research papers
the use of cocaine/crack, two illegal drugs, is much less than
that of legal drugs such as nicotine and alcohol, its social
impact is disproportionately high. Continuous use of the drug
drives the individual to develop social incapacitation, followed
by serious physical and psychological problems, such as
cardiac arrhythmia, stroke, heart attack, difficulty breathing,
hallucinations and paranoia (Kauer & Malenka, 2007).
Most individuals who use cocaine or crack also use ethanol.
From the perspective of the user, concomitant cocaine and
ethanol use has the effect of enhancing and prolonging the
euphoria associated with cocaine while ameliorating the
unwanted agitation and paranoia. In addition, low levels of
ethanol and cocaine have been found to have a synergistic
effect that surpasses the pleasant state felt as a result of using
either one alone (McCance-Katz et al., 1993). This is due to the
fact that the concomitant use of cocaine and ethanol produces
another psychoactive substance known as cocaethylene, which
has pharmacological properties similar to those of cocaine but
which has a plasma half-life three to five times longer than that
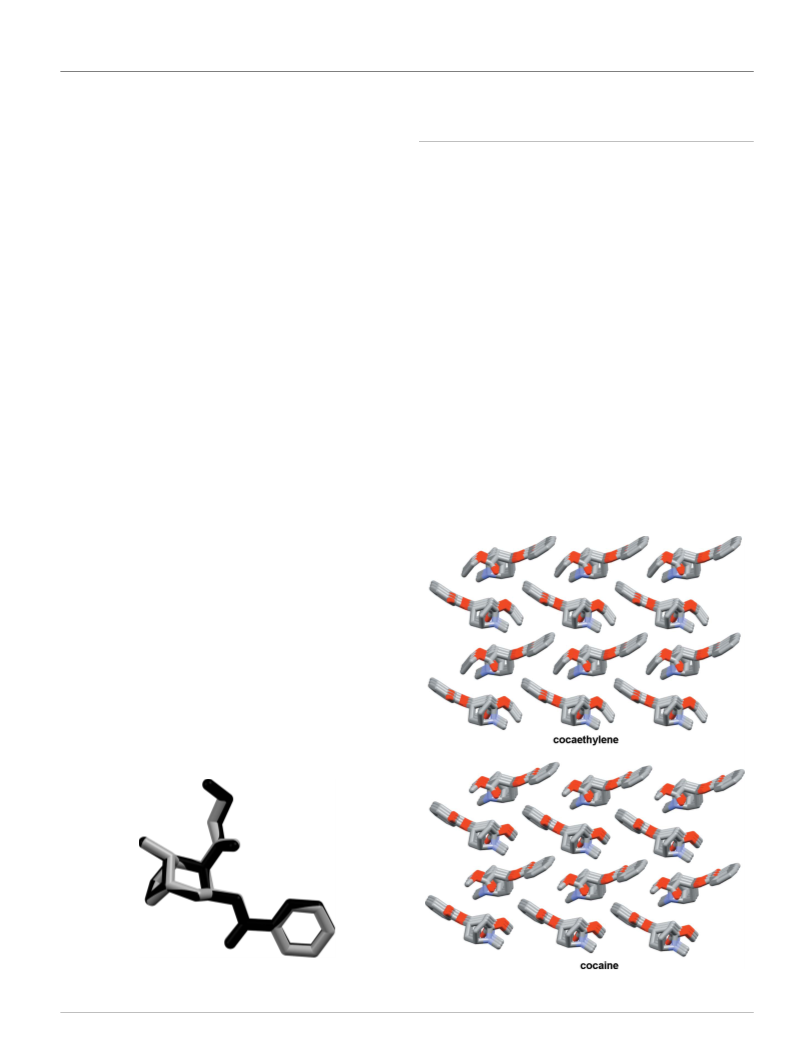
of cocaine (Jatlow, 1993). From a synthetic point of view,
cocaethylene is the ethyl ester of benzoylecgonine, obtained
by a transesterification reaction between cocaine and ethanol.
Cocaethylene has an elimination half-life of 150 min
Table 1
Experimental details.
Crystal data
Chemical formula
18 4
C H23NO
M
r
317.37
Monoclinic, P2
1
296
8.4533 (2), 10.2380 (3), 10.2552 (2)
107.881 (1)
844.66 (4)
2
Cu Kꢀ
0.72
Crystal system, space group
Temperature (K)
˚
a, b, c (A)
ꢀ
ꢁ
V (A )
( )
˚
3
Z
Radiation type
À1
ꢂ (mm
Crystal size (mm)
)
0.35 Â 0.15 Â 0.10
Data collection
Diffractometer
Bruker APEXII CCD area
detector
Multi-scan (SADABS; Krause et
al., 2015)
0.725, 0.931
Absorption correction
T
No. of measured, independent and
min, Tmax
3969, 2133, 2026
observed [I > 2ꢃ(I)] reflections
R
int
0.024
0.595
˚
À1
)
(sin ꢄ/ꢅ)max (A
Refinement
2
2
2
R[F > 2ꢃ(F )], wR(F ), S
No. of reflections
0.036, 0.101, 1.03
2133
208
1
No. of parameters
No. of restraints
H-atom treatment
Áꢆmax, Áꢆmin (e A
Absolute structure
(compared with approximately 90 min for cocaine), which
H-atom parameters constrained
0.15, À0.17
explains the prolonged euphoric effects reported after use of
cocaine and ethanol together (Boghdadi et al., 1997). Both
drugs increase heart rate and systolic blood pressure, but
cocaethylene tends to have a greater effect on blood pressure
˚
À3
)
Flack x parameter determined
+
À
using 568 quotients [(I ) À (I )]/
+
(I ) + (I )] (Parsons et al., 2013)
À
[
Absolute structure parameter
À0.03 (14)
(McCance et al., 1995). The increased cardiovascular effects of
Computer programs: APEX2 (Bruker, 2003), SAINT (Bruker, 2003), SIR92 (Altomare et
al., 1993), SHELXL2014 (Sheldrick, 2015), ORTEP-3 for Windows (Farrugia, 2012),
Mercury (Macrae et al., 2006) and WinGX (Farrugia, 2012).
cocaethylene contribute to the high mortality reported in
chronic cocaine users (Farre et al., 1993).
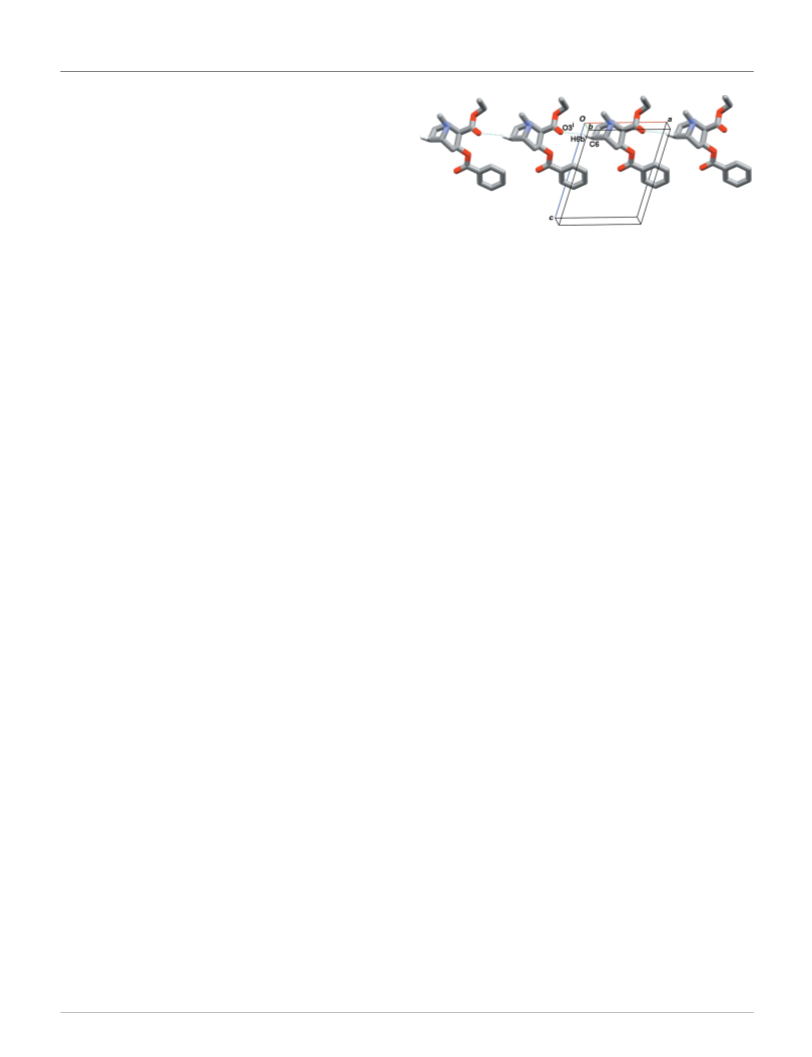
Despite the toxicological and social impact of cocaethylene,
its molecular conformation and supramolecular architecture
are unknown thus far. Here, we fill this gap through the first
single-crystal structure determination of cocaethylene, (I),
which is an even more potent narcotic than its precursor
cocaine. The knowledge derived from this study will be useful
for understanding the psychoactive profile of cocaethylene
and for providing an analytical pattern for narcotic control
purposes.
2
. Experimental
2.1. Synthesis and crystallization
Cocaethylene was synthesized following the protocol
available in the literature (Everhart et al., 1999). Benzoyl-
ecgonine (500 mg), previously synthesized according to the
methodology described by Findlay (1954), was stirred together
with oxalyl chloride (2.5 ml) for 20 min at room temperature.
Next, the mixture was cooled to 273 K prior to the dropwise
addition of ethanol (5 ml). After complete addition of ethanol
at 273 K, the mixture was again stirred at room temperature
for 12 h. The residual solvent was then withdrawn under
reduced pressure in a rotary evaporator and the obtained
product was dissolved in acidified water (2 ml of water and
0.1 ml of H SO ). This solution was then mixed together with a
2 4
mixture of diethyl ether (4 ml) and saturated ammonium
hydroxide aqueous solution (0.5 ml). The remaining aqueous
phase was washed twice with diethyl ether (5 ml). The organic
phases were pooled and then subjected to silica-gel column
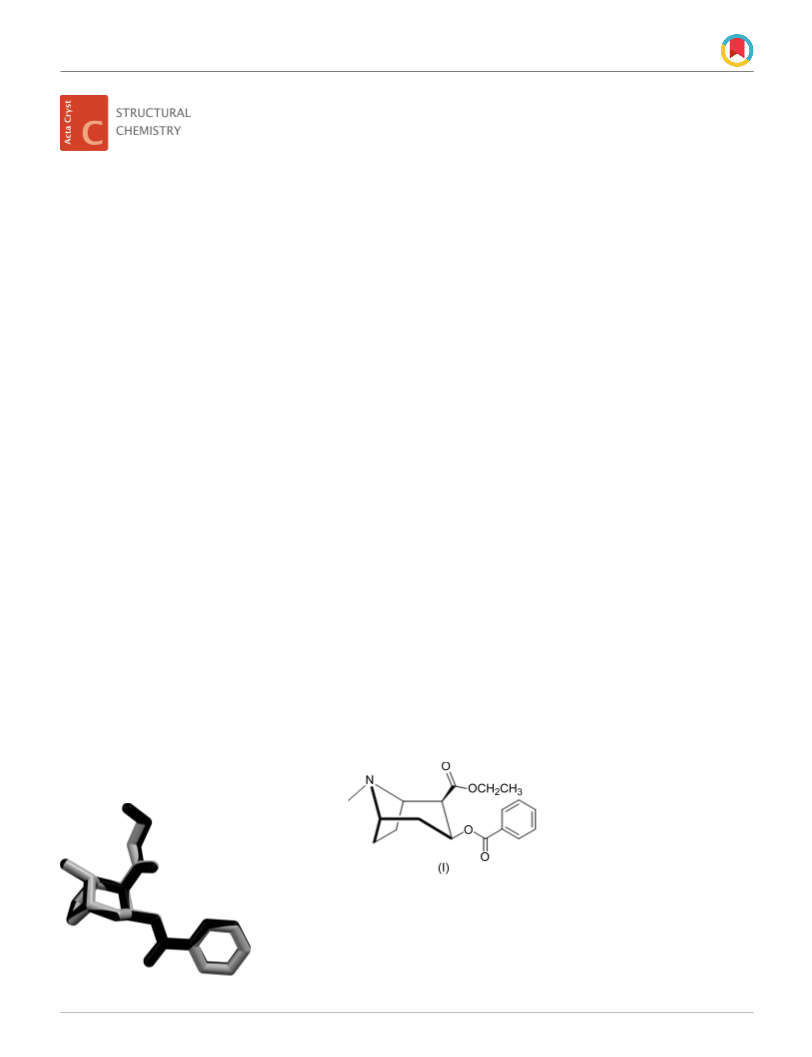
Figure 1
The molecular structure of cocaethylene, showing the atom-labelling
scheme. Displacement ellipsoids are drawn at the 30% probability level
and H atoms are shown as spheres of arbitrary radii.
ꢁ
Acta Cryst. (2017). C73, 780–783
Maia et al.
C
18
H
23NO
4
781

 Da Silva Maia, Angélica Faleiros
Da Silva Maia, Angélica Faleiros
 Martins, Felipe T.
Martins, Felipe T.
 Da Silva Neto, Leonardo
Da Silva Neto, Leonardo
 Alves, Rosemeire Brondi
Alves, Rosemeire Brondi
 De Fátim, Angelo
De Fátim, Angelo