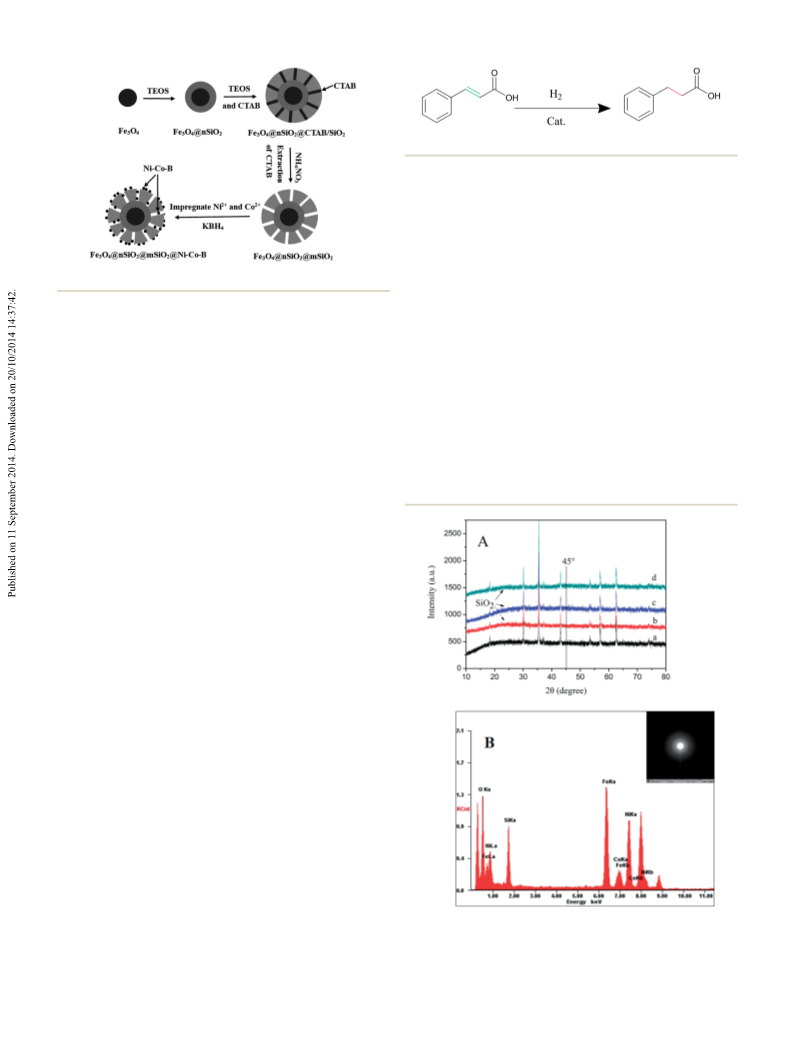
RSC Advances
Paper
Notes and references
1 Z. H. Xu, C. X. Li, X. J. Kang, D. M. Yang, P. P. Yang, Z. Y. Hou
and J. Lin, J. Phys. Chem. C, 2010, 114, 16343–16350.
2 J. P. Ge, Q. Zhang, T. R. Zhang and Y. D. Yin, Angew. Chem.,
Int. Ed., 2008, 47, 8924–8928.
3 D. G. He, X. X. He, K. M. Wang, Y. X. Zhao and Z. Zou,
Langmuir, 2013, 29, 5896–5904.
4 P. P. Yang, Z. W. Quan, Z. Y. Hou, C. X. Li, X. J. Kang,
Z. Y. Cheng and J. Lin, Biomaterials, 2009, 30, 4786–4795.
5 Y. H. Deng, Y. Cai, Z. K. Sun, J. Liu, C. Liu, J. Wei, W. Li, C. Liu,
Y. Wang and D. Y. Zhao, J. Am. Chem. Soc., 2010, 132, 8466–8473.
6 W. Li, B. L. Zhang, X. J. Li, H. P. Zhang and Q. Y. Zhang, Appl.
Catal., A, 2013, 459, 65–72.
Fig. 7 Reusability studies of Fe3O4@nSiO2@mSiO2@Ni–Co–B on the
selective hydrogenation of cinnamic acid to hydrocinnamic acid.
Reaction conditions: 3.0 g cinnamic acid, 0.1 g catalyst (20 wt%
Ni–Co–B), 200 mL tert-butanol, temperature at 373 K, initial P(H2) ¼
3.0 MPa, and reaction time 1 h.
7 H. M. Chen, S. S. Liu, H. L. Yang, Y. Mao, C. H. Deng,
X. M. Zhang and P. Y. Yang, Proteomics, 2010, 10, 930–939.
8 J. B. Zheng, Z. Q. Xia, J. J. Li, W. K. Lai, X. D. Yi, B. H. Chen,
W. P. Fang and H. L. Wan, Catal. Commun., 2012, 21, 18–21.
9 H. Liu, J. Deng and W. Li, Catal. Lett., 2010, 137, 261–266.
10 G. Y. Bai, L. J. Shi, Z. Zhao, Y. L. Wang, M. D. Qiu and
H. X. Dong, Mater. Lett., 2013, 96, 93–96.
the mSiO2 shell, which can not only benet the self-assembly of
Ni–Co–B by modulating the surface negatively charged,27 but
also promote the dispersion of Ni–Co–B nanoparticle on its
mesoporous surface. The good catalytic performance of Ni–Co–
B/mSiO2 can support this assumption, which also shows 100%
conversion and selectivity in the hydrogenation of cinnamic
acid. However, it must be separated by centrifugation, which
is not as convenient as magnetic separation of Fe3O4@
nSiO2@mSiO2@Ni–Co–B. Furthermore, we think the low BET
surface area (5.1 m2 gꢁ1) and pore volume (0.009 cm3 gꢁ1) of
Fe3O4@nSiO2 should account for the lower activity of Fe3O4@
nSiO2@Ni–Co–B, compared to Ni–Co–B. Finally, the stability of
Fe3O4@nSiO2@mSiO2@Ni–Co–B was investigated in the
hydrogenation of cinnamic acid and the results were depicted
in Fig. 7. As expected, this core–shell nanocomposite can be
easily separated by magnet and recycled ve times without
signicant loss of its initial catalytic activity with the selectivity
for hydrocinnamic acid still keeping 100%.
11 G. Y. Bai, H. X. Dong, Z. Zhao, H. L. Chu, X. Wen, C. Liu and
F. Li, RSC Adv., 2014, 4, 19800–19805.
12 G. Y. Bai, H. X. Dong, Z. Zhao, Y. L. Wang, Q. Z. Chen and
M. D. Qiu, J. Nanosci. Nanotechnol., 2013, 13, 5012–5016.
13 K. Lan and Z. X. Shan, Synth. Commun., 2007, 37, 2171–2177.
14 K. J. P. Narayana, P. Prabhakar, M. Vijayalakshmi,
Y. Venkateswarlu and P. S. J. Krishna, Pol. J. Microbiol.,
2007, 56, 191–197.
15 Y. H. Deng, D. W. Qi, C. H. Deng, X. M. Zhang and
D. Y. Zhao, J. Am. Chem. Soc., 2008, 130, 28–29.
16 C. Wang, S. Y. Tao, W. Wei, C. G. Meng, F. Y. Liu and M. Han,
J. Mater. Chem., 2010, 20, 4635–4641.
17 H. Li, J. Liu, H. X. Yang and H. Li, J. Mater. Res., 2009, 24,
3300–3307.
18 S. Z. Li, Y. Ma, X. L. Yue, Z. Cao and Z. F. Dai, New J. Chem.,
2009, 33, 2414–2418.
19 X. H. Zhu, Y. Z. Chen, F. W. Zhang, J. R. Niu, M. Xie and
J. T. Ma, RSC Adv., 2014, 4, 2509–2514.
20 J. P. Yang, F. Zhang, Y. R. Chen, S. Qian, P. Hu, W. Li,
Y. H. Deng, Y. Fang, L. Han, M. Luqmanb and D. Y. Zhao,
Chem. Commun., 2011, 47, 11618–11620.
21 P. J. Branton, K. Kaneko and N. Setoyama, Langmuir, 1996,
12, 599–600.
22 S. S. Liu, H. M. Chen, X. H. Lu, C. H. Deng, X. M. Zhang and
P. Y. Yang, Angew. Chem., Int. Ed., 2010, 49, 7557–7561.
23 Z. N. Liu, H. H. Yang, H. Zhang, C. J. Huang and L. F. Li,
Cryogenics, 2012, 52, 699–703.
24 H. F. Liu, S. F. Ji, H. Yang, H. Zhang and M. Tang, Ultrason.
Sonochem., 2014, 21, 505–512.
4. Conclusions
In conclusion, a novel magnetic mesoporous core–shell nano-
composite Fe3O4@nSiO2@mSiO2@Ni–Co–B was prepared and
showed excellent activity in the selective hydrogenation of cin-
namic acid to hydrocinnamic acid. The existence of the mSiO2
shell affords this nanocomposite larger surface area and pore
volume, making the active Ni–Co–B species highly dispersed on
its surface, and then accounting for its high activity. Meanwhile,
this nanocomposite can be easily separated by magnet aer
reaction due to its high magnetism and recycled ve times
without signicant loss of its initial catalytic activity, demon-
strating its good stability.
25 R. Xu, G. Y. Sun, Q. Y. Li, E. B. Wang and J. M. Gu, Solid State
Sci., 2010, 12, 1720–1725.
26 X. H. Zhang and L. Jiang, J. Mater. Chem., 2011, 21, 10653–10657.
27 D. W. Wang, X. M. Zhu, S. F. Lee, H. M. Chan, H. W. Li,
S. K. Kong, J. C. Yu, C. H. K. Cheng, Y. X. J. Wang and
K. C. F. Leung, J. Mater. Chem. B, 2013, 1, 2934–2942.
Acknowledgements
Financial support by the National Natural Science Foundation
of China (21376060), the Natural Science Foundation of Hebei
Province (B2014201024), and the Science Foundation of Hebei
University (3333112) are gratefully acknowledged.
44306 | RSC Adv., 2014, 4, 44302–44306
This journal is © The Royal Society of Chemistry 2014

 Liu, Xiaofang
Liu, Xiaofang
 Shi, Lingjuan
Shi, Lingjuan
 Feng, Wenhui
Feng, Wenhui
 Niu, Libo
Niu, Libo
 Liu, Chen
Liu, Chen
 Bai, Guoyi
Bai, Guoyi