INTERACTION OF ANTHOCYANINS AND ANTHOCYANIDINS
389
Gradient elution mode of separation of radiolysis products
electronic absorption spectra of the solutions were
recorded on an SFꢀ2000 spectrophotometer in special
quartz cells (10 mm in optical path length) connected
with an ampoule for evacuation.
Time, min Flow rate, mL/min
A, %
B, %
The products of radiationꢀinduced transformations
of anthocyanins were identified by liquid chromatogꢀ
raphy–mass spectrometry using an Acquity UPLC
chromatograph with a tandem quadrupole detector.
The separation was performed on an ACQUITY
0
0.3
0.3
0.3
0.3
95
95
30
0
5
5
0.25
6.50
7.60
70
UPLC
®
R BEH C18 50
×
2
mm 1.7 m column in the
µ
gradient elution mode as specified in the table. The
mobile phase was composed of (A) 0.1% formic acid in
water and (B) acetonitrile.
100
Spectrophotometric scans were in the range of
200–800 nm. Mass spectrometric detection was carꢀ
ried out under the following conditions (found by
optimizing the determination of MalvꢀGl using the
program InteliStart): the ESI+ operating mode, a
voltage across the capillary of 3.0 kV, a cone voltage of
30 V, a source temperature of 150°С, a drying gas
(nitrogen) flow rate of 1000 L/h, and a collisionꢀcell
voltage of 30 V (highꢀpurity argon gas).
Acetaldehyde was determined as its hydrazone with
2,4ꢀdinitrophenylhydrazine using HPLC.
2,4ꢀDinitrophenylhydrazine, purified by recrystalꢀ
lization from its ethanol solution saturated at a temꢀ
(4)
OH + C2H5OH → CH3CHOH + H2O.
Thus, it may be assumed that HER is the only radꢀ
ical that can react with the test substance in this sysꢀ
tem. In addition, the presence of the acid ensures the
stability of anthocyanins and anthocyanidins, which
degrade in a neutral or alkaline solution. In the
absence of additives, HERs are consumed in combiꢀ
nation (5) and disproportionation reactions (6) to give
2,3ꢀbutanediol and acetaldehyde, respectively:
CH3CHOH + CH3CHOH
(5)
→ CH3CH(OH)CH(OH)CH3,
perature of 50⎯60°С, was dissolved in acetonitrile to
CH3CHOH + CH3CHOH
(6)
have a concentration of 1 mg/mL. The 2,4ꢀdinitroꢀ
phenylhydrazine solution and phosphoric acid (1 : 7)
in an amount of 0.2 mL each were added to 1 mL of an
→ CH3CHO + CH3CH2OH.
The irradiation of MalvꢀGl and CyꢀGl glycoside
alcohol solution, and 1 L of the mixture was injected solutions results in the most significant changes in the
μ
into the chromatograph after 20 minutes.
The mobile phase was the 60/40 acetonitrile–water
blend used at a flow rate of 0.3 mL/min. The
visible region of their electronic absorption spectra, as
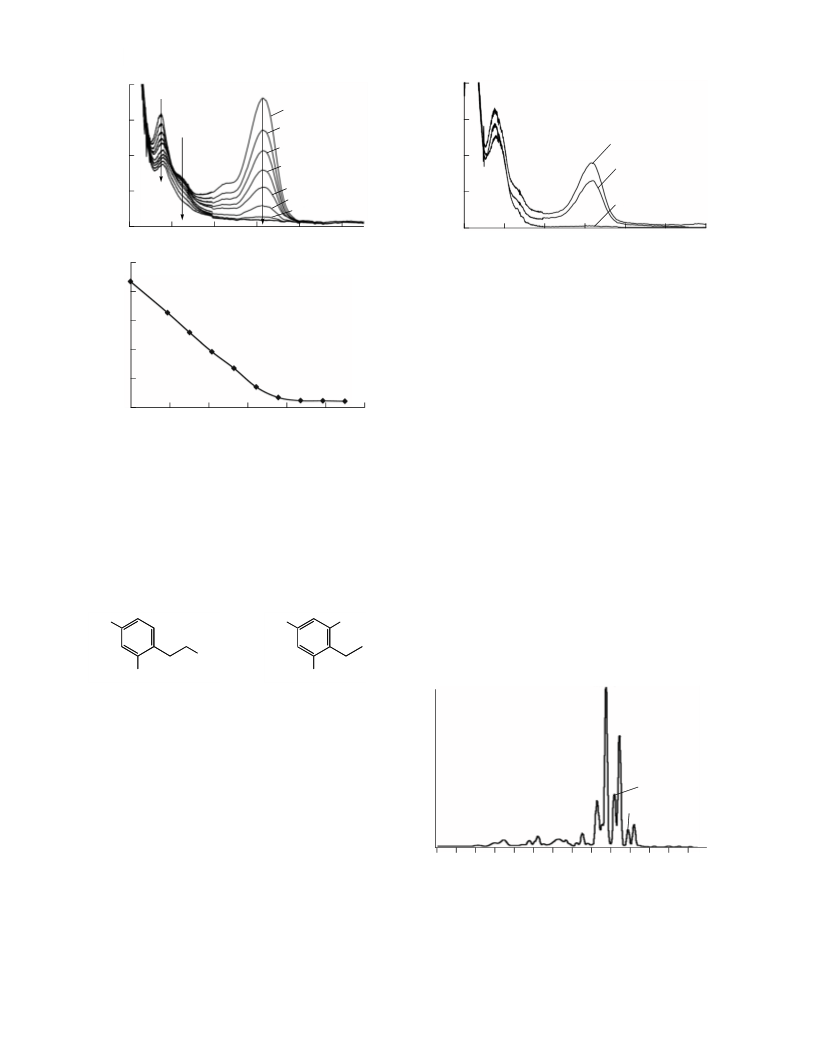
shown in Fig. 1a, and these changes are linearly
related to the absorbed dose (Fig. 1b). The disappearꢀ
ance of the absorption band in the visible part of the
spectrum implies that the reaction of MalvꢀGl with
HER destroys conjugation between ring B and the rest
of the molecule, since it is this conjugation that is
responsible for the electronic transition in anthocyaꢀ
nins at 520 nm [13].
ACQUITY UPLC
column temperature was 40
photometric at a wavelength of 360 nm, and the samꢀ
®
BEH C18
(
50
×
2 mm, 1.7
µ
m)
°С, detection was spectroꢀ
ple chamber temperature was 20°С.
RESULTS
After unsealing the ampoule and saturating the
irradiated solution with atmospheric oxygen, the color
is restored and the absorption spectra of the initial
unirradiated solution and the aerated irradiated soluꢀ
tion differ for the most part in the intensity alone, as
shown in Fig. 2.
Because of the low concentration of alcohol and
other solutes, most (~95%) of the ionizing radiation
energy incident on the system was absorbed by water,
generating a significant amount of free radicals, whose
yield is well documented [12]:
The products of the MalvꢀGl reaction with HER
were separated chromatographically (Fig. 3a). Two
major products with retention times of 2.55 and
2.69 min had the mass spectrometric characteristics
H2O
e
(2.8–2.9),
·
H(0.6),
O
H(2.8–2.9),
(1)
H2(4.5), H2O2(0.75).
similar to those of MalvꢀGl (m/z = 655). These species
were supposed to be protonated ions (M + H)+, not
the molecular ions as in the case of MalvꢀGl. To verify
this hypothesis, the flow from the chromatograph was
mixed in the mass spectrometric detector with an
ammonia solution, an operation that would lead to the
formation of the (M + NH3)+ ion and an increase in
Since the alcohol and hydrochloric acid concenꢀ
trations are large compared with the test substances,
the hydroxyl radical and the electron react with them,
giving ultimately HER:
+
(2)
(3)
e + H → H,
m/z by 16. However, m/z remained unchanged: thus, it
H + C2H5OH → CH3CHOH + H2,
HIGH ENERGY CHEMISTRY
Vol. 49
No. 6
2015

 Domnina
Domnina
 Fenin
Fenin
 Ermakov
Ermakov
 Magomedbekov
Magomedbekov