3929
Scheme 2. Reagents and conditions. (a) DIBAL, toluene, −78°C, 1 h, 95%; (b) Ph3P_CHCOOMe, benzene, 23°C, 1 h, 64%;
(c) I2, Ph3P, imidazole, toluene, 23°C, 1 h, 79%; (d) DIBAL, toluene, −78°C, 1 h; (e) DHP, p-TsOH, CH2Cl2, 23°C, 1 h, 90% in
two steps; (f) cycloheptanone N,N-dimethylhydrazone, n-BuLi, THF, 23°C, 2 h; (g) silica gel, CH2Cl2, 23°C, 19 h, 92% in two
steps; (h) conc. HCl, dioxane, 23°C, 5 h, 94%; (i) t-BuOK, t-BuOH, reflux, 3.5 h, 28% for 10a, 43% for 10b; (j) allyl iodide,
KHMDS, THF, 0°C, 3 h, 81% (3:11=7:1); (k) RuCl2(_CHPh)(PCy3)2, toluene, reflux, 20% for 12, 76% for 13; (l) H2, Pd/C,
EtOH, 23°C, 1 h, 55% for 2, 68% for 14
References
1. Zeichmeister, K.; Brandl, F.; Hoffe, W.; Hecker, E.; Opferkuch, H. J.; Adolf, W. Tetrahedron Lett. 1970, 4075–4078.
Uemura, D.; Hirata, Y. Tetrahedron Lett. 1971, 3673–3676. Opferkuch, H.; Hecker, E. Tetrahedron Lett. 1974, 261–264.
Uemura, D.; Ohwaki, H.; Hirata, Y.; Chen, Y.-P.; Hsu, H.-Y. Tetrahedron Lett. 1974, 2527–2528. Uemura, D.; Hirata, Y.;
Chen, Y.-P.; Hsu, H.-Y. Tetrahedron Lett. 1974, 2529–2532. Upadhyay, R. R.; Mohadded, J. Curr. Sci. 1987, 56, 1058–1059.
2. Hasler, C. M.; Acs, G.; Blumberg, M. Cancer Res. 1992, 52, 202–208.
3. Fujiwara, M.; Ijichi, K.; Tokuhisa, K.; Katsuura, K.; Shigeta, S.; Konno, K.; Wang, G.-Y.-S.; Uemura, D.; Yokota, T.; Baba,
M. Antimicrob. Agents Chemother. 1996, 40, 271–273. Fujiwara, M.; Ijichi, K.; Tokuhisa, K.; Katsuura, K.; Wang, G.-Y.-S.;
Uemura, D.; Shigeta, S.; Konno, K.; Yokota, T.; Baba, M. Antiviral Chemistry & Chemotherapy 1996, 7, 230–236.
4. Review: Kim, S.; Winkler, J. D. Chem. Soc. Rev. 1997, 26, 387–399.
5. (a) Winkler, J. D.; Hey, J. P.; Williard, P. G. J. Am. Chem. Soc. 1986, 108, 6425–6426. (b) Winkler, J. D.; Henegar, K. E.;
Williard, P. G. J. Am. Chem. Soc. 1987, 109, 2850–2851. (c) Winkler, J. D.; Henegar, K. E.; Hong, B.-C.; Williard, P. G. J.
Am. Chem. Soc. 1994, 116, 4183–4188. (d) Winkler, J. D.; Hong, B.-C.; Bahador, A.; Kazanietz, M. G.; Blumberg, P. M.
J. Org. Chem. 1995, 60, 1381–1390. (e) Winkler, J. D.; Kim, S.; Harrison, S.; Lewin, N. E.; Blumberg, P. M. J. Am. Chem.
Soc. 1999, 121, 296–300.
6. (a) Funk, R. L.; Olmstead, T. A.; Parvez, M. J. Am. Chem. Soc. 1988, 110, 3298–3300. (b) Funk, R. L.; Olmstead, T. A.;
Parvez, M.; Stallman, J. B. J. Org. Chem. 1993, 58, 5873–5875.
7. (a) Rigby, J. H.; de Sainte Claire, V.; Cuisiat, S. V.; Heeg, M. J. J. Org. Chem. 1996, 61, 7992–7993. (b) Rigby, J. H.; Hu,
J.; Heeg, M. J. Tetrahedron Lett. 1998, 39, 2265–2268.
8. Nakamura, T.; Matsui, T.; Tanino, K.; Kuwajima, I. J. Org. Chem. 1997, 62, 3032–3033.
9. Review: Grubbs, R. H.; Chang, S. Tetrahedron 1998, 54, 4413–4450.
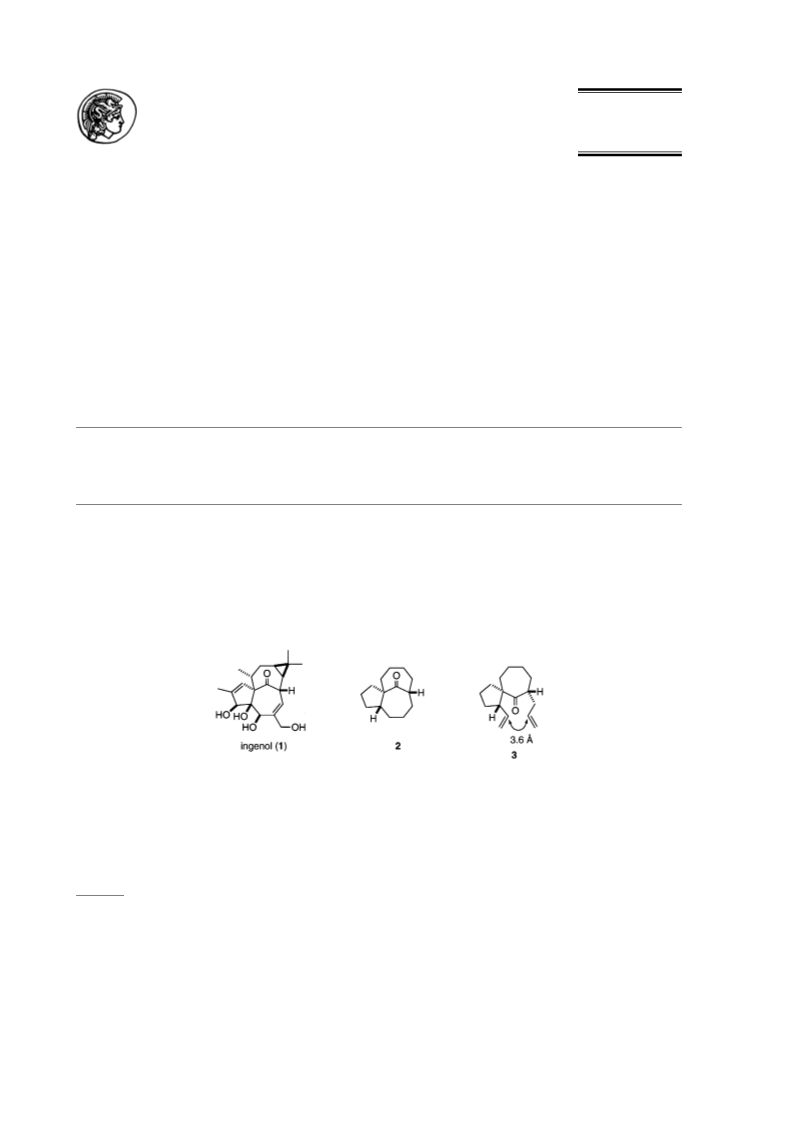
10. The global minima were calculated in a Multiconformer conformational search using MacroModel (Version 6.0).

 Kigoshi, Hideo
Kigoshi, Hideo
 Suzuki, Yuto
Suzuki, Yuto
 Aoki, Kenta
Aoki, Kenta
 Uemura, Daisuke
Uemura, Daisuke