Organometallics
Article
ether = 1:1), pure product was obtained as a yellow solid (27.1 mg,
1.33−1.32 (m, 4H), 0.86 (t, J = 6.9 Hz, 3H). 13C NMR (100 MHz,
1
4
8
7
1
1
7%), mp 63−65 °C. H NMR (400 MHz, CDCl ) δ 10.31 (s, 1H),
CDCl ) δ 174.5, 143.9, 139.0, 137.0, 128.8, 128.5, 128.1, 126.5, 118.5,
3
3
.50 (dd, J = 8.5, 1.6 Hz, 1H), 8.22 (dd, J = 6.5, 1.0 Hz, 1H), 7.35−
.31 (m, 1H), 7.24−7.11 (m, 5H), 6.99−6.95 (m, 1H), 3.16 (d, J =
3.3 Hz, 1H), 2.78 (d, J = 13.4 Hz, 1H), 1.98−1.91 (m, 1H), 1.62−
.53 (m, 1H), 1.28 (s, 3H), 0.95 (t, J = 7.4 Hz, 3H). 13C NMR (100
114.7, 51.1, 38.8, 32.3, 29.5, 22.7, 13.9. HRMS (positive ESI) Calcd for
C H N O (M + H) 299.1760, found 299.1758.
18
23
2
2
2-(2-Phenylcyclopropanecarboxamido)pyridine 1-oxide (3ha).
After purification by column chromatography (dichloromethane/
MHz, CDCl ) δ 175.9, 144.2, 137.1, 137.0, 130.2, 128.2, 128.1, 126.7,
acetone = 3:1), pure product was obtained as a white solid (13.2 mg,
3
1
1
18.5, 114.5, 49.3, 45.6, 32.6, 19.9, 9.1. HRMS (positive ESI) Calcd for
26%), mp 147−148 °C. H NMR (400 MHz, CDCl ) δ 10.15 (s, 1H),
3
C H N O (M + H) 285.1603, found 285.1600.
8.21−8.18 (m, 2H), 7.31−7.17 (m, 6H), 6.92−6.88 (m, 1H), 2.71 (dd,
1
7
21
2
2
2
-(2,2-Dibenzylbutanamido)pyridine 1-oxide (4ca). After purifi-
J = 16.9, 8.6 Hz, 1H), 2.29−2.23 (m, 1H), 1.92−1.87 (m, 1H), 1.49−
cation by column chromatography (dichloromethane/acetic ether =
1.44 (m, 1H). 13C NMR (100 MHz, CDCl ) δ 168.4, 144.1, 136.9,
3
:1), pure product was obtained as a white solid (7.3 mg, 10%), mp
135.8, 129.2, 128.2, 128.1, 126.8, 118.1, 114.6, 27.0, 25.0, 11.4. HRMS
(positive ESI) Calcd for C H N O (M + H) 255.1134, found
255.1130.
1
0−81 °C. H NMR (400 MHz, CDCl ) δ 10.15 (s, 1H), 8.51 (dd, J =
3
15 15
2
2
.5, 1.7 Hz, 1H), 8.16 (dd, J = 6.5, 1.1 Hz, 1H), 7.35−7.31 (m, 1H),
.22−7.13 (m, 10H), 6.97−6.93 (m, 1H), 3.23 (d, J = 13.9 Hz, 2H),
.96 (d, J = 13.9 Hz, 2H), 1.70 (q, J = 7.4 Hz, 2H), 1.17 (t, J = 7.2 Hz,
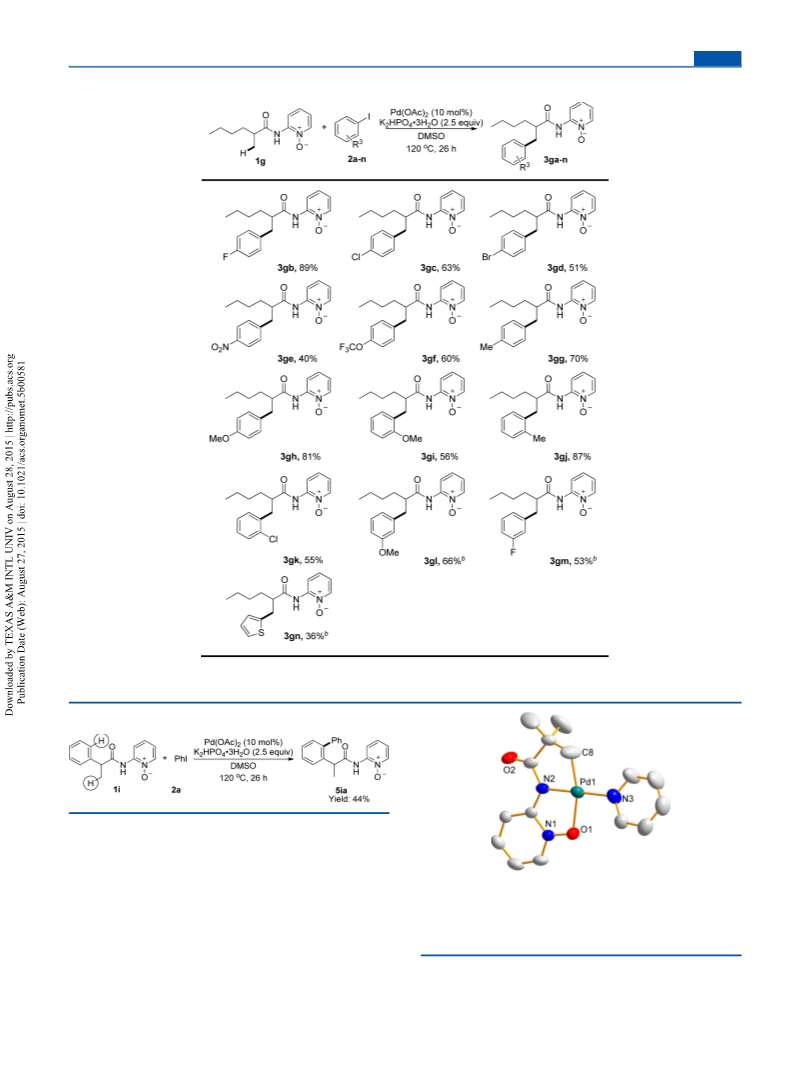
2-(2-([1,1′-Biphenyl]-2-yl)propanamido)pyridine 1-oxide (5ia).
After purification by column chromatography (aceticether/petroleum
H). 13C NMR (100 MHz, CDCl ) δ 175.3, 144.0, 137.0, 136.9, 129.9,
ether = 5:1), pure product was obtained as a pale yellow oil (28.0 mg,
3
1
28.3, 128.2, 126.7, 118.5, 114.4, 53.7, 41.6, 23.6, 8.6, 9.1. HRMS
44%). H NMR (400 MHz, CDCl ) δ 9.78 (s, 1H), 8.37 (dd, J = 8.5,
3
1.7 Hz, 1H), 8.13 (dd, J = 6.5, 1.1 Hz, 1H), 7.48 (m, 3H), 7.41−7.28
23
25
2
2
61.1913.
(m, 6H), 7.26−7.24 (m, 1H), 6.92−6.88 (m, 1H), 3.97 (q, J = 7.0 Hz,
2-(2-Methyl-3-phenylpropanamido)pyridine 1-oxide (3da). After
1H), 1.54 (d, J = 7.0 Hz, 3H). 13C NMR (100 MHz, CDCl ) δ 173.2,
3
purification by column chromatography (dichloromethane/acetic
144.2, 142.1, 140.8, 137.9, 136.9, 130.5, 129.2, 128.7, 128.4, 128.0,
127.6, 127.4, 126.9, 118.4, 114.4, 43.9, 18.5. HRMS (positive ESI)
Calcd for C H N O (M + H) 319.1447, found 319.1445.
ether = 1:1), pure product was obtained as a pale yellow solid (21.7
1
mg, 42%), mp 86−88 °C. H NMR (400 MHz, CDCl ) δ 10.01 (s,
3
20 19
2
2
1
H), 8.45 (dd, J = 8.5, 1.7 Hz, 1H), 8.21 (dd, J = 6.5, 1.0 Hz, 1H), 7.33
2-(2-(4-Fluorobenzyl)hexanamido)pyridine 1-oxide (3gb). After
(
t, J = 1.24 Hz, 1H), 7.30−7.18 (m, 5H), 6.98−6.94 (m, 1H), 3.14
purification by column chromatography (dichloromethane/acetone =
(
dd, J = 13.3, 7.0 Hz, 1H), 2.89−2.82 (m, 1H), 2.76 (dd, J = 13.4, 7.5
3:1), pure product was obtained as a pale yellow oil (56.6 mg, 89%).
13
1
Hz, 1H), 1.29 (d, J = 6.8 Hz, 3H). C NMR (100 MHz, CDCl ) δ
H NMR(400 MHz, CDCl ) δ 9.95 (s, 1H), 8.45 (dd, J = 8.5, 1.7 Hz,
3
3
1
4
2
74.9, 144.1, 138.8, 137.0, 129.0, 128.5, 128.1, 126.6, 118.6, 114.8,
4.6, 39.8, 17.5. HRMS (positive ESI) Calcd for C H N O (M + H)
1H), 8.20 (dd, J = 6.5, 1.8 Hz, 1H), 7.34−7.29 (m, 1H), 7.16−7.13
(m, 2H), 6.99−6.91 (m, 3H), 3.02 (dd, J = 13.7, 8.6 Hz, 1H), 2.81
(dd, J = 13.7, 6.2 Hz, 1H), 2.73−2.66 (m, 1H), 1.81−1.72 (m, 1H),
15
17
2
2
57.1290, found 257.1286.
-(2-Benzyl-3-phenylpropanamido)pyridine 1-oxide (4da). After
1
3
2
1.63−1.56 (m, 1H), 1.38−1.32 (m, 4H), 0.87 (t, J = 7.0 Hz, 3H). C
purification by column chromatography (dichloromethane/acetic
ether = 1:1), pure product was obtained as a white solid (8.4 mg,
NMR (100 MHz, CDCl ) δ 174.3, 161.6 (d, J
= 242.9 Hz), 143.9,
3
F−C
137.0, 134.6 (d, JF−C = 3.1 Hz), 130.3 (d, JF−C = 7.9 Hz), 128.1, 118.6,
1
19
1
8
7
2
3%), mp 115−117 °C. H NMR (400 MHz, CDCl ) δ 9.70 (s, 1H),
115.3 (d, JF−C = 21.1 Hz), 114.7, 51.1, 37.9, 32.4, 29.5, 22.6, 13.9. F
3
.39 (dd, J = 8.5, 1.7 Hz, 1H), 8.12 (dd, J = 6.5, 1.0 Hz, 1H), 7.29−
.23 (m, 5H), 7.12−7.17 (m, 6H), 6.93−6.89 (m, 1H), 3.12−3.09 (m,
NMR (376 MHz, CDCl3) δ −116.51. HRMS (positive ESI) Calcd for
C H FN O (M + H) 317.1665, found 317.1664.
18
22
2
2
13
H), 3.05−3.02 (m, 1H), 2.88 (dd, J = 13.0, 5.5 Hz, 2H). C NMR
2-(2-(4-Chlorobenzyl)hexanamido)pyridine 1-oxide (3gc). After
purification by column chromatography (dichloromethane/acetone =
(
100 MHz, CDCl ) δ 173.6, 143.7, 138.6, 137.0, 128.8, 128.6, 128.0,
3
1
26.7, 118.6, 114.7, 53.0, 38.5. HRMS (positive ESI) Calcd for
3:1), pure product was obtained as a pale yellow oil (42.1 mg, 63%).
1
C H N O (M + H) 333.1603, found 333.1601.
H NMR (400 MHz, CDCl ) δ 9.97 (s, 1H), 8.44 (dd, J = 8.5, 1.7 Hz,
2
1
21
2
2
3
2
-(2-Benzylbutanamido)pyridine 1-oxide (3ea). After purification
1
H), 8.21 (dd, J = 6.5, 1.1 Hz, 1H), 7.34−7.30 (m, 1H), 7.23−7.20
m, 2H), 7.12 (d, J = 8.4 Hz, 2H), 6.99−6.95 (m, 1H), 3.03 (dd, J =
3.6, 8.6 Hz, 1H), 2.80 (dd, J = 13.7, 6.2 Hz, 1H), 2.74−2.69 (m, 1H),
1.79−1.71 (m, 1H), 1.61−1.54 (m, 1H), 1.33−1.29 (m, 4H), 0.87 (t, J
7.0 Hz, 3H). 13C NMR (100 MHz, CDCl ) δ 174.1, 143.9, 137.4,
by column chromatography (dichloromethane/acetone = 6:1), pure
product was obtained as a yellow oil (41.7 mg, 77%). H NMR (400
MHz, CDCl ) δ 9.97 (s, 1H), 8.46 (d, J = 8.5 Hz, 1H), 8.18(d, J = 6.4
(
1
1
3
Hz, 1H), 7.31−7.29 (m, 1H), 7.25−7.18 (m, 5H), 6.95−6.91 (m, 1H),
=
3
3
1
.06 (q, J = 8.3 Hz, 1H), 2.86−2.81 (m, 1H), 2.73−2.66 (m, 1H),
1
37.0, 132.3, 130.2, 128.7, 128.2, 118.7, 114.7, 50.9, 38.0, 32.4, 29.5,
2.7, 13.9. HRMS (positive ESI) Calcd for C H ClN O (M + H)
13
.83−1.75 (m, 1H), 1.67−1.62 (m, 1H), 0.99−0.95 (m, 3H).
C
2
18
22
2
2
NMR (100 MHz, CDCl ) δ 174.3, 143.9, 139.0, 137.0, 128.8, 128.5,
3
333.1370, found 333.1368.
1
28.1, 126.5, 118.5, 114.7, 52.3, 38.4, 25.6, 11.8. HRMS (positive ESI)
2
-(2-(4-Bromobenzyl)hexanamido)pyridine 1-oxide (3gd). After
Calcd for C H N O (M + H) 271.1447, found 271.1444.
16
19
2
2
purification by column chromatography (dichloromethane/acetone =
2
-(2-Benzylpentanamido)pyridine 1-oxide (3fa). After purification
3
8
8
:1), pure product was obtained as a white solid (38.8 mg, 51%), mp
1
by column chromatography (dichloromethane/acetone = 3:1), pure
product was obtained as a pale yellow oil (43.3 mg, 76%). H NMR
6−88 °C. H NMR (400 MHz, CDCl ) δ 9.95 (s, 1H), 8.44 (dd, J =
3
1
.5, 1.7 Hz, 1H), 8.21 (dd, J = 6.4, 0.9 Hz, 1H), 7.38 (s, 1H), 7.36−
(
400 MHz, CDCl ) δ 9.93 (s, 1H), 8.45 (d, J = 8.4 Hz, 1H), 8.19 (d, J
3
7.30 (m, 2H), 7.07 (d, J = 8.3 Hz, 2H), 7.00−6.96 (m, 1H), 3.01 (dd, J
=
6.5 Hz, 1H), 7.32−7.30 (m, 1H), 7.25−7.16 (m, 5H), 6.97−6.93 (m,
=
13.6, 8.6 Hz, 1H), 2.78 (dd, J = 13.6, 6.2 Hz, 1H), 2.72−2.65 (m,
1
H), 3.06 (q, J = 8.3 Hz, 1H), 2.85−2.71 (m, 2H), 1.81−1.71 (m,
H), 1.61−1.53 (m, 1H), 1.42−1.35 (m, 2H), 0.91 (t, J = 7.2 Hz, 3H).
1
0
H), 1.81−1.71 (m, 1H), 1.62−1.54 (m, 1H), 1.38−1.28 (m, 4H),
.87 (t, J = 7.0 Hz, 3H). 13C NMR (100 MHz, CDCl ) δ 174.1, 143.9,
1
3
1
3
C NMR (100 MHz, CDCl ) δ 174.5, 143.9, 139.0, 137.0, 128.8,
3
138.0, 137.0, 131.6, 130.6, 128.2, 120.4, 118.7, 114.7, 50.8, 38.0, 32.4,
1
28.5, 128.1, 126.5, 118.5, 114.7, 50.9, 38.8, 34.7, 20.7, 14.0. HRMS
29.5, 22.7, 13.9. HRMS (positive ESI) Calcd for C H BrN O (M +
18
22
2
2
(
positive ESI) Calcd for C H N O (M + H) 285.1603, found
H) 377.0865, found 377.0842.
2-(2-(4-Nitrobenzyl)hexanamido)pyridine 1-oxide (3ge). After
purification by column chromatography (dichloromethane/acetone =
17
21
2
2
2
85.1600.
2-(2′-Benzylhexanamido)pyridine 1-oxide (3ga). Prepared accord-
ing to the general direct arylation reaction experiment procedure. After
purification by column chromatography (dichloromethane/acetone =
3:1), pure product was obtained as a pale yellow solid (26.4 mg, 40%),
1
mp 137−138 °C. H NMR (400 MHz, CDCl ) δ 9.99 (s, 1H), 8.42
3
3
:1), pure product was obtained as a pale yellow oil (50.0 mg, 83%).
(dd, J = 8.5, 1.8 Hz, 1H), 8.21 (dd, J = 6.5, 1.0 Hz, 1H), 8.14−8.11 (m,
2H), 7.38−7.32 (m, 3H), 7.02−6.98 (m, 1H), 3.17 (dd, J = 13.6, 9.0
Hz, 1H), 2.79 (dd, J = 13.7, 5.8 Hz, 1H), 2.82−2.75 (m, 1H), 1.86−
1.77 (m, 1H), 1.66−1.57 (m, 1H), 1.41−1.31 (m, 4H), 0.88 (t, J = 7.0
1
H NMR (400 MHz, CDCl ) δ 9.91 (s, 1H), 8.45 (dd, J = 8.5, 1.7 Hz,
3
1
H), 8.19 (dd, J = 6.5, 1.1 Hz, 1H), 7.33−7.28 (m, 1H), 7.25−7.16
(
m, 5H), 6.97−6.93 (m, 1H), 3.05 (q, J = 8.4 Hz, 1H), 2.83 (q, J = 6.4
Hz, 1H), 2.76−2.71 (m, 1H), 1.80−1.73 (m, 1H), 1.62−1.55 (m, 1H),
Hz, 3H). 13C NMR (100 MHz, CDCl ) δ 173.5, 146.8, 146.7, 143.7,
3
F
Organometallics XXXX, XXX, XXX−XXX

 Zhang, Shou-Kun
Zhang, Shou-Kun
 Yang, Xin-Yan
Yang, Xin-Yan
 Zhao, Xue-Mei
Zhao, Xue-Mei
 Li, Peng-Xiang
Li, Peng-Xiang
 Niu, Jun-Long
Niu, Jun-Long
 Song, Mao-Ping
Song, Mao-Ping