(3H, s, CH -28), 1.60 (3H, s, CH -21), 1.61 (3H, s, CH -27), 1.64 (3H, s, CH -26), 4.85 (1H, d, J = 7.6, H-1ꢂ), 4.93 (1H, d,
3
3
3
3
J = 6.0, H-1ꢂꢂꢂꢂ), 5.07 (1H, d, J = 7.6, H-1ꢂꢂꢂ), 5.28 (1H, d, J = 7.6, H-1ꢂꢂ) (only characteristic proton resonances are given).
+
Mass spectrum: FAB, m/z 1101 [M + Na] .
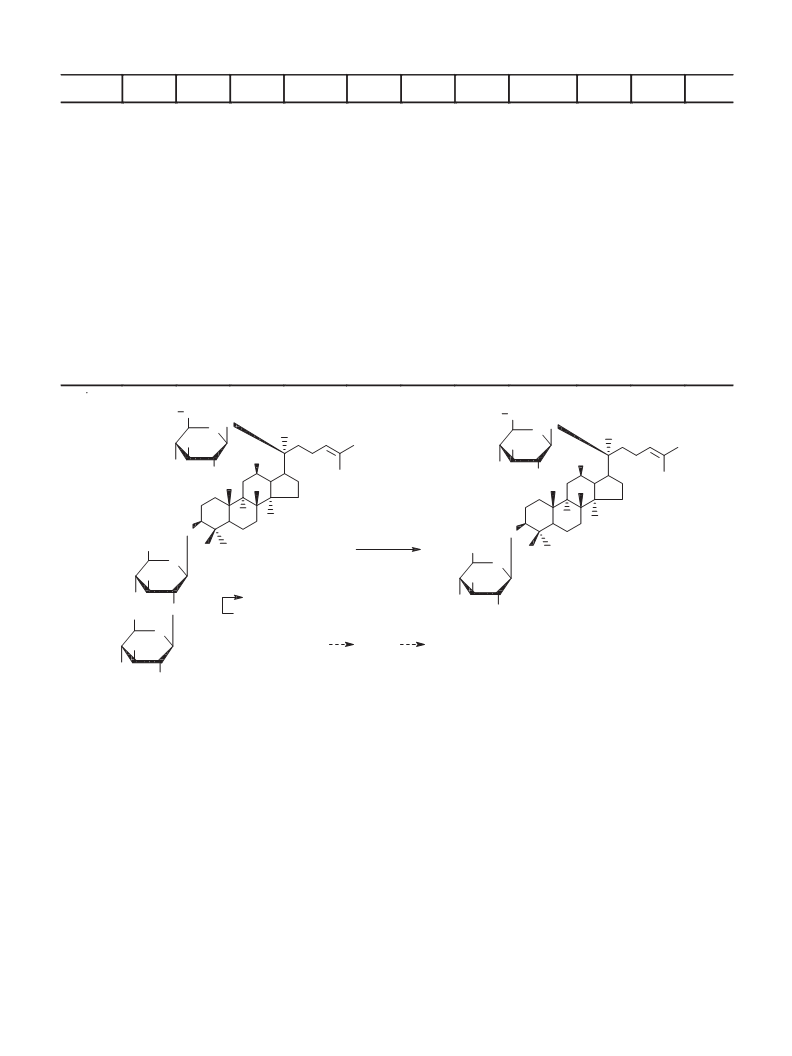
3-O-(ꢀ-D-Glucopyranosyl)-20-O-[ꢅ-L-arabinopyranosyl-(1ꢄ6)-ꢀ-D-glucopyranosyl]-3ꢀ,12ꢀ,20ꢀ-
1
trihydroxydammar-24-ene (6). Yield 69%. C H O . White powder, mp 185–186°C. Í NMR spectrum (500 MHz,
47 80 17
Py-d , ꢁ, ppm, J/Hz): 0.86 (3H, s, CH -19), 0.93 (3H, s, CH -18), 0.98 (3H, s, CH -30), 1.01 (3H, s, CH -29), 1.22 (3H, s,
5
3
3
3
3
CH -28), 1.64 (3H, s, CH -21), 1.66 (3H, s, CH -27), 1.69 (3H, s, CH -26), 4.85 (1H, d, J = 7.6, H-1ꢂ), 4.92 (1H, d, J = 6.0,
3
3
3
3
H-1ꢂꢂꢂꢂ), 5.07 (1H, d, J = 7.6, H-1ꢂꢂꢂ) (only characteristic proton resonances are given). Mass spectrum: FAB, m/z 939
+
[M + Na] .
3-O-(ꢀ-D-Glucopyranosyl)-20-O-[ꢀ-D-glucopyranosyl-(1ꢄ6)-ꢀ-D-glucopyranosyl]-3ꢀ,12ꢀ,20ꢀ-
1
trihydroxydammar-24-ene (8). C H O . White powder, mp 179–181°C. Í NMR spectrum (500 MHz, Py-d , ꢁ, ppm,
48 82 18
5
J/Hz): 0.76 (3H, s, CH -19), 0.92 (3H, s, CH -18), 0.94 (3H, s, CH -30), 0.95 (3H, s, CH -29), 1.26 (3H, s, CH -28), 1.57 (3H,
3
3
3
3
3
s, CH -26), 1.62 (6H, s, CH -21, 27), 4.90 (1H, d, J = 7.6, H-1ꢂ), 5.04 (1H, d, J = 7.6, H-1ꢂꢂꢂꢂ), 5.09 (1H, d, J = 7.6, H-1ꢂꢂꢂ) (only
3
3
+
characteristic proton resonances are given). Mass spectrum: FAB, m/z 969 [M + Na] .
ACKNOWLEDGMENT
This work partially supported by the Brain Pool Program of 2014, Republic Korea.
REFERENCES
1.
2.
K. T. Choi, Acta Pharmacol. Sin., 29, 1109 (2008).
L. P. Christensen, Adv. Food Nutr. Res., 55, 1 (2009).
3.
4.
H. S. Jee, K. H. Chang, S. H. Park, K. T. Kim, and H. D. Paik, Food. Rev. Int., 30, 91 (2014).
J. D. Park, D. K. Rhee, and Y. H. Lee, Phytochem. Rev., 4, 159 (2005).
5.
6.
7.
8.
K. W. Leung, Natural Products, K. G. Ramawat and J. M. Merillon (eds.), Springer, Berlin, 2013, p. 3498.
Y. Zheng, H. Nan, M. Hao, C. Song, Y. Zhou, and Y. Gao, Biomed. Rep., 1, 555 (2013).
T. T. Mai, J. Moon, Y. Song, P. Q. Viet, and P. V. Phuc, Cancer Lett., 321, 144 (2012).
Q. Mao, P. H. Zhang, Q. Wang, and S. L. Li, Phytomedicine, 21, 515 (2014).
F. Gao, J. M. Zhang, Z. G. Wang, W. Peng, H. L. Hu, and C. M. Fu, Asian Pac. J. Cancer Prev., 14, 5599 (2013).
L. N. Ten, S. H. Baek, W. T. Im, M. Lee, H. W. Oh, and S. T. Lee, Int. J. Syst. Evol. Microbiol., 56, 2677 (2006).
M. H. Yoon, L. N. Ten, and W. T. Im, Int. J. Syst. Evol. Microbiol., 57, 1810 (2007).
L. N. Ten, S. M. Chae, and S.-A. Yoo, Chem. Nat. Compd., 49, 773 (2013).
L. N. Ten, S. M. Chae, and S.-A. Yoo, Chem. Nat. Compd., 49, 1168 (2014).
L. N. Ten, S. M. Chae, and S.-A. Yoo, Chem. Nat. Compd., 50, 565 (2014).
X. Zhao, J. Gao, C. Song, Q. Fang, N. Wang, T. Zhao, D. Liu, and Y. Zhou, Phytochemistry, 78, 65 (2012).
C. H. Cui, S. C. Kim, and W. T. Im, Appl. Microbiol. Biotechnol., 97, 649 (2013).
S. J. Park, S. Y. Youn, G. E. Ji, and M. S. Park, Food Sci. Biotechnol., 21, 839 (2012).
X. F. Jin, H. S. Yu, D. M. Wang, T. Q. Liu, C. Y. Liu, D. S. An, W. T. Im, S. G. Kim, and F. X. Jin, J. Microbiol.
Biotechnol., 22, 343 (2012).
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
20.
21.
22.
C. S. Park, M. H. Yoo, K. H. Noh, and D. K. Oh, Appl. Microbiol. Biotechnol., 87, 9 (2010).
H. Koizumi, S. Sanada, Y. Ida, and J. Shoji, Chem. Pharm. Bull., 30, 2393 (1982).
K. C. Shin and D. K. Oh, J. Biotechnol., 172, 30 (2014).
L. Wang, Q. M. Liu, B. H. Sung, D. S. An, H. G. Lee, S. G. Kim, S. C. Kim, S. T. Lee, and W. T. Im,
J. Biotechnol., 156, 125 (2011).
23.
D. S. An, C. H. Cui, H. G. Lee, L. Wang, S. C. Kim, S. T. Lee, F. Jin, H. Yu. Y. W. Chin, H. K. Lee, W. T. Im,
and S. G. Kim, Appl. Environ. Microbiol., 76, 5827 (2010).
24.
25.
M. C. Yang, D. S. Seo, J. Hong, S. H. Hong, Y. C. Kim, and K. R. Lee, Nat. Prod. Sci., 14, 171 (2008).
L. N. Ten, S. M. Chae, and S.-A. Yoo, Chem. Nat. Compd., 50, 562 (2014).
696

 Ten
Ten
 Chae
Chae
 Yoo
Yoo