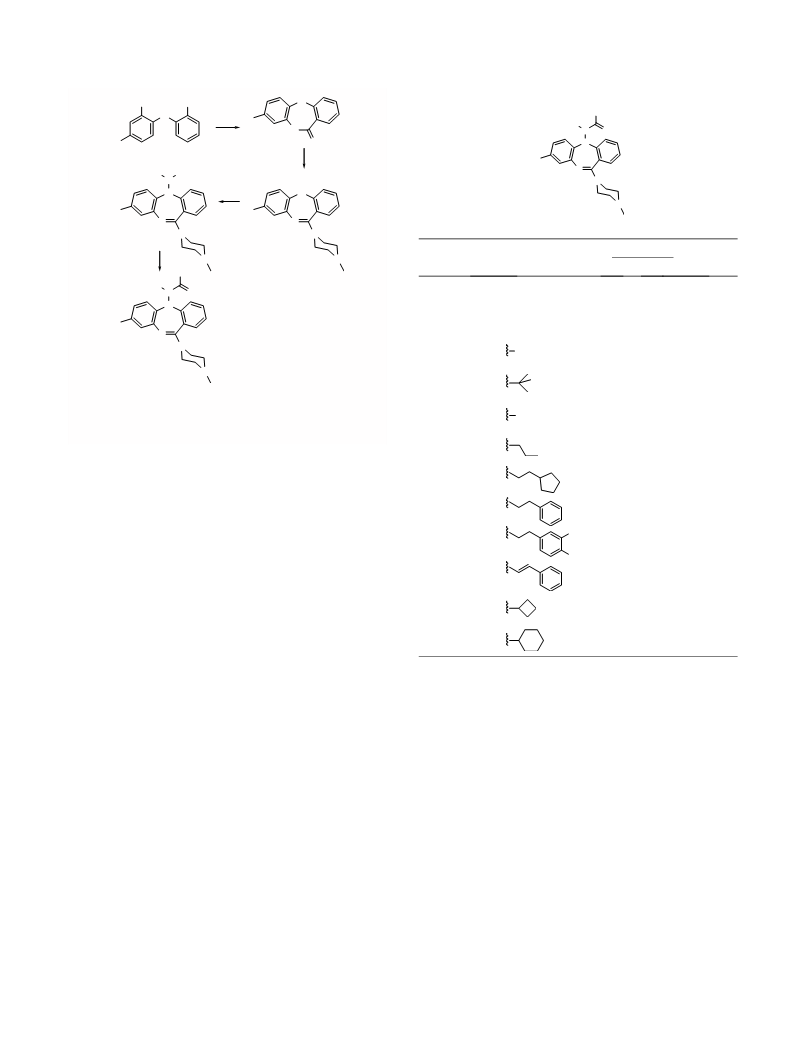
T. K. Sasikumar et al. / Bioorg. Med. Chem. Lett. 16 (2006) 4543–4547
4547
Spring, R.; Greenlee, W. J.; Smith, M.; Favreau, L.;
Fawzi, A.; Zhang, H.; Lachowicz, J. E. J. Med. Chem.
2005, 48, 680; (d) Asproni, B.; Pau, A.; Bitty, M.; Melosu,
M.; Cerri, R.; Dazzi, L.; Seu, E.; Maciocco, E.; Sanna, E.;
Busonero, F.; Talani, G.; Pusceddu, L.; Altomare, C.;
Trapani, G.; Biggio, G. J. Med. Chem. 2002, 45, 4655.
6. Baldessarini, R. J.; Frankenburg, F. R. N. Eng. J. Med.
1991, 324, 746.
7. Hippius, H. J. Clin. Psychiatry 1999, 60, 22.
8. Remington, G.; Kapur, S. Psychopharmacology 2000,
148, 3.
9. Ashby, C. R.; Wang, R. Y. Synapse 1996, 24, 349.
10. Millan, J. Pharmacol. Exp. Ther. 2000, 295, 853.
11. Squires, R. F.; Saederup, E. Neurochem. Res. 1998, 23, 1283.
12. Michel, F. J.; Trudeau, L.-E. Neuropharmacology 2000,
39, 1536.
13. Carlsson, A.; Waters, N.; Holm-Waters, S.; Tedroff, J.;
Nilsson, M.; Carlsson, M. L. Annu. Rev. Toxicol. 2001, 41,
237.
14. Kalkman, H. O.; Neumann, V.; Hoyer, D.; Tricklebank,
M. D. Br. J. Pharmacol. 1998, 124, 1550.
15. Idanpaan-Heikkila, J.; Alhava, E.; Olkinoura, M.; Palva,
P. Eur. J. Clin. Pharmacol. 1977, 11, 193.
16. Lieberman, J. A.; Johns, C. A.; Kane, J. M.; Rai, K.;
Pisciotta, A. V.; Saltz, B. L.; Howard, A. J. Clin.
Psychiatry 1988, 49, 271.
17. Alvir, J. M.; Lieberman, J. A.; Safferman, A.; Schwimmer,
J. L.; Schaaf, J. A. N. Eng. J. Med. 1993, 329, 162.
18. Buchanan, R. W. Schizophr. Bull. 1995, 57, 57.
19. Casey, D. E. J. Clin. Psychiatry 1996, 57, 40.
20. Lindstrom, L. H. Acta Psychiatr. Scand. 1988, 77, 524.
21. Liao, Y.; DeVoer, P.; Meier, E.; Wikstrom, H. J. Med.
Chem. 1997, 40, 4146.
22. Kricka, L. J.; Ledwith, A. Chem. Rev. 1974, 74, 101.
23. Hartman, W. W.; Roll, L. J. Org. Synth. 1943, 11, 418.
24. Two methods have been generally employed for the
formation of hydrazide link. The hydrazine 5 was treated
with the corresponding acid chloride in presence of TEA
followed by silica gel purification affording the hydrazide.
In the case of acids, we used EDCI or DCC coupling
strategy. (In all cases, the yield of the product was
60–80%.)
25. For experimentals: Ltk- cells stably expressing D1 and D2
receptors at a density of 4–7 pmol/mg protein were lysed in
hypotonic buffer and centrifuged at 48,000g. Membrane
pellets were frozen and stored at À80 ꢁC for use in binding
assays. Receptor affinities were determined by equilibrium
binding experiments in which bound and free radioligands
were separated by rapid filtration, and bound counts were
quantified by liquid scintillation counting. For D1 binding,
the radioligand was [3H] SCH 23390 (0.3 nM), and
nonspecific binding was defined by addition of 10 lM
unlabeled SCH 23390. For D2 binding, the radioligand
was [3H]methylspiperone (0.5 nM) and nonspecific bind-
ing was defined using 10 lM (À)-sulpride. Test com-
pounds, radioligand, and membrane homogenates
prepared from CHO cells expressing each receptor subtype
were incubated in a 200 lL volume for 1 h at room
temperature prior to filtration on GF-C plates. Competi-
tion binding data were analyzed using Graphpad Prism, in
which curves fit a one-site competition model with a Hill
Slope equal to or approximately 1. Mean Ki values from
four separate determinations are reported. The SEM was
below 15% in each case. LC–MS analysis was performed
on an Applied Biosystems API-100 mass spectrometer and
Shimadzu SCL-10A LC column: Altech platinum C18, 3
micron, 33 mm · 7 mm ID; gradient flow: 0 min—10%
CH3CN, 5 min—95% CH3CN, 7 min—95% CH3CN,
7.5 min—10% CH3CN, and 9 min—stop. Chromatogra-
phy was performed with Selecto Scientific flash silica gel,
32–63 lM.
26. A parallel synthesis method was developed for SAR
determination; see Su, J. et al., following paper.
27. Korfmacher, W. A.; Cox, K. A.; Ng, K. J.; Veals, J.;
Hsein, Y.; Wainhaus, S.; Broske, L.; Prelusky, D.;
Nomeir, A.; White, R. E. Rapid Commun. Mass Spectrom.
2001, 15, 335, Data are from pooled samples from two rats
in cassette-accelerated rapid rat protocol as described in
the above reference. Briefly, two male Sprauge–Dawley
rats were dosed orally at a dose of 10 mg/kg. Blood
samples were collected at different time points and
analyzed according to reference 27. Compound plasma
levels for individual animals used to calculate PK param-
eters were generally within 25% of average values.

 Sasikumar
Sasikumar
 Burnett
Burnett
 Zhang
Zhang
 Smith-Torhan
Smith-Torhan
 Fawzi
Fawzi
 Lachowicz
Lachowicz