J . Org. Chem. 1998, 63, 8565-8569
8565
Dioxir a n e Ep oxid a tion s of
Sch em e 1. Con cer ted Ep oxid a tion by Dioxir a n es
1
,1-Disu bstitu ted Eth ylen es. P r obin g for
Ra d ica l P a th w a ys by Com p u ta tion s a n d
Exp er im en ts
J ian Liu and K. N. Houk*
Department of Chemistry and Biochemistry, University of
California, Los Angeles, California 90095
Anna Dinoi, Caterina Fusco, and Ruggero Curci*
Centro CNR “MISO”, Department of Chemistry, University
of Bari, v. Amendola 173, I-70126 Bari, Italy
Received J une 2, 1998
Sch em e 2. Step w ise Bir a d ica l Rea ction of
Alk en es w ith Dioxir a n es
In tr od u ction
The epoxidation of alkenes under extremely mild and
1
neutral conditions using dioxiranes is of particular
interest due to the value of this transformation in
2
synthesis. Dimethyldioxirane (DMD) (1) and methyl-
(
trifluoromethyl)dioxirane (TFD)3 are frequently em-
ployed to carry out epoxidations of a variety of substrates,
providing access to even highly sensitive epoxides.1
Mechanistic studies of this reaction have continued
unabated during the past decade; much experimental
evidence, including the syn-stereospecific course of diox-
1
irane epoxidations, the greater reactivity of cis alkenes
4
5
than their trans isomers, kinetic H/D isotope effects,
and the outcome of radical-clock experiments,6a all have
consinstently pointed to a concerted mechanism; a spiro
transition state for dioxirane approach to the double bond
was suggested to be consistent with these data (Scheme
1
). Following the pioneering theoretical studies by Bach
7
dation of ethene, propene, and cis- and trans-2-butene.7,8
This is well understood in terms of S 2-like attack by
N
the alkene at the dioxirane peroxide bond. FMO interac-
tions provide a guide to the mechanism: the alkene
HOMO interacts in a stabilizing fashion with the OO σ*
orbital, while the O lone pair interaction with the alkene
and co-workers, increasingly accurate computational
methods have provided support to the view of an es-
sentially concerted mechanism for the dioxirane epoxi-
(
1) For reviews, see: (a) Adam, W.; Curci, R.; Edwards, J . O. Acc.
Chem. Res. 1989, 22, 205. (b) Murray, R. W. Chem. Rev. 1989, 89, 1187.
c) Curci, R. In Advances in Oxygenated Processes; Baumstark, A. L.,
(
LUMO strongly favors a spiro geometry in the transition
Ed.; J AI: Greenwich, CT, 1990; Vol 2, Chapter I, pp 1-59. (d) Adam,
W.; Hadjiarapoglou, L. P.; Curci, R.; Mello, R. In Organic Peroxides;
Ando, W., Ed.; Wiley: New York, 1992; Chapter 4, pp 195-219. (e)
Adam, W.; Hadjiarapoglou, L. Top. Curr. Chem. 1993, 164, 4552. (f)
Curci, R.; Dinoi, A.; Rubino, M. F. Pure Appl. Chem. 1995, 67, 811.
See references therein.
state (TS).1
f,7,8
Unsymmetrical substitution of the alkene
can cause the transition state to become unsymmetrical,
but no intermediates were located on the potential
surface.8
a
(2) (a) Murray, R. W.; J eyaraman, R. J . Org. Chem. 1985, 50, 2847.
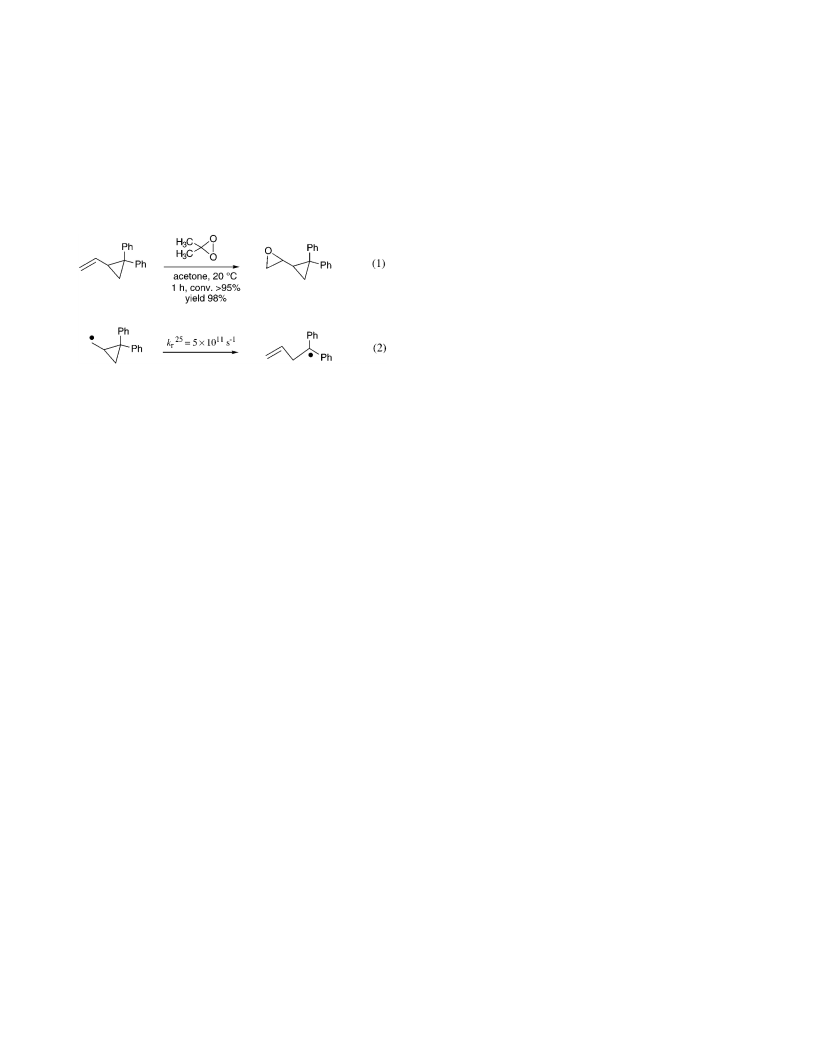
At odds with a concerted epoxidation mechanism,
(b) Cassidei, L.; Fiorentino, M.; Mello, R.; Sciacovelli, O.; Curci, R. J .
9a,b
Minisci et al.
recently envisaged a radical pair mech-
Org. Chem. 1987, 52, 699. (c) Adam, W.; Chan, Y.-Y.; Cremer, D.;
Gauss, J .; Scheutzow, D.; Schindler, M. J . Org. Chem. 1987, 52, 2800.
anism for the epoxidation of an unsymmetrically substi-
(3) (a) Mello, R.; Fiorentino, M.; Sciacovelli, O.; Curci, R. J . Org.
tuted alkene such as R-methylstyrene (2b) with DMD
Chem. 1988, 53, 3890. (b) Mello, R.; Fiorentino, M.; Fusco, C.; Curci,
R. J . Am. Chem. Soc. 1989, 111, 6749. (c) Mello, R.; Cassidei, L.;
Fiorentino, M.; Fusco, C.; H u¨ mmer, W.; J a¨ ger, V.; Curci, R. J . Am.
Chem. Soc. 1991, 113, 2205.
(Scheme 2); this conclusion was based on their detection
of a minor amount of allylic oxidation byproducts, i.e.,
-phenylpropenol, PhC(dCH )CH OH (4a ), and 2-phen-
ylpropenal, PhC(dCH )CHdO (4b), along with epoxide
2
2
2
(4) (a) Baumstark, A. L.; McCloskey, C. J . Tetrahedron Lett. 1987,
2
2
5
2
8, 3311. (b) Baumstark, A. L.; Vasquez, P. C. J . Org. Chem. 1988,
3, 3437. (c) Murray, R. W.; Shiang, D. L. J . Chem. Soc., Perkin Trans.
1990, 349.
3b. The proposed epoxidation mechanism involves alk-
ene-induced homolysis of the dioxirane O-O bond and
(5) (a) Angelis, Y.; Zhang, X.; Orfanopulos, M. Tetrahedron Lett.
1
996, 37, 5991. (b) Murray, R. W.; Shiang, D. L.; Singh, M. J . Org.
Chem. 1991, 56, 3677.
6) (a) Adam, W.; Curci, R.; D’Accolti, L.; Dinoi, A.; Fusco, C.;
Gasparrini, F.; Kluge, R.; Paredes, R.; Schulz, M.; Smerz, A. K.; Veloza,
L. A.; Weink o¨ tz, S.; Winde, R. Chem. Eur. J . 1997, 3, 105. (b) Newcomb,
M. Tetrahedron 1993, 49, 1151.
(8) (a) Houk, K. N.; Liu, J .; DeMello, N. C.; Condroski, K. R. J . Am.
Chem. Soc. 1997, 119, 10147. (b) J enson, C.; Liu, J .; Houk, K. N.;
J orgensen, W. L. J . Am. Chem. Soc. 1997, 119, 12982. See also
references therein.
(9) (a) Bravo, A.; Fontana, F.; Fronza, G.; Minisci, F. Tetrahedon
Lett. 1995, 38, 6945. (b) Bravo, A.; Fontana, F.; Fronza, G.; Minisci,
F.; Zhao, L. J . Org. Chem. 1998, 63, 254. (c) Walling, C.; Chang, Y. W.
J . Am. Chem. Soc. 1954, 76, 4878. (d) Walling, C.; Heaton, L.; Tanner,
D. D. J . Am. Chem. Soc. 1965, 87, 1715.
(
(7) (a) Bach, R. D.; Andr e´ s, J . L.; Owensby, A.; Schlegel, H. B. J .
Am. Chem. Soc. 1992, 114, 7207. (b) Bach, R. D.; Glukhovtsev, M. N.;
Gonzalez, C.; Marquez, M.; Estev e´ z, C. M.; Baboul, A. G.; Schlegel, H.
B. J . Phys. Chem. 1997, 101, 6092. See also other articles of the series.
1
0.1021/jo981050d CCC: $15.00 © 1998 American Chemical Society
Published on Web 10/27/1998

 Liu
Liu
 Houk
Houk
 Dinoi
Dinoi
 Fusco
Fusco
 Curci
Curci