N. Şener, M. Gür, M. S. Çavuş, M. Zurnaci, and İ. Şener
Vol 000
[23] Hassan, A. S.; Mady, M. F.; Awad, H. M.; Hafes, T. S.
Chinese Chem Lett 2017, 28, 388.
[24] Laikhter, A.; Behlke, M. A.; Walder, J.; Roberts, K. W.
US Patent No 9,540,515.10 Jan. 2017.
[25] Karcı, F.; Sener, N.; Yamaç, M.; Sener, I.; Demirçalı, A. Dyes
and Pigments 2009, 80, 47.
[26] Kees, K. L.; Fitzgerald, J. J.; Steiner, K. E.; Mattes, J. F.;
Mihan, B.; Tosi, T.; Mondoro, D.; McCaleb, M. L. J Med Chem 1996,
39, 3920.
[27] Kelekçi, N. G.; Yabanoğlu, S.; Küpeli, E.; Salgın, U.; Özgen,
Ö.; Uçar, G.; Yesilada, E.; Kendi, E.; Yesilada, A.; Bilgin, A. A. Bioorgan
Med Chem 2007, 15, 5775.
[28] Faidallah, H. M.; Rostom, S. A. F. Arch Pharm 2017, 350,
e1700025.
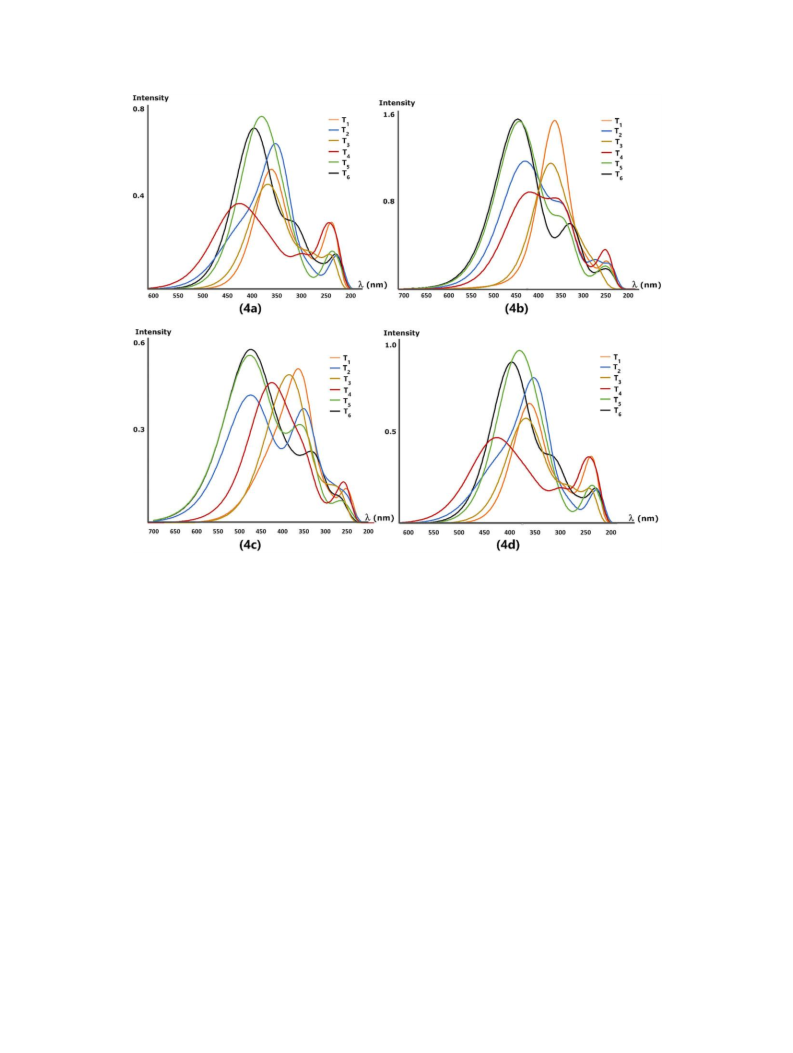
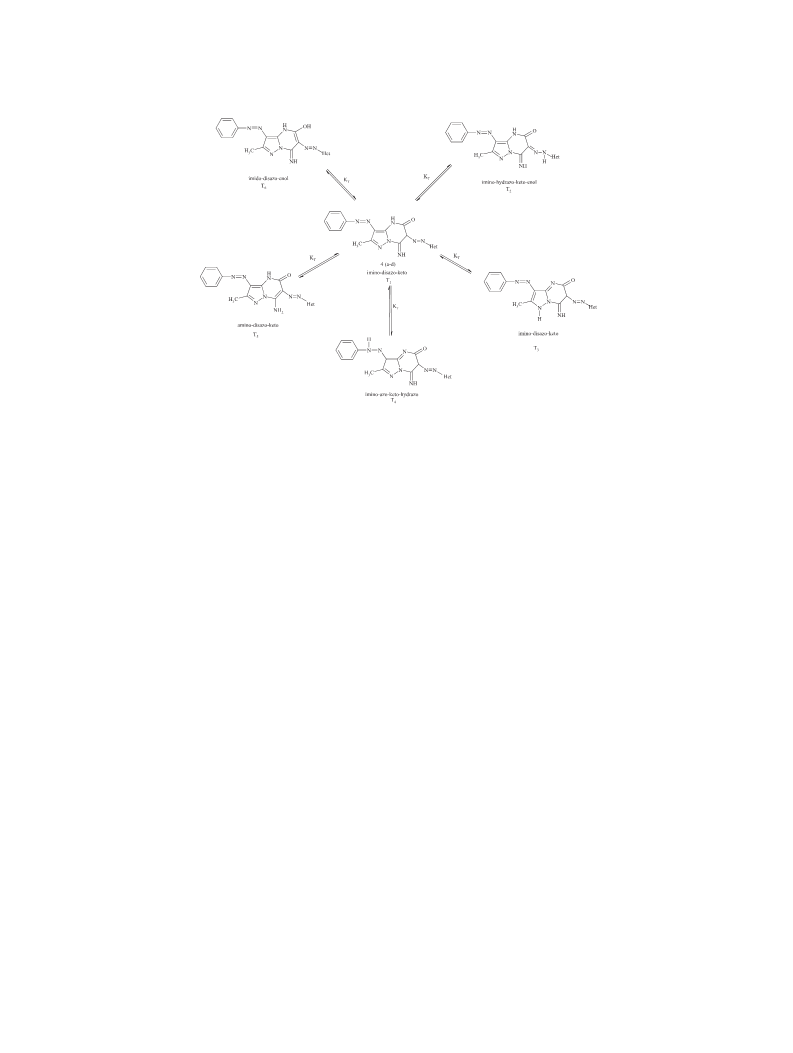
The HOMO and LUMO energies of both keto forms and
tautomeric structures were also calculated with the titled
method and basis set. FMO energy eigenvalues of the
compounds and their tautomers were used to calculate
electronic parameters such as energy band gab ΔE,
chemical hardness (η), and electronegativity (χ). The
correlation between tautomer structures and electronic
parameters of the compounds was investigated. All
calculations were performed by using the GAUSSIAN 09
[41] software package program.
[29] Malvar, D. C.; Ferreira, R. T.; Castro, R. A.; Castro, L. L.;
Freitas, A. C. C.; Costa, E. A.; Florentino, I. F.; Mafra, J. C. M.; Souza,
G. E. P.; Vanderlinde, F. A. Life Sci 2014, 95, 81.
[30] Shehry, M. F. E.; Ghorab, M. M.; Abbas, S. Y.; Fayed, E. A.;
Shedid, S. A.; Ammar, Y. A. Eur J Med Chem 2018, 143, 1463.
[31] Hernández-Vázquez, E.; Aguayo-Ortiz, R.; Ramírez-
Espinosa, J. J.; Estrada-Soto, S.; Hernández-Luis, F. Eur J Med Chem
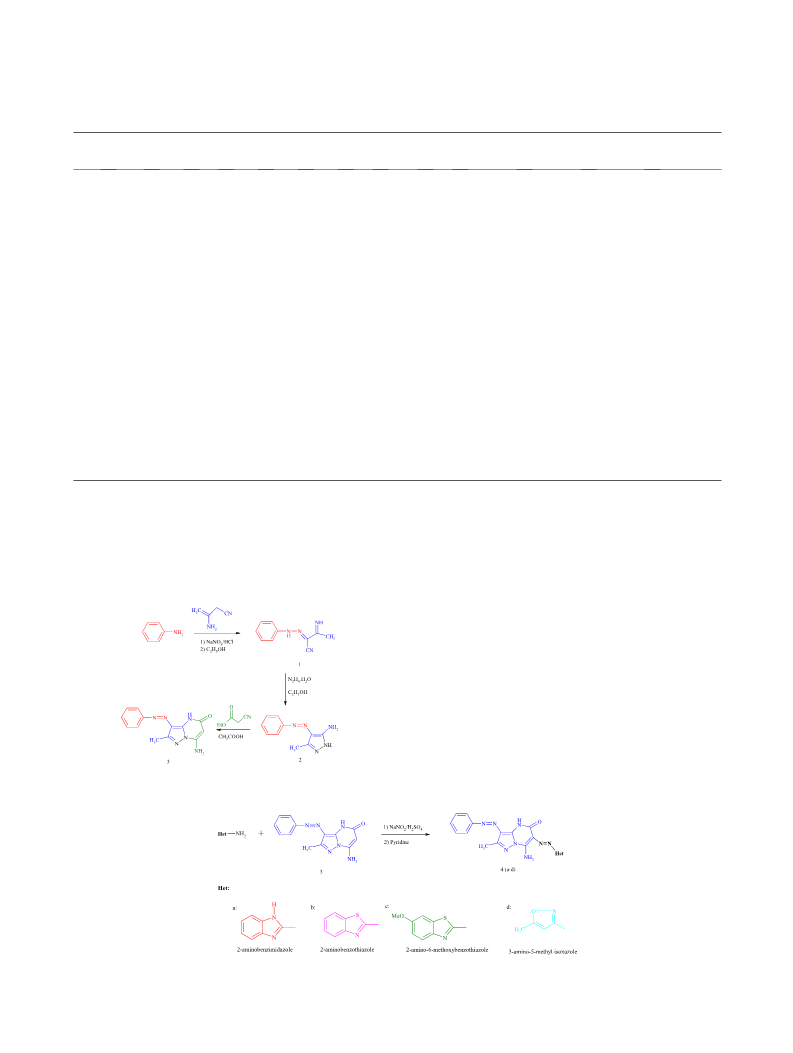
2013, 69, 10.
[32] Soliman, R.; Mokhtar, H.; Mohamed, H. F. J Pharm Sci 1983,
72, 1004.
[33] Şener, N.; Erişkin, S.; Yavuz, S.; Şener, İ. J Heterocyclic
Chem 2017, 54, 3538.
[34] Ma, M.; Johnson, K. E. J Electroanal Chem 1993, 355, 97.
[35] Elnagdi, M. H.; Sallam, M. M. M.; Fahmy, H. M.; Ibrahim,
S. A. M. Helv Chim Acta 1976, 59, 551.
[36] Elnagdi, M. H.; Elgemeie, G. E.; Abd-Elaal, F.-M. Heterocy-
cles 1985, 23, 3121.
Acknowledgment. The authors are grateful to the Scientific
Research Projects Council of Kastamonu University (KU.
BAP01/2017-31).
REFERENCES AND NOTES
[1] Rawat, A. D.; Sharma, R. S.; Karmakar, S.; Arora, L. S.;
Mishra, V. Ecotoxicol Environ Saf 2018, 148, 528.
[2] Özkınalı, S.; Çavuş, M. S.; Sakin, B. JOTCSA, Section A:
Chemistry 2018, 5, 159.
[3] Rotaru, A.; Dumitru, M. J Therm Anal Calorim 2017, 127, 21.
[4] Bakan, E.; Karci, F.; Avinç, O. Int J Eng Appl Sci 2016, 10, 8269.
[5] Kocaokutgen, H.; Gür, M.; Soylu, M. S. Lönnecke Acta
Crystallogr E 2003, E59, 1613.
[37] Hohenberg, P.; Kohn, W. Phys Rev 1964, 136, B864.
[38] Kohn, W.; Sham, L. Phys. Rev. 1965, 140, A1133.
[39] Becke, A. D. J Chem Phys 1993, 98, 1372.
[6] Geng, J.; Xu, D.; Chang, F.; Tao, T.; Huang, W. Dyes and
Pigments 2017, 137, 101.
[7] Akiyama, H.; Kawara, T.; Takada, H.; Takatsu, H.; Chigrinov,
V.; Prudnikova, E.; Kozenkov, V.; Kwok, H. Liq Cryst 2002, 29, 1321.
[8] Ledoux, I.; Zyss, J.; Barni, E.; Barolo, C.; Diulgheroff, N.;
Quagliotto, P.; Viscardi, G. Synth Met 2000, 115, 1 213.
[9] Aliabadi, A.; Eghbalian, E.; Kiani, A. Iran J. Basic Med Sci
2013, 16, 1133.
[10] Zayed, M. A.; Mohamed, G. G.; Abdullah, S. A. M.
Spectrochim Acta A 2011, 78, 1027.
[11] Kumar, K.; Chinnagiri, T.; Keshavayya, J.; Rajesh, T. N.;
Peethambar, S. K.; Ali, S.; Angadi, R. Org Chem Int 2013, 2013, 1.
[12] Maradiya, H. R. JSerbChemSoc 2002, 67, 709.
[13] Otutu, J. O. IJRRAS 2013, 15, 292.
[14] Castro, M. C. R.; Schellenberg, P.; Belsley, M.; Fonseca,
A. M. C.; Fernandes, S. S. M.; Raposo, M. M. M. Dyes and Pigments
2012, 95, 392.
[15] Hallas, G.; Choi, J. H. Dyes and Pigments 1999, 42, 249.
[16] Hallas, G.; Towns, A. D. A. Dyes and Pigments 1996, 31, 273.
[17] Xu, D.; Li, Z.; Peng, Y.; Geng, J.; Qian, H.; Huang, W. Dyes
and Pigments 2016, 133, 143.
[40] Lee, C.; Yang, W.; Parr, R. G. Phys Rev, B 1988, 37, 785.
[41] Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.;
Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Mennucci, B.;
Petersson, G. A.; Nakatsuji, H.; Caricato, M.; Li, X.; Hratchian, H. P.;
Izmaylov, A. F.; Bloino, J.; Zheng, G.; Sonnenberg, J. L.; Hada, M.;
Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima,
T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Montgomery, J. A.;
Peralta, J. E.; Ogliaro, F.; Bearpark, M.; Heyd, J. J.; Brothers, E.; Kudin,
K. N.; Staroverov, V. N.; Keith, T.; Kobayashi, R.; Normand, J.;
Raghavachari, K.; Rendell, A.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.;
Cossi, M.; Rega, N.; Millam, J. M.; Klene, M.; Knox, J. E.; Cross,
J. B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann,
R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski,
J. W.; Martin, R. L.; Morokuma, K.; Zakrzewski, V. G.; Voth, G. A.;
Salvador, P.; Dannenberg, J. J.; Dapprich, S.; Daniels, A. D.; Farkas,
O.; Foresman, J. B.; Ortiz, J. V.; Cioslowski, J.; Fox, D. J. Gaussian 09,
Revision B.01; Gaussian, Inc: Wallingford CT, 2010.
[18] Abdou, M. M. Am J Chem 2013, 3, 126.
SUPPORTING INFORMATION
[19] Ünal, A.; Eren, B.; Eren, E. J Mol Struct 2013, 1049, 303.
[20] Dostanic, J.; Mijin, D.; Uscumlic, G.; Jovanovic, D. M.; Zlatar,
M.; Loncarevic, D. Spectro Chim Acta Part A 2014, 123, 37.
[21] Özkinali, S.; Çavuş, M. S.; Ceylan, A.; Gür, M. J Mol Struct
2017, 1149, 206.
Additional supporting information may be found online
in the Supporting Information section at the end of the
article.
[22] Tsai, P. C.; Wang, J. Dyes and Pigments 2008, 76, 575.
Journal of Heterocyclic Chemistry
DOI 10.1002/jhet

 ?ener, Nesrin
?ener, Nesrin
 Gür, Mahmut
Gür, Mahmut
 ?avu?, M. Serdar
?avu?, M. Serdar
 Zurnaci, Merve
Zurnaci, Merve
 ?ener, ?zzet
?ener, ?zzet