Journal of the American Chemical Society
Page 4 of 5
3. (a) Lifschitz, A. M.; Young, R. M.; Mendez-Arroyo, J.; Stern, C.
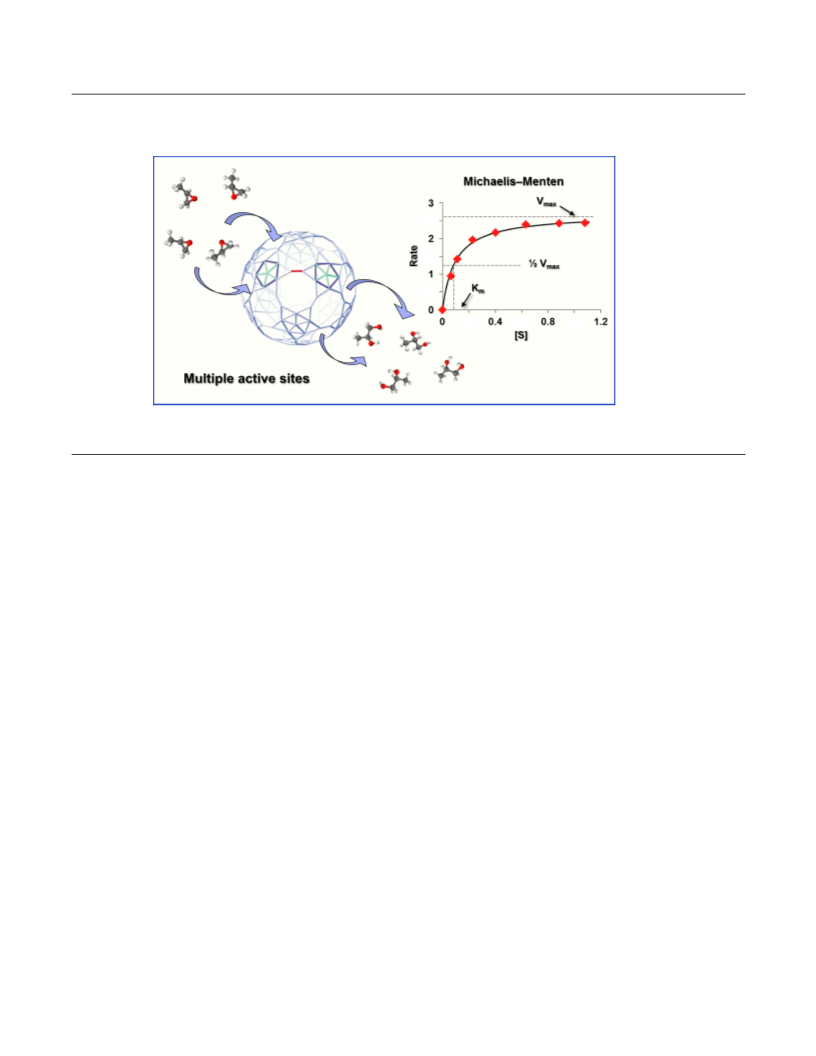
numbers of active sites should increase the rate accelera-
tion at Vmax (kcat / kuncat) by a factor of 2.6, from 16,000 to
43,200, far short of the observed value of 182,800.
Notably, however, the decrease from 23 to 12 acetate
L.; McGuirk, C. M.; Wasielewski, M. R.; Mirkin, C. A. Nat. Commun.
2015, 6, 6541; (b) Preston, D.; Fox-Charles, A.; Lo, W. K. C.;
Crowley, J. D. Chem. Commun. 2015, 51, 9042-9045; (c) Kremer, C.;
Luetzen, A. Chem. - Eur. J. 2013, 19, 6162-6196.
4. (a) Hastings, C. J.; Pluth, M. D.; Bergman, R. G.; Raymond, K.
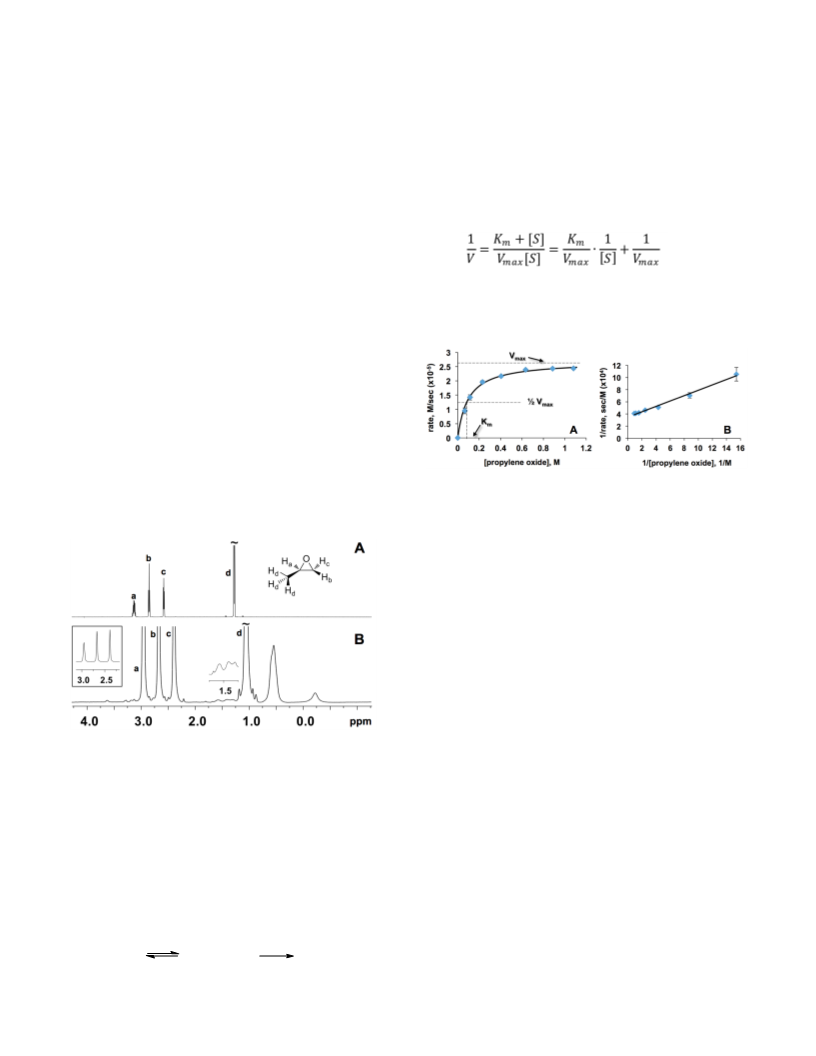
N. J. Am. Chem. Soc. 2010, 132, 6938-6940; (b) Marinescu, L. G.;
Bols, M. Angew. Chem. Int. Ed. 2006, 45, 4590-4593; (c) Trainor, G.
L.; Breslow, R. J. Am. Chem. Soc. 1981, 103, 154-158.
1
2
3
4
5
6
7
8
ligands (to give 18 active MoV sites) considerably in-
2
creases the space available for substrate molecules inside
the capsule. As result, the steady-state number of PO
“guests” inside the capsule at saturation nearly doubles
from 5 ± 0.5 (23 acetate ligands), to 9 ± 0.5 (12 acetate
ligands). Hence, not only are more MoV2 active sites pre-
sent, but the concentration of encapsulated substrate in-
creases as well. This increases the effective molarity of
the reaction, thereby amplifying the rate acceleraton per
5. Horiuchi, S.; Murase, T.; Fujita, M. Angew. Chem., Int. Ed. 2012,
51, 12029-12031.
9
6. This is emphasized in the "Future Directions" section of a 2011
review by Mirkin (ref. 1g), and in the "Conclusions and Outlook"
section of a 2015 review by Brown, Troste, Bergman and Raymond
(ref. 1b). For examples, see: (a) Kohyama, Y.; Murase, T.; Fujita, M.
J. Am. Chem. Soc. 2014, 136, 2966-2969; (b) Metherell, A. J.; Ward,
M. D., Chem. Commun. 2014, 50, 6330-6332; (c) Lee, S. J.; Cho, S.-
H.; Mulfort, K. L.; Tiede, D. M.; Hupp, J. T.; Nguyen, S. T. J. Am.
Chem. Soc. 2008, 130, 16828-16829; (d) Brisig, B.; Sanders, J. K. M.;
Otto, S. Angew. Chem., Int. Ed. 2003, 42, 1270-1273; (e) Nakash, M.;
Clyde-Watson, Z.; Feeder, N.; Davies, J. E.; Teat, S. J.; Sanders, J. K.
M. J. Am. Chem. Soc. 2000, 122, 5286-5293; (f) Mackay, L. G.;
Wylie, R. S.; Sanders J. K. M. J. Am. Chem. Soc. 1994, 116, 3141-
3142; (g) Walter, C. J.; Anderson, H. L.; Sanders, J. K. M. Chem.
Commun. 1993, 458-460.
7. (a) A. Müller, P. Gouzerh, Chem. Eur. J. 2014, 20, 4862– 4873; (b)
A. Müller, P. Gouzerh, Chem. Soc. Rev. 2012, 41, 7431- 7463.
8. (a) Johnson, A. M.; Wiley, C. A.; Young, M. C.; Zhang, X.;
Lyon, Y.; Julian, R. R.; Hooley, R. J. Angew. Chem. Int. Ed. 2015, 54,
5641-5645; (b) Jayamurugan, G.; Roberts, D. A.; Ronson, T. K.;
Nitschke, J. R. Angew. Chem., Int. Ed. 2015, 54, 7539-7543; (c) Gütz,
C.; Hovorka, R.; Klein, C.; Jiang, Q.-Q.; Bannwarth, C.; Engeser, M.;
Schmuck, C.; Assenmacher, W.; Mader, W.; Topić, F.; Rissanen, K.;
Grimme, S.; Lützen, A. Angew. Chem. Int. Ed. 2014, 53, 1693-1698;
(d) Bruns, C. J.; Fujita, D.; Hoshino, M.; Sato, S.; Stoddart, J. F.;
Fujita, M. J. Am. Chem. Soc. 2014, 136, 12027-12034; (e) Zhao, L.;
Ghosh, K.; Zheng, Y.-R.; Stang, P. J. J. Org. Chem. 2009, 74, 8516-
8521; (f) Sato, S.; Iida, J.; Suzuki, K.; Kawano, M.; Ozeki, T.; Fujita,
M. Science 2006, 313, 1273-1276.
9. (a) Grego, A.; Mueller, A.; Weinstock, I. A. Angew. Chem., Int.
Ed. 2013, 52, 8358-8362; (b) Petina, O.; Rehder, D.; Haupt, E. T. K.;
Grego, A.; Weinstock, I. A.; Merca, A.; Boegge, H.; Szakacs, J.;
Mueller, A. Angew. Chem., Int. Ed. 2011, 50, 410-414.
10. Ziv, A.; Grego, A.; Kopilevich, S.; Zeiri, L.; Miro, P.; Bo, C.;
Muller, A.; Weinstock, I. A. J. Am. Chem. Soc. 2009, 131, 6380-6382.
11. Kopilevich, S.; Gil, A.; Garcia-Ratés, M.; Bonet-Ávalos, J.; Bo,
C.; Müller, A.; Weinstock, I. A. J. Am. Chem. Soc. 2012, 134, 13082-
13088.
12. H-1 NMR data showed 4.5 ± 0.5 equiv. of PO in the capsules
with 23 acetate ligands and 21 propanoate ligands (entries 1 and 3),
and 5 ± 1 equiv in the capsule with 29 propanoate ligands (entry 4). At
small [PO], reaction by the 12-acetate capsule is too rapid for reliable
quantification, but the number of equiv. of encapsulated PO is < 7.
13. Kobayashi, S.; Manabe, K. Acc. Chem. Res. 2002, 35, 209-217.
14. (a) Dydio, P.; Detz, R. J.; Reek, J. N. H. J. Am. Chem. Soc.
2013, 135, 10817-10828; (b) Pluth, M. D.; Bergman, R. G.; Raymond,
K. N. Acc. Chem. Res. 2009, 42, 1650-1659; (c) Ortega-Caballero, F.;
Rousseau, C.; Christensen, B.; Petersen, T. E.; Bols, M. J. Am. Chem.
Soc. 2005, 127, 3238-3239.
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54
55
56
57
58
59
60
MoV active site. This cage effect increases the rate ac-
2
celeration from 43,200 (based on the larger number of
MoV2 sites) to an enzyme-like4c value of 182,000.
The present findings demonstrate how the structural
framework of a porous capsule can stabilize reactive
metal fragments in water, and through encapsulation,
bring numerous guest substrates in close proximity to
multiple active sites. We further show how the removal
of endohedrally bound blocking ligands increases turno-
ver rates by providing a larger number of reactive (lig-
and-free) metal sites, whose activity, in turn, is simulta-
neously amplified by a larger steady-state concentration
of encapsulated substrates, more of which now occupy
the larger interior space of the ligand-depleted cage.
ASSOCIATED CONTENT
Supporting Information.
Materials and methods, and spectra. This material is availa-
AUTHOR INFORMATION
Corresponding Author
Notes
No competing financial interests have been declared.
ACKNOWLEDGMENT
Support from the ISF (190/13 to I.A.W.), the Deutsche For-
schungsgemeinschaft (A.M and I.A.W.), the Kreitman
Foundation (S.K.) and the ERC (Advanced Grant to A.M.).
REFERENCES
1. For cages and containers: (a) Cook, T. R.; Stang, P. J. Chem. Rev.
2015, 115, 7001-7045; (b) Brown, C. J.; Toste, F. D.; Bergman, R. G.;
Raymond, K. N. Chem. Rev. 2015, 115, 3012-3035; (c) Leenders, S.
H. A. M.; Gramage-Doria, R.; de Bruin, B.; Reek, J. N. H. Chem. Soc.
Rev. 2015, 44, 433-448; (d) Ballester, P.; Fujita, M.; Rebek, J., Jr.
Chem. Soc. Rev. 2015, 44, 392-393; (e) Amouri, H.; Desmarets, C.;
Moussa, J. Chem. Rev. 2012, 112, 2015-2041; (f) Ajami, D.; Rebek, J.,
Acc. Chem. Res. 2013, 46, 990-999.; (g) Wiester, M. J.; Ulmann, P. A.;
Mirkin, C. A. Angew. Chem. Int. Ed. 2011, 50, 114-137.
2. (a) Garcia-Simon, C.; Gramage-Doria, R.; Raoufmoghaddam, S.;
Parella, T.; Costas, M.; Ribas, X.; Reek, J. N. H. J. Am. Chem. Soc.
2015, 137, 2680-2687; (b) Zhao, C.; Toste, F. D.; Raymond, K. N.;
Bergman, R. G. J. Am. Chem. Soc. 2014, 136, 14409-14412.
15. Schäffer, C.; Todea, A. M.; Bögge, H.; Petina, O. A.; Rehder,
D.; Haupt, E. T. K.; Müller, A. Chem. Eur. J. 2011, 17, 9634-9639.
16. Kozik, M.; Casan-Pastor, N.; Hammer, C. F.; Baker, L. C. W. J.
Am. Chem. Soc. 1988, 110, 7697-7701.
17. This was done by dialysis against water (see the Supporting
Information). Notably, "flow" methods could be used with porous
membranes to continuously modify capsule reactivity.
18. To our knowledge, this is the largest rate acceleration reported
to date for a cage or container in pure water at room temperature. See
ref. 1b for a discussion of reported rate accelerations.
19. Cacciapaglia, R.; Di Stefano, S.; Mandolini, L. Acc. Chem. Res.
2004, 37, 113-122.
ACS Paragon Plus Environment

 Kopilevich, Sivil
Kopilevich, Sivil
 Müller, Achim
Müller, Achim
 Weinstock, Ira A.
Weinstock, Ira A.