V. N. Povalishev et al. / Bioorg. Med. Chem. Lett. 16 (2006) 1236–1239
1239
11. Shadyro, O. I.; Yurkova, I. L.; Kisel, M. A.; Brede, O.;
Arnhold, J. Free Radical Biol. Med. 2004, 36, 1612.
12. Shadyro, O. I.; Sosnovskaya, A. A.; Edimecheva, I. P.;
Grintsevich, I. B.; Lagutin, P. Yu.; Alekseev, A. V.;
Kazem, K. Free Radical Res. 2005, 39, 713.
points to the presence of a carbonyl group in the above-
mentioned compounds as a factor increasing significant-
ly the ability to oxidize a-hydroxyethyl radicals by reac-
tion (11):
13. Shadyro, O. I.; Edimecheva, I. P.; Glushonok, G. K.;
Ostrovskaya, N. I.; Polozov, G. I.; Murase, H.; Kagiya, T.
Free Radical Res. 2003, 37, 1087.
14. Lau, P. T. S.; Kestner, M. J. Org. Chem. 1968, 33, 4426.
15. Maslovskaya, L. A.; Petrikevich, D. K.; Timoschuk, V.
A.; Shadyro, O. I. Zh. Obshch. Khim. 1996, 66, 1899, in
Russian.
16. Greene, T. W.; Wuts, P. G. M. Protective Groups in
Organic Synthesis; John Wiley and Sons: New York, 1980,
158–163.
RO
RO
S
S
.
+
CH3CHOH
OH +
CH3CHO
O
ð11Þ
O
O
R = H, CH 3
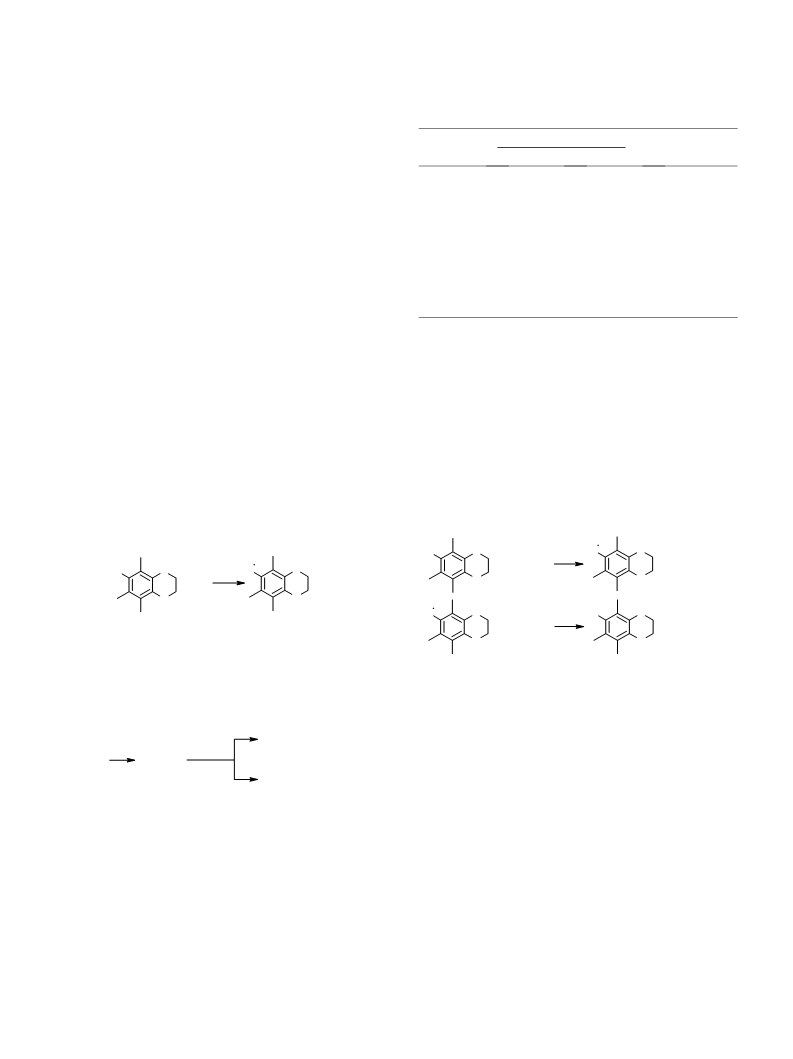
The ability of carbonyl-containing compounds to oxi-
dize various radicals formed from alcohols has been
shown earlier.26
17. Compound I: 1H NMR (100 MHz, CDCl3) d 4.40–4.26 (m,
CH2, OH), 3.20–3.00 (m, CH2), 2.15 (s, 3CH3), MS (m/z, I%)
210 (M+, 89), 195 (MꢀCH3, 5), 182 (MꢀCO, 6), 154 (100),
mp 104 °C. Compound II: 1H NMR (100 MHz, CDCl3) d
4.30–4.00 (m, CH), 3.05 (s, OH), 2.95 (d, J = 1.7 Hz, CH2),
2.15 (s, 3CH3), 1.47 (d, J = 6.2, CH3), MS (m/z, I%) 224
(M+, 60), 195 (27), 183 (17), 154 (100), mp 88 °C.
On confronting the data obtained in radiolysis studies of
hexane and ethanol, the following conclusion can be
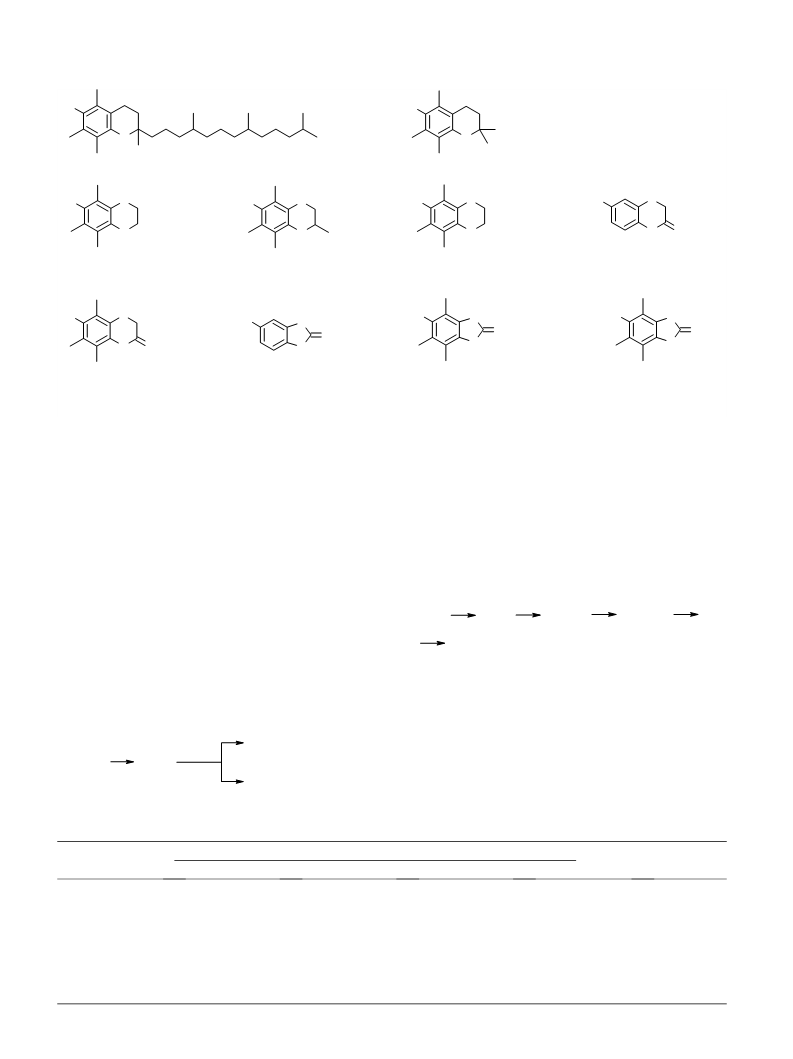
made: analogues of a-tocopherol, in particular com-
pounds of types IV–VIII, are unique agents regulating
free-radical reactions of various types. Thus, the pres-
ence of a hydroxyl group in the structure imparts to such
agent the ability to reduce organic radicals to the initial
molecules. The presence of a carbonyl group makes an
agent capable of oxidizing alcohol radicals. As a rule,
substances having such properties can block fragmenta-
tion reactions of a-hydroxyl-containing radicals occur-
ring in biologically important molecules. Taking into
account the convenience of preparative methods for
compounds of type (IV–VII), as well as the presence
of pharmacological activity,27 they may be of interest
as potential radioprotectors, medicinal drug products,
and industrial antioxidants.
1
Compound III: H NMR (100 MHz, CDCl3) d 4.39–4.29
(m, CH2), 3.63 (s, CH3O), 3.19–3.05 (m, CH2), 2.14 (s,
3CH3), MS (m/z, I%) 224 (M+, 100), 209 (MꢀCH3, 79), mp
49 °C. Compound IV: 1H NMR (100 MHz, CDCl3) d 7.00–
6.60 (m, 3H), 3.45 (s, CH2), MS (m/z, I%) 182 (M+, 54), 154
(MꢀCO, 75), 153 (100), mp 170 °C. Compound V: 1H
NMR (100 MHz, CDCl3) d 4.60 (br, OH), 3.40 (s, CH2),
2.30–2.10 (m, 3CH3), MS (m/z, I%) 224 (M+, 75), 209
(MꢀCH3, 1.7), 196 (MꢀCO, 100), mp 199 °C. Compound
VI: 1H NMR (100 MHz, CDCl3) d 7.25 (s, OH), 7.20–6.60
(m, 3H), MS (m/z, I%) 168 (M+, 100), 140 (MꢀCO, 29), 112
(Mꢀ2CO, 91), mp 174 °C. Compound VII: 1H NMR
(100 MHz, CDCl3) d 4.70 (br, OH), 2.40–2.10 (m, 3CH3),
MS (m/z, I%) 210 (M+, 78), 182 (MꢀCO, 13), 154 (Mꢀ2CO,
100), mp 159 °C. Compound Vlll: 1H NMR (100 MHz,
CDCl3) d 3.67 (s, CH3O), 2.30–2.15 (m, 3CH3), MS (m/z,
I%) (M+, 100), mp 83 °C.
References and notes
1. Halliwell, B.; Gutteridge, J. M. C. Free Radicals in Biology
and Medicine, 3rd ed.; Clarendon Press: Oxford, 1999.
2. Burton, G. W.; Doba, T.; Gabe, E. G.; Hughes, L.; Lee,
F. L.; Prasad, L.; Ingold, K. U. J. Am. Chem. Soc. 1985,
107, 7053.
3. Burton, G. W.; Ingold, K. U. Acc. Chem. Res. 1986, 19, 194.
4. Hussain, H. H.; Babic, G.; Durst, T.; Wright, J. S.;
Flueraru, M.; Chichirau, A.; Chepelev, L. L. J. Org.
Chem. 2003, 68, 7023.
5. Davies, M. J.; Forni, L. G.; Willson, R. L. Biochem. J.
1988, 255, 513.
6. Kapoor, S.; Mukherjee, T.; Kagiya, T. V.; Nair, C. K. K.
J. Radiat. Res. J. 2002, 43, 99.
7. Muller, S. N.; Batra, R.; Senn, M.; Giese, B.; Kisel, M. A.;
Shadyro, O. I. J. Am. Chem. Soc. 1997, 119, 2795.
8. Edimecheva, I. P.; Kisel, M. A.; Shadyro, O. I.; Vlasov, A.
P.; Yurkova, I. L. Int. J. Rad. Biol. 1997, 71, 555.
9. Shadyro, O. I.; Yurkova, I. L.; Kisel, M. A. Int. J. Rad.
Biol. 2002, 78, 211.
10. Shadyro, O. I. In Free Radicals in Biology and Environ-
ment; Minisci, F., Ed.; Academic: Netherlands, 1997; pp
317–329.
19. Schmidt, M. W.; Baldridge, K. K.; Boatz, J. A.; Elbert, S.
T.; Gordon, M. S.; Jensen, J. J.; Koseki, S.; Matsunaga,
N.; Nguyen, K. A.; Su, S.; Windus, T. L.; Dupuis, M.;
Montgomery, J. A. J. Comput. Chem. 1993, 14, 1347.
20. Wright, J. S.; Johnson, E. R.; DiLabio, G. A. J. Am.
Chem. Soc. 2001, 123, 1173.
21. Shinsaka, K.; Shida, S. Bull. Chem. Soc. Jpn. 1970, 43,
3728.
22. Saraeva, V. V. Radiolysis of hydrocarbons in liquid phase.
Publ. of Moscow University: Moscow, 1986, in Russian.
23. Kunjappu, I. T.; Rao, K. N. Radiat. Phys. Chem. 1979, 13,
97.
24. Kennerly, G. W.; Patterson, W. L. Ind. Eng. Chem. 1956,
48, 1917.
25. Freeman, G. R. Radiation chemistry of ethanol. Wash.:
NBS, 1974.
26. Oyama, M. A. J. Org. Chem. 1965, 30, 2429.
27. Shadyro, O. I.; Timoschuk, V. A.; Polozov, G. I.;
Povalishev, V. N.; Andreeva, O. T.; Zhelobkovich, V. E.
Khim. Pharm. Zh. 1999, 33, 25, in Russian.

 Povalishev
Povalishev
 Polozov
Polozov
 Shadyro
Shadyro