Y. Liu et al. / Process Biochemistry 46 (2011) 390–394
393
Acknowledgments
This work was supported by the Program for New Century
Excellent Talents in University of China (no. NCET-07-0341). The
language was polished by Dr. Zafar Ali Siddiqui.
Appendix A. Supplementary data
Supplementary data associated with this article can be found, in
References
[1] Romano P, Suzzi G. Origin and production of acetoin during wine yeast fermen-
tation. Appl Environ Microbiol 1996;62:309–15.
[2] Rivas B, Torre P, Dominguez JM, Perego P, Converti A, Parajo JC. Carbon material
and bioenergetic balances of xylitol production from corncobs by Debaryomyces
hansenii. Biotechnol Prog 2003;19:706–13.
[3] Toda F, Tanaka K, Tange H. New reduction method of diketones, oxo amides,
and quinones with Zn–EtOH in the presence of a salt. J Chem Soc, Perkin Trans
1989;1:1555–6.
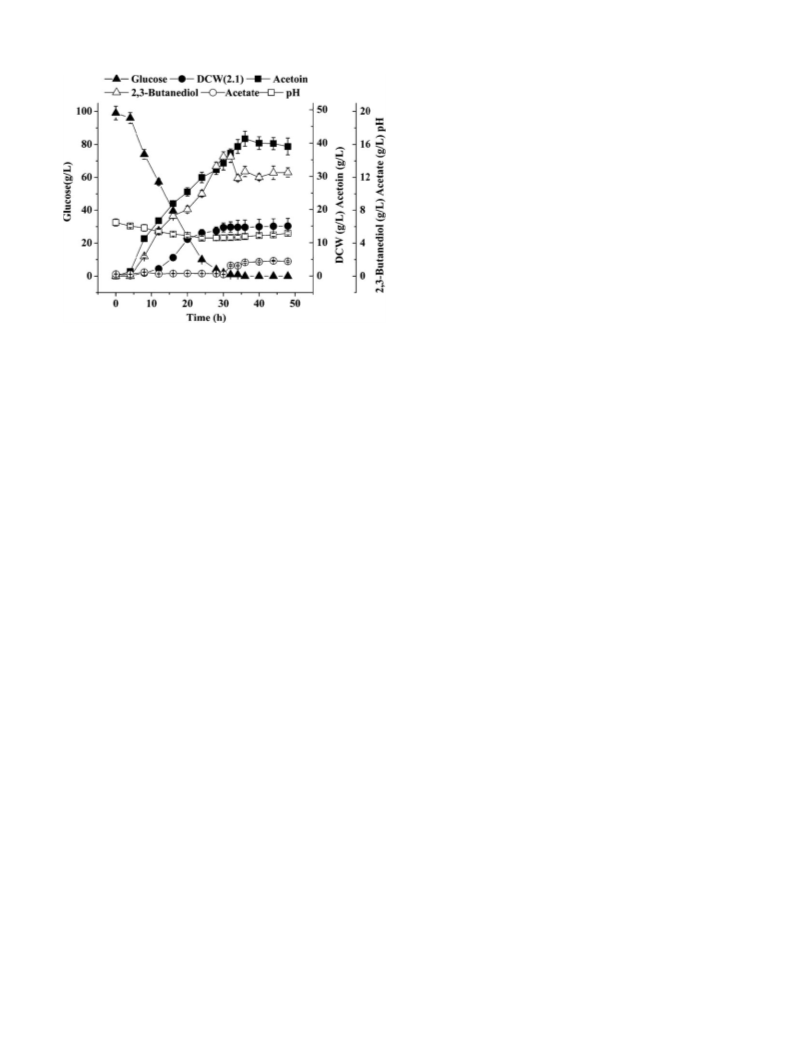
Fig. 3. Time course of acetoin fermentation by B. licheniformis MEL09 at 37 ◦C.
[4] Blom RH. Configuration of acetylmethylcarbinol.
1945;67(8):494–8.
J
Am Chem Soc
[5] Tomita T, Ozawa T, Tomita I. The cause of acetoin production by myo-
inositol deficient Saccharomyces carlsbergensis. J Vitaminol (Kyoto) 1969;15:
215–21.
[6] Garibaldi JA, Bayne HG. Production of acetoin and diacetyl by the genus
Salmonella. Appl Microbiol 1970;20:855–6.
3.3. Time course of acetoin fermentation by B. licheniformis
MEL09
[7] Branen AL, Keenan TW. Diacetyl and acetoin production by Lactobacillus casei.
Appl Microbiol 1971;22:517–21.
The time course of acetoin fermentation by B. licheniformis
MEL09 was monitored under optimal fermentation conditions at
37 ◦C for 48 h. The cell growth was slow during the initial 8 h,
decreased rapidly after 8 h and dropped to zero at 36 h. Acetoin
production increased after a short lag phase (after 4 h), reached a
maximum concentration of 41.26 g l−1 at 36 h, and then decreased
slowly. As shown in Fig. 3, acetoin production may help to maintain
neutral pH, similar to the report on Lactobacillus plantarum [35].
The byproducts formed during acetoin production by B. licheni-
formis MEL09, such as the organic acids diacetyl and 2,3-butanediol,
were also determined. According to the HPLC analysis, for-
mate, lactate, succinate, malate, fumarate and diacetyl were not
detected during the acetoin fermentation. Although acetate could
be detected during the early exponential growth phase, its con-
centration was lower than 1.0 g l−1 during the whole fermentation
process. It was previously proposed that the conversion of pyruvate
to acetoin or 2,3-butanediol prevents overacidification of the cul-
ture during exponential growth [36]. The production of the main
byproduct 2,3-butanediol increased after 8 h, reached a maximum
concentration of 14.52 g l−1 at 30 h, and then decreased slowly.
[8] Collins EB, Speckman RA. Influence of acetaldehyde on growth and acetoin
production by Leuconostoc citrovorum. J Dairy Sci 1974;57:1428–31.
[9] Bassit N, Boquien CY, Picque D, Corrieu G. Effect of initial oxygen concentration
on diacetyl and acetoin production by Lactococcus lactis subsp. lactis biovar
[10] Can˜as A, Owens JD. Acetoin production in growing Leuconostoc mesenteroides.
World J Microbiol Biotechnol 1999;62:339–44.
[11] Romano P, Suzzi G, Zironi R, Comi G. Biometric study of acetoin production in
Hanseniaspora guilliermondii and Kloeckera apiculata. Appl Environ Microbiol
1993;59:1838–41.
[12] Teixeira RM, Cavalheiro D, Ninow JL, Furigo AJ. Optimization of acetoin produc-
tion by Hanseniaspora guilliermondii using experimental design. Braz J Chem
Eng 2002;19:181–6.
[13] Yu EK, Saddler JN. Fed-batch approach to production of 2,3-butanediol by
Klebsiella pneumoniae grown on high substrate concentrations. Appl Environ
Microbiol 1983;46:630–5.
[14] Qin J, Xiao Z, Ma C, Xie N, Liu P, Xu P. Production of 2,3-butanediol by Kleb-
siella pneumoniae using glucose and ammonium phosphate. Chin J Chem Eng
2006;14(1):132–6.
[15] Zeng AP, Biebl H, Deckwer WD. Production of 2,3-butanediol in a membrane
bioreactor with cell recycle. Appl Microbiol Biotechnol 1991;34:463–8.
[16] Dettwiler B, Dunn IJ, Heinzle E, Prenosil JE. A simulation model for the continu-
ous production of acetoin and butanediol using Bacillus subtilis with integrated
pervaporation separation. Biotechnol Bioeng 1993;41:791–800.
[17] Xiao ZJ, Liu PH, Qin JY, Xu P. Statistical optimization of medium components
for enhanced acetoin production from molasses and soybean meal hydrolysate.
Appl Microbiol Biotechnol 2007;74:61–8.
[18] Schallmey M, Singh A, Ward OP. Developments in the use of Bacillus species for
industrial production. Can J Microbiol 2004;128(50):17–51.
[19] Tourney J, Ngwenya BT, Fred Mosselmans JW, Magennis M. Physical and
chemical effects of extracellular polymers (EPS) on Zn adsorption to Bacillus
licheniformis S-86. J Colloid Interface Sci 2009;337:381–9.
4. Conclusions
[20] Bajaj IB, Lele SS, Singhal RS. Enhanced production of poly (gamma-glutamic
acid) from Bacillus licheniformis NCIM 2324 in solid-state fermentation. J Ind
Microbiol Biotechnol 2008;35:1581–6.
[21] Kalishwaralal K, Deepak V, Ram Kumar Pandian S, Gurunathan S. Biologi-
cal synthesis of gold nanocubes from Bacillus licheniformis. Bioresour Technol
2009;100:5356–8.
[22] Li Z, Zhong S, Lei HY, Chen RW, Yu Q, Li HL. Production of a novel bioflocculant
by Bacillus licheniformis X14 and its application to low temperature drinking
water treatment. Bioresour Technol 2009;100:3650–6.
[23] Perego P, Converti A, Del Borghi M. Effects of temperature, inoculum size and
starch hydrolyzate concentration on butanediol production by Bacillus licheni-
formis. Bioresour Technol 2003;89:125–31.
[24] Nicholson WL. The Bacillus subtilis ydjL (bdhA) gene encodes ace-
toin reductase/2,3-butanediol dehydrogenase. Appl Environ Microbiol
2008;74:6832–8.
[25] Underwood SA, Zhou S, Causey TB, Yomano LP, Shanmugam KT, Ingram
LO. Genetic changes to optimize carbon partitioning between ethanol
and biosynthesis in ethanologenic Escherichia coli. Appl Environ Microbiol
2002;68:6263–72.
A new acetoin-producing strain of B. licheniformis, MEL09, was
isolated and identified. By screening different factors affecting the
production of acetoin, optimal media and culture conditions were
found, and the highest acetoin production yet by B. licheniformis
MEL09 was obtained. Under optimized conditions, the acetoin
concentration, GCE and productivity were at 41.26 g l−1, 41.26%
and 1.15 g l−1 h−1, respectively (an increase of 84.86%, 47.89% and
85.48% as compared with the initial cultural conditions). Moreover,
the maximum cell growth (g l−1) was significantly improved (from
6.36 to 10.10), and Ya/x (g g−1) was enhanced from 3.50 to 4.08. In
addition, B. licheniformis MEL09 showed favorable characteristics
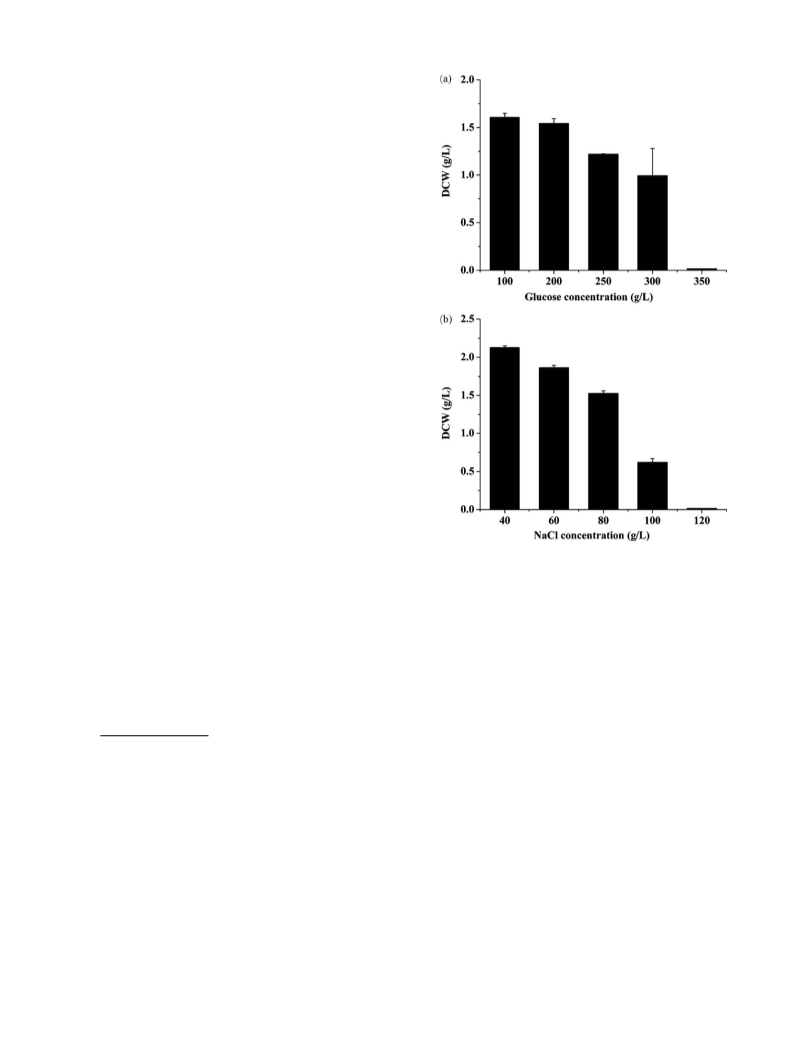
for industrial application, such as resistance to high osmolarity.
Future goals include reducing levels of byproducts such as 2,3-
butanediol and increasing GCE under high glucose concentration.

 Liu, Yongfeng
Liu, Yongfeng
 Zhang, Shuling
Zhang, Shuling
 Yong, Yang-Chun
Yong, Yang-Chun
 Ji, Zhixia
Ji, Zhixia
 Ma, Xin
Ma, Xin
 Xu, Zhenghong
Xu, Zhenghong
 Chen, Shouwen
Chen, Shouwen