2
736
D. M. Cermak et al. / Bioorg. Med. Chem. 8 (2000) 2729±2737
2
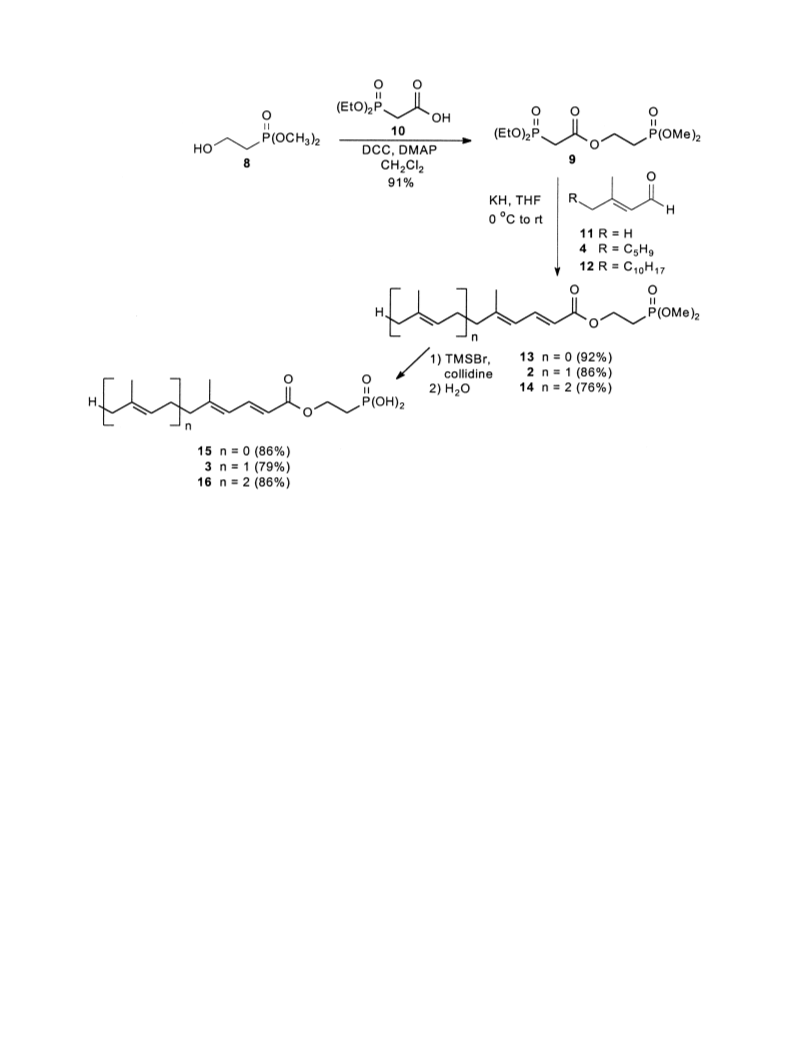
-(Dimethoxyphosphinyl)ethyl (E)-5,9-dimethyl-4,8-deca-
Science 1993, 260, 1934. (d) James, G. L.; Goldstein, J. L.;
Brown, M. S.; Rawson, T. E.; Somers, T. C.; McDowell, R. S.;
Crowley, C. W.; Lucas, B. K.; Levinson, A. D.; Marsters, J.
C., Jr. Science 1993, 260, 1937. (e) Garcia, A. M.; Rowell, C.;
Ackermann, K.; Kowalczyk, J. J.; Lewis, M. D. J. Biol. Chem.
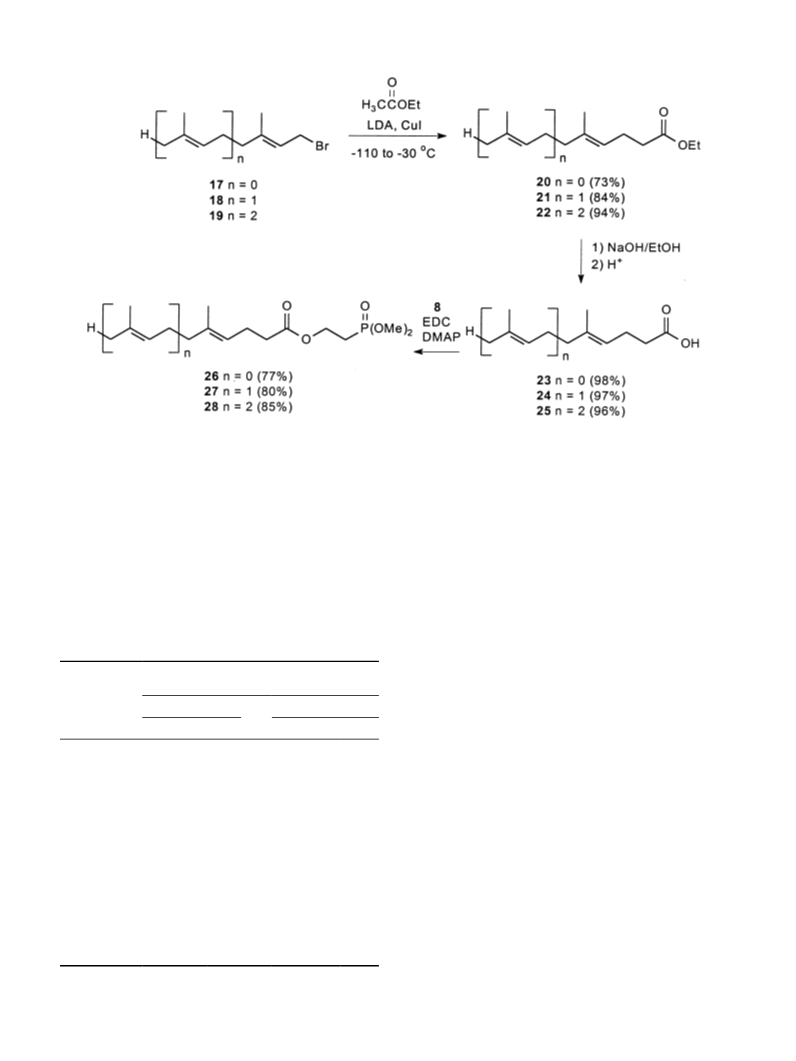
dienoate (27). A solution of acid 24 (165 mg, 0.84
mmol), phosphonate 8 (116 mL, 0.931 mmol), DCC
(
191 mg, 0.926 mmol) and DMAP (11 mg, 0.09 mmol) in
dichloromethane (8.4 mL) was allowed to react as
described for compound 26. After 4 h, standard work up
and ®nal puri®cation by ¯ash column chromatography
1
993, 268, 18415. (f) Nigam, M.; Seong, C.; Qian, Y.; Hamil-
ton, A.; Sebti, S. J. Biol. Chem. 1993, 268, 20695. (g) Qian Y.
M.; Marugan J. J.; Fossum, R. D.; Vogt, A.; Sebti, S. M.;
Hamilton, A. D. Bioorg. Med. Chem. 1999, 7, 3011.
(
ethyl acetate) gave compound 27 as a clear oil (224 mg,
1
8
7
4
1
3
5
0%): H NMR d 5.03±5.14 (m, 2H), 4.30 (dt, J=12.8,
.4 Hz, 2H), 3.76 (d, J=10.9 Hz, 6H), 2.29±2.37 (m,
H), 2.17 (dt, J=18.8, 7.4 Hz, 2H), 1.93±2.10 (m, 4H),
.67 (d, J=0.9 Hz, 3H), 1.62 (d, J=0.9 Hz, 3H), 1.60 (s,
H); C NMR d 172.7, 136.5, 131.1, 123.9, 121.9, 58.0,
2.2 (d, JCP=6.7 Hz), 39.4, 34.1, 26.4, 25.4, 24.8 (d,
7
. (a) Singh, S. B.; Zink, D. L.; Liesch, J. M.; Goetz, M. A.;
Jenkins, R. G.; Nallin-Omstead, M.; Silverman, K. C.; Bills,
G. F.; Mosley, R. T.; Gibbs, J. B.; Alberts-Shonberg, G.;
Lingham, R. B. Tetrahedron 1993, 49, 5917. (b) Singh, S. B.
Tetrahedron Lett. 1993, 34, 6521.
1
3
8. Jayasuria, H.; Bills, G. F.; Cascales, C.; Zink, D. L.; Goetz,
M. A.; Jenkins, R. G.; Silverman, K. C.; Lingham, R. B.;
3
1
JCP=141.0 Hz), 23.2, 17.4, 15.7; P NMR d 30.3. Anal.
calcd for C H O P: C, 57.80; H, 8.80. Found C, 57.80;
H, 8.84.
Singh, S. B. Bioorg. Med. Chem. 1996, 6, 2081.
1
6
29
5
9
. Singh, S. B.; Liesch, J. M.; Lingham, R. B.; Goetz, M. A.;
Gibbs, J. B. J. Am. Chem. Soc. 1994, 116, 11606.
0. Patel, D. V.; Schmidt, R. J.; Biller, S. A.; Gordon, E. M.;
1
2
1
1
-(Dimethoxyphosphinyl)ethyl (E,E)-5,9,13-trimethyl-4,8,
2-tetradecatrienoate (28). A solution of acid 25 (500mg,
.89mmol), phosphonate 8 (270mL, 2.17mmol), and
Robinson, S. S.; Manne, V. J. Med. Chem. 1995, 38, 2906.
11. (a) Mu, Y.-Q.; Omer, C. A.; Gibbs, R. A. J. Am. Chem.
Soc. 1996, 118, 1817. (b) Zahn, T. J.; Ksebati, M. B.; Gibbs,
R. A. Tetrahedron Lett. 1998, 39, 3991. (c) Mechelke, M. F.;
Wiemer, D. F. Tetrahedron Lett. 1998, 39, 9609. (d) Edelstein,
R. L.; Weller, V. A.; Distefano, M. D.; Tung, J. S. J. Org.
Chem. 1998, 63, 5298. (e) Mechelke, M. F.; Wiemer, D. F. J.
Org. Chem. 1999, 64, 4821.
DCC (429 mg, 2.08 mmol) and DMAP (25 mg, 0.20
mmol) in dichloromethane (8.4 mL) was allowed to
react as described for compound 26. After 4 h, standard
work up and ®nal puri®cation by ¯ash column chro-
matography (ethyl acetate) gave compound 28 as a clear
1
2. (a) McClard, R. W.; Fujita, T. S.; Stremler, K. E.; Poulter,
1
oil (647 mg, 85%): H NMR d 5.05±5.13 (m, 3H), 4.30
C. D. J. Am. Chem. Soc. 1987, 109, 5544. (b) Pompliano, D.
L.; Rands, E.; Schaber, M. D.; Mosser, S. D.; Anthony, N. J.;
Gibbs, J. B. Biochemistry 1992, 31, 3800. (c) Gibbs, R. A.;
Krishnan, U. Tetrahedron Lett. 1994, 35, 2509. (d) Mu, Y.;
Gibbs, R. A.; Eubanks, L. M.; Poulter, C. D. J. Org. Chem.
1996, 61, 8010. (e) Kang, M. S.; Stemerick, D. M.; Zwolshen,
J. H.; Harry, B. S.; Sunkara, P. S.; Harrison, B. L. Biochem.
Biophys. Res. Comm. 1995, 217, 245. (f) Valentijn, A. R. P. M.;
van den Berg, O.; van der Marel, G. A.; Cohen, L. H.; van
Boom, J. H. Tetrahedron 1995, 51, 2099. (g) Gaon, I.; Turek,
T. C.; Weller, V. A.; Edelstein, R. L.; Singh, S. K.; Distefano,
M. D. J. Org. Chem. 1996, 61, 7738. (h) Hohl, R. J.; Lewis, K.
A.; Cermak, D. M.; Wiemer, D. F. Lipids 1998, 33, 39. (i).
Holstein, S. A.; Cermak, D. M.; Wiemer, D. F.; Lewis, K.;
Hohl, R. J. Bioorg. Med. Chem. 1998, 6, 687. (j) Cermak, D.
M.; Du, Y.; Wiemer, D. F. J. Org. Chem. 1999, 64, 388.
13. Long, S. B.; Casey, P. J.; Beese, L. S. Biochemistry 1998,
(
2
2
dt, J=12.8, 7.4 Hz, 2H), 3.76 (d, J=11.0 Hz, 6H),
.29±2.37 (m, 4H), 2.16 (dt, J=18.9, 7.4 Hz, 2H), 1.93±
.10 (m, 8H), 1.67 (d, J=0.9 Hz, 3H), 1.62 (d,
1
3
J=1.0 Hz, 3H), 1.59 (s, 6H); C NMR d 172.5, 136.4,
1
6
JCP=140.4 Hz), 23.0, 17.3, 15.6, 15.6; P NMR d 30.2.
Anal. calcd for C H O P: C, 62.96; H, 9.32. Found C,
62.71; H, 9.35.
34.6, 130.7, 124.0, 123.7, 121.8, 57.8, 52.0 (d, JCP=
.7 Hz), 39.3, 39.2, 33.9, 26.4, 26.1, 25.3, 24.7 (d,
3
1
2
1
37
5
Acknowledgements
Financial support from the Roy J. Carver Charitable
Trust and from the Leukemia & Lymphoma Society is
gratefully acknowledged.
3
1
1
1
1
1
7, 9612.
4. Corey, E. J.; Schmidt, G. Tetrahedron Lett. 1979, 5, 399.
5. Maryano, B. E.; Reitz, A. B. Chem. Rev. 1989, 89, 863.
6. Rathke, M. W.; Nowak, M. J. Org. Chem. 1985, 50, 2624.
7. (a) Bortolussi, M.; Seyden-Penne, J. Synth. Comm. 1989,
9, 2355. (b) Satoh, Y.; Tayano, T.; Hara, S.; Suzuki, A. Tet-
References and Notes
1
2
. Gelb, M. H. Science 1997, 275, 1750.
. (a) Oli, A. Biochem. Biophys. Acta 1999, 1423, c19. (b)
rahedron Lett. 1989, 30, 5153.
18. Galinis, D. L.; Wiemer, D. F. J. Org. Chem. 1993, 58,
7804.
19. (a) Desai, M. C.; Stramiello, L. M. S. Tetrahedron Lett.
1993, 34, 7685. (b) Sheehan, J. C.; Cruickshank, P. A.; Bosh-
art, G. L. J. Org. Chem. 1961, 26, 2525.
20. Kuwajima, I.; Doi, Y. Tetrahedron Lett. 1972, 1163.
21. Hohl, R. J.; Larson, R. A.; Mannickarottu, V.; Yachnin,
S. Blood 1991, 77, 1064.
Rowinsky, E. K.; Windle, J. J.; Von Ho, D. D. J. Clin. Onc.
1
999, 17, 3631.
. (a) Hancock, J. F.; Magee, A. I.; Childs, J. E.; Marshall, C.
3
J. Cell 1989, 57, 1167. (b) Schafer, W. R.; Kim, R.; Sterne, R.;
Thorner, J.; Kim, S.-H.; Rine, J. Science 1989, 245, 379.
4. Bos, J. L. Cancer Research 1989, 49, 4682.
5. Kato, K.; Cox, A. D.; Hisaka, M. M.; Graham, S. M.;
Buss, J. E.; Der, C. J. Proc. Natl. Acad. Sci. USA 1992, 89,
22. (a) Moores, S. L.; Schaber M. D.; Mosser, S. D.; Rands,
E.; O'Hara, M. B.; Garsky, V. M.; Marshall, M. S.; Pom-
pliano, D. L.; Gibbs, J. B. J. Biol. Chem. 1991, 266, 14603. (b)
Khan, S. G.; Mukhtar, H.; Agarwal, R. J. Biochem. Biophys.
Methods 1995, 30, 133.
23. Harwood, Jr., H. J. Anal. Biochem. 1995, 226, 268.
24. Meisters, A.; Wailes, P. C. Aust. J. Chem. 1960, 13, 110.
25. (a) Sato, K.; Inoue, S.; Sakamoto, T. Synthesis 1981, 796.
6
6
403.
. (a) Reiss, Y.; Goldstein, J. L.; Seabra, M. C.; Casey, P. J.;
Brown, M. S. Cell 1990, 62, 81. (b) Gibbs, J. B.; Pompliano,
D. L.; Mosser, S.; Rands, E.; Lingham, R.; Singh, S.; Scol-
nick, E.; Kohl, N.; Oli, A. J. Biol. Chem. 1993, 268, 7617. (c)
Kohl, N.; Mosser, S.; deSolms, S.; Giuliani, E.; Pompliano,
D.; Graham, S.; Smith, R.; Scolnick, E.; Oli, A.; Gibbs, J. B.

 Cermak, Diana M.
Cermak, Diana M.
 Wiemer, David F.
Wiemer, David F.
 Lewis, Kriste
Lewis, Kriste
 Hohl, Raymond J.
Hohl, Raymond J.