 Nadtochii
Nadtochii
 Melent'eva
Melent'eva