20
BRINKEVICH et al.
5. Shadyro, O., Yurkova, I., Kisel, M., Brede, O., and
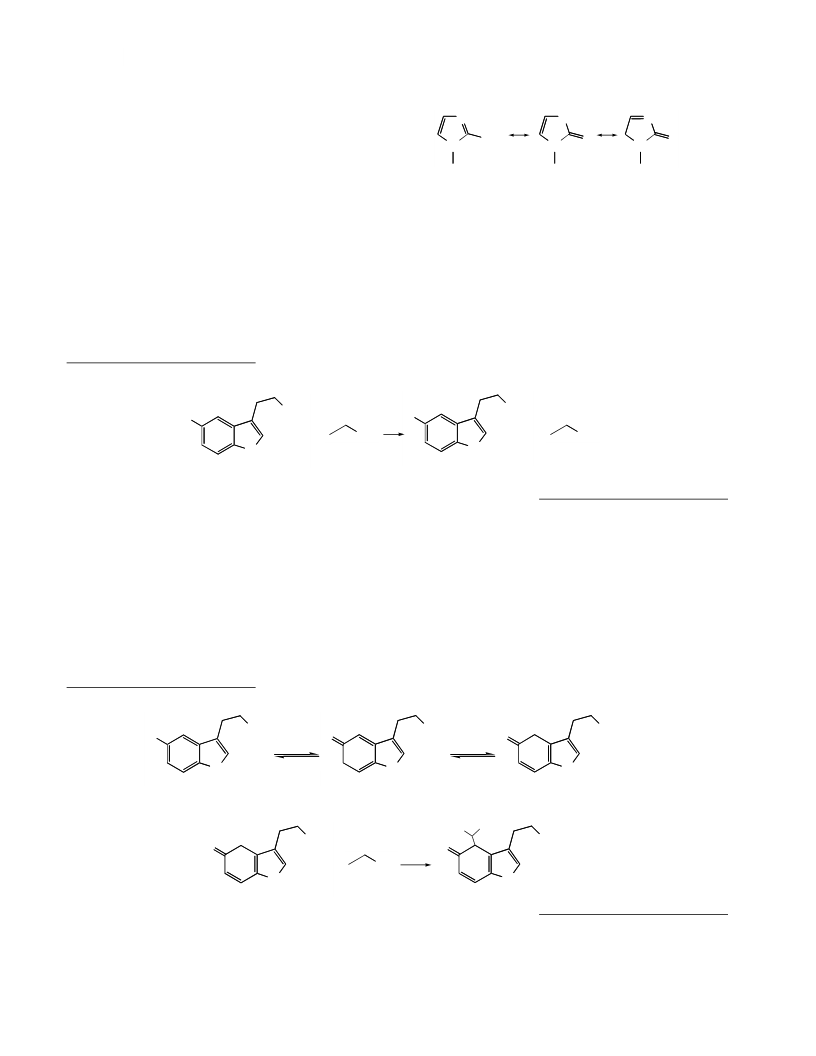
The nitrogenꢀcentered radical cations of comꢀ
Arnhold, J., Free Radical Biol. Med, 2004, vol. 36,
no. 12, p. 1612.
pounds (V) and (VI) are considerably stabilized due to
the delocalization of an unpaired electron over the
aromatic system, which creates the possibility of the
formation of adducts with
Indeed, compounds with the molecular weights
M = Madditive + M ꢀhydroxyethyl radical and M = Madditive
6. Shadyro, O.I., Sosnovskaya, A.A., and Vrublevskaya, O.N.,
Int. J. Radiat. Biol., 2003, vol. 79, no. 4, p. 269.
α
ꢀhydroxyethyl radicals.
7. Edimecheva, I.P., Kisel, M.A., Shadyro, O.I.,
+
Kazem, K., Murase, H., and Kagiya, T., J. Radiat. Res.
,
α
2M ꢀhydroxyethyl radical are formed in insignificant yields as
2005, vol. 46, no. 3, p. 319.
α
the molecular products of the radiolysis and peroxideꢀ
induced transformations of harmine (V) and harman
(VI), respectively, in deaerated ethanol (Figs. 2c, 2d).
8. Shadyro, O.I. and Kisel’, R.M., High Energy Chem.
,
2007, vol. 41, no. 5, p. 318.
9. Yurkova, I., Kisel, M., Arnhold, J., and Shadyro, O.,
The ratio between the yields of these adducts at
D =
Chem. Phys. Lipids, 2005, vol. 134, no. 1, p. 41.
2 kGy is 3 : 1 assumed that the sensitivity of a mass
spectrometer to the substances is the same.
10. Brinkevich, S.D., Sverdlov, R.L., Lagutin, P.Yu., and
Shadyro, O.I., High Energy Chem., 2011, vol. 45, no. 5,
p. 380.
CONCLUSIONS
11. Ksendzova, G.A., Sorokin, V.L., Edimecheva, I.P., and
Shadyro, O.I., Free Radical Res., 2004, vol. 38, no. 11,
p. 1183.
The above set of experimental and theoretically
calculated data suggests that harmine (V), harman
(VI), and harmaline (VII) oxidize
radicals to suppress their recombination. In this case,
an increase in the oxidizing properties in this series of
ꢀcarboline alkaloids is observed. The main molecular
12. Dominique, B.ꢀR., Toxicology, 2010, vol. 278, no. 1,
α
ꢀhydroxyethyl
p. 55.
13. Rihui Cao, Wenlie Peng, Zihou Wang, and Anlong Xu,
Curr. Med. Chem., 2007, vol. 14, no. 4, p. 479.
β
product of the freeꢀradical transformations of harmaꢀ
line (VII) induced by radiolysis or peroxide thermolyꢀ
sis in deaerated ethanol is its dimer; in the case of
harmine (V) and harman (VI), the addition products
14. Kahmann, C., Wunderlich, G., Freudenberg, R.,
Zophel, K., Oehme, L., and Kotzerke, J., Int. J. Radiat.
Biol., 2010, vol. 86, no. 10, p. 811.
15. Brinkevich, S.D. and Shadyro, O.I., High Energy
Chem., 2011, vol. 45, no. 2, p. 93.
of
α
ꢀhydroxyethyl radicals are formed. The possibility
of the regeneration of harmine (V) and harman (VI)
16. Freeman, G.R., Radiation Chemistry of Ethanol: A
Review of Data on Yields, Reaction Rate Parameters, and
Spectral Properties of Transients, Washington: NBS,
1974.
upon the interaction of additives with
radicals is noted.
α
ꢀhydroxyethyl
Serotonin (III) can suppress the formation of ethꢀ
anol radiolysis products due to the reduction and addiꢀ
17. Hryntsevich, I.B. and Shadyro, O.I., Bioorg. Med.
Chem. Lett., 2005, vol. 15, no. 19, p. 4252.
tion of ꢀhydroxyethyl radicals. We found that pyrrole
α
(I), indole (II), melatonin (IV), imidazole (VIII), 1ꢀ
methylimidazole (IX), and 2ꢀmercaptoꢀ1ꢀmethylimiꢀ
18. Brinkevich, S.D., Samovich, S.D., and Shadyro, O.I.,
High Energy Chem., 2011, vol. 45, no. 6, p. 532.
dazole (X) exhibit low reactivity toward
ethyl radicals.
α
ꢀhydroxyꢀ
19. Pikaev, A.K., Sovremennaya radiatsionnaya khimiya.
Radioliz gazov i zhidkostei (Moder Radiation Chemisꢀ
try: Radiolysis of Gases and Liquids), Moscow: Nauka,
1986.
REFERENCES
20. Duplatre, G. and Jonah, C.D., Radiat. Phys. Chem.
1984, vol. 24, p. 557.
,
1. Halliwell, B. and Gutteridge, J.M.C., Free Radicals [n
Biology snd Medicine. Oxford: Oxford University Press,
1999.
2. Von Sonntag, C., The Chemical Basis of Radiation Biolꢀ
ogy. London: Taylor and Francis, 1987.
3. Von Sonntag, C., FreeꢀRadicalꢀInduced DNA Damage
and Its Repair, Berlin: Springer, 2006.
4. Edimecheva, I.P., Kisel, M.A., Shadyro, O.I., Vlasov, A.P.,
and Yurkova, I.L., Int. J. Radiat. Biol., 1997, vol. 71,
no. 5, p. 555.
21. Greenstock, C.L., Ng, M., and Hunt, J.W., Adv. Chem.
Ser., 1968, vol. 81, p. 397.
22. Rao, P.S., Simic, M., and Hayon, E.J., Phys. Chem.
1975, vol. 79, p. 1260.
,
23. Vladimirov, V.G., Krasil’nikov, I.I., and Arapov, O.V.,
Radioprotektory: struktura i funktsii (Radioprotectors:
Structure and Functions), Kiev: Naukova Dumka,
1989.
HIGH ENERGY CHEMISTRY
Vol. 47
No. 1
2013

 Brinkevich
Brinkevich
 Sverdlov
Sverdlov
 Shadyro
Shadyro