1
72
E. LONGHINOTTI ET AL.
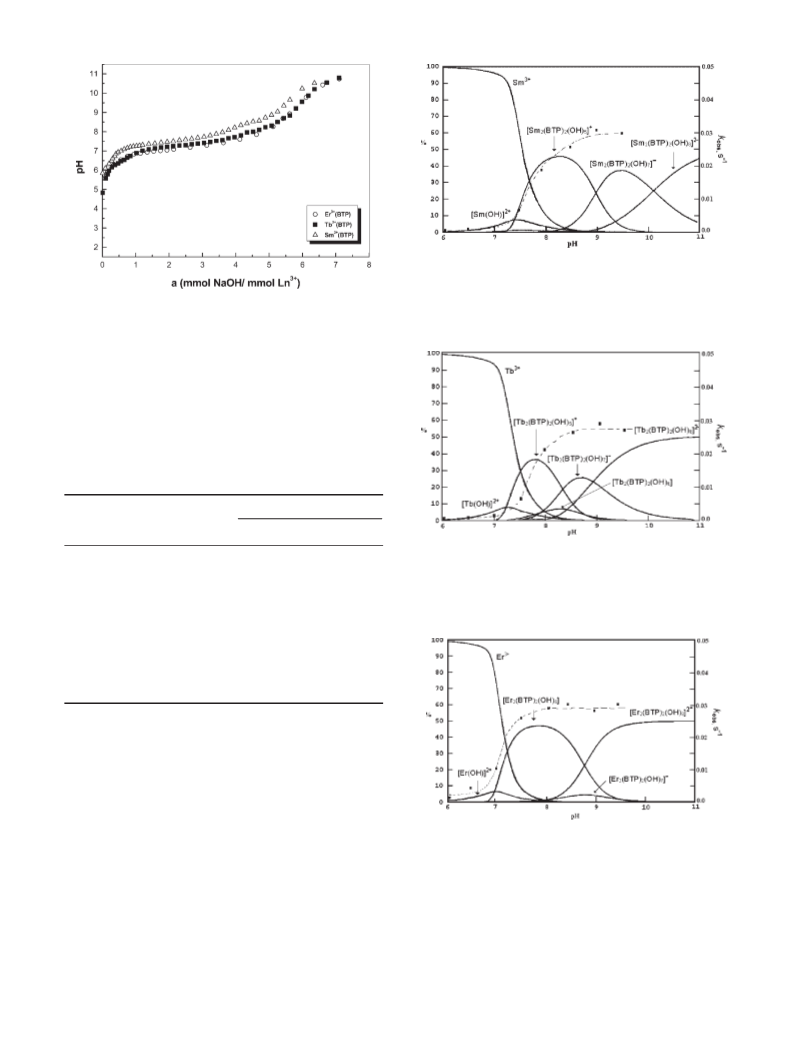
pH 9.4; above this pH, the predominant species is
[
The simple observation that the dinuclear hydroxo
3þ
2
ꢀ
Sm (BTP) (OH) ] .
2
complexes of Ln are the active species in the catalysis
of the BDNPP hydrolysis reaction is interesting from the
2
8
3
Species distribution diagrams for Tb –BTP show
þ
3
þ
similar results to those for the Sm
system. The
complex reaches a maximum
point of view of the bimetallic cooperativity presented by
1,21
þ
[
(
Tb (BTP) (OH) ]
2
some natural metalloenzymes.
dinuclear complexes in phosphate ester hydrolysis reac-
The participation of
2
5
36.5%) at pH 7.8, and at this pH there are also small
7
tions was proposed by Yatsimirsky and co-workers and
amounts of the [Tb (BTP) (OH) ] and [Tb (BTP)
2
2
2
6
2
ꢀ
ꢀ
(
OH)7] complexes. The [Tb (BTP) (OH) ] species
2
allows one of the lanthanides to promote the binding
act efficiently, while the second metal ion through as-
sociation with a nucleophilic species induces a synchro-
nic effect on the hydrolysis reaction rate. The overall
effect results in both the ability of the metal ion to
polarize P—O bonds and the placement of a nucleophilic
species in a suitable position, an effect which results
in charge, high coordination numbers and binding
directionality.
2
7
reaches its maximum at pH 8.7 with 25.6% formation,
whereas the [Tb (BTP) (OH) ] species only reaches
2
2
6
6
.9% at pH 8.3 (Fig. 5).
Figure 6 shows the formation of 47% of the [Er2
BTP) (OH) ] complex at pH 7.8. The yield of the
(
[
2
6
ꢀ
Er (BTP) (OH) ] complex is only 4.3% at pH 8.7. At
7
2
2
2ꢀ
this pH the [Tb (BTP) (OH) ] species is formed and it
2
2
7
prevails at higher pH.
The species distribution diagrams with the plot of
observed rate constant for the catalytic reaction make it
possible to identify the reactive species present in those
systems. For Sm , Tb and Er we can see that the
pH–kobs profiles are closely related to the distribution
curves for the hydroxo complexes.
The equilibrium results were used to interpret the
kinetics of the BDNPP hydrolysis reaction, where kobs
is the sum of the specific catalytic constants of each
species present in solution:
Acknowledgements
3
þ
3þ
3þ
The authors are indebted to PRONEX, CAPES and
CNPq, Brazil, for financial support of this work. The
authors are grateful to Professor A. K. Yatsimirsky for
helpful discussions.
REFERENCES
þ
kobs½Ln ðBTPÞ ꢃ
¼ k ½Ln ðBTPÞ ðOHÞ ꢃ
2
2 total
1
2
2
5
1. Wilcox DE. Chem. Rev. 1996; 96: 2435–2458.
2
3
4
. Kirby AJ. Angew. Chem. Int. Ed. Engl. 1996; 35: 707–724.
. Breslow R. Acc. Chem. Res. 1995; 28: 146–153.
. (a) Cowan JA. Curr. Opin. Chem. Biol. 2001; 5: 634–642; (b)
Bonf a´ L, Gatos M, Mancin F, Tecilla P, Tonellato U. Inorg. Chem.
þ k ½Ln ðBTPÞ ðOHÞ ꢃ
2
2
2
6
ð1Þ
ꢀ
þ k ½Ln ðBTPÞ ðOHÞ ꢃ
3
2
2
7
2
ꢀ
2
003; 42: 3943–3949; (c) Lahiri SD, Zhang G, Dunaway-Mariano
þ k ½Ln ðBTPÞ ðOHÞ ꢃ
4
2
2
8
D, Allen KN. Science 2003; 229: 2067–2071.
. Komiyama M, Matsumura K, Matsumoto Y. J. Chem. Soc., Chem.
Commun. 1992; 640–641.
5
Hydroxo complexes concentrations used in Eqn (1)
were calculated from the total metal concentrations at
each pH value. The specific rate constants of each species
6
. Moss RA, Bracken K, Zhang J. Chem. Commun. 1997; 1639–
1
7. (a) Gomes-Tagle P, Yatsimirsky AK. Inorg. Chem. 2001; 40:
640.
3
2
786; (b) Mej ´ı a-Radillo Y, Yatsimirsky AK. Inorg. Chim. Acta
003; 351: 97–106; (c) Gomes-Tagle P, Yatsimirsky AK. J. Chem.
Soc., Dalton Trans. 2001; 2663–2670.
(
k –k ) are shown in Table 3.
1 4
From the specific rate constants of each species, it is
possible to make comparisons of the catalytic effects of
all species, which have direct implications for the pro-
posed reaction mechanism. However, the specific rate
constants did not show significant variation for the
different dinuclear hydroxo species present in solution.
These results suggest that all dinuclear species are active
species, starting from the pentahydroxide species.
8. Franklin SJ. Curr. Opin. Chem. Biol., 2001; 5: 201–208.
9
. Chin J, Takasaki BK. J. Am. Chem. Soc. 1995; 117: 8582–8585.
0. Komiyama M, Kamitani J, Sumaoka J, Asanuma H. J. Chem.
Soc., Perkin Trans. 2 1998; 523–527.
11. Nakamura Y, Hashimoto S. J. Chem. Soc., Perkin Trans. 1 1996;
623–2628.
2. Morrow JR, Buttrey LA, Berbach KA. Inorg. Chem. 1992; 31:
6–20.
13. Bunton CA, Farber SJ. J. Org. Chem. 1969; 34: 767–772.
1
2
1
1
1
4. Martell AE, Motekaitis RJ. Determination and Use of Stability
Constants. VCH: New York, 1992.
1
1
5. Komiyama M, Matsumura K. J. Biochem. 1997; 122: 387–394.
6. Chin J, Takasaki BK, Hurst P. J. Am. Chem. Soc. 1996; 118: 9982–
Table 3. Rate constants for BDNPP hydrolysis by different
Ln complexes with BTP at 25 C
3þ
ꢂ
9
7. Liu T, Schneider H-J. Supramol. Chem. 2002; 14: 231–236.
983.
1
2
0 k (s
ꢀ1
)
1
18. Bruice TC, Tsubouchi A. J. Am. Chem. Soc. 1995; 117: 7399–
411.
7
3
þ
3þ
3þ
3þ
1
2
9. Komiyama M, Matsumura K. J. Biochem. 1997; 122: 387–394.
0. Smith RM, Martell AE, Motekaitis RJ. Critical Stability
Constants of Metal Complexes. Database Version 5.0, NIST
Standard Reference Database 46. US Department of Commerce,
Gaithersburg, MD, 1998.
Sm
Tb
Er
La
2.7
k1
k2
k3
k4
2.7
—
3.1
2.9
2.3
2.7
2.8
2.8
—
2.9
—
2
1. Strater N, Lipscomb WN, Klabunde T, Krebs B. Angew. Chem.
Int. Ed. Engl. 1996; 35: 2024–2055.
3.1
Copyright # 2004 John Wiley & Sons, Ltd.
J. Phys. Org. Chem. 2005; 18: 167–172

 Longhinotti, Elisane
Longhinotti, Elisane
 Domingos, Josiel B.
Domingos, Josiel B.
 Da Silva, Pedro L.F.
Da Silva, Pedro L.F.
 Szpoganicz, Bruno
Szpoganicz, Bruno
 Nome, Faruk
Nome, Faruk