10.1002/anie.201907209
Angewandte Chemie International Edition
COMMUNICATION
(Figure 2), which is in the same range as for BAL-catalyzed
benzoin condensation (free enzyme) reported in literature [5].
Assuming that the estimation of enzyme amounts within the
catalytic active cells was correct the equal conversion indicates a
very small diffusional barrier in all reaction systems. It is
noteworthy to mention, that the enantiomeric excess (ee) of the
reaction product (R)-benzoin was >99 % in all batches.
The advantages of using catalytic active whole cells in the
PE instead of isolated enzyme became evident upon repetitive
use. For repetition, we removed a defined amount of organic
solvent from the PE after a reaction time of one hour and replaced
it with the same amount of fresh solvent (see Supporting
Information for details). Again, the enantiomeric excess of (R)-
benzoin was >99 % in all cycles. However, the catalytic
performance of the isolated enzyme dropped dramatically yielding
a conversion of only ~41 % after the first and ~21 % after the fifth
recycling. Conversion also dropped when PE with catalytic active
whole-cells were used repeatedly, but considerably less than with
the free enzyme (Figure 3). The activity loss is probably due to
the repeated exposition to the organic solvent, which in the whole
cells was cushioned by the protective environment of the cell
envelope.
extending the reaction time to 24 hours, small amounts of product
were also obtained with unmodified and modified cells in pure
CPME (19.6±12.4 mmol⋅L-1 and 79.9±33.1 mmol⋅L-1, respectively).
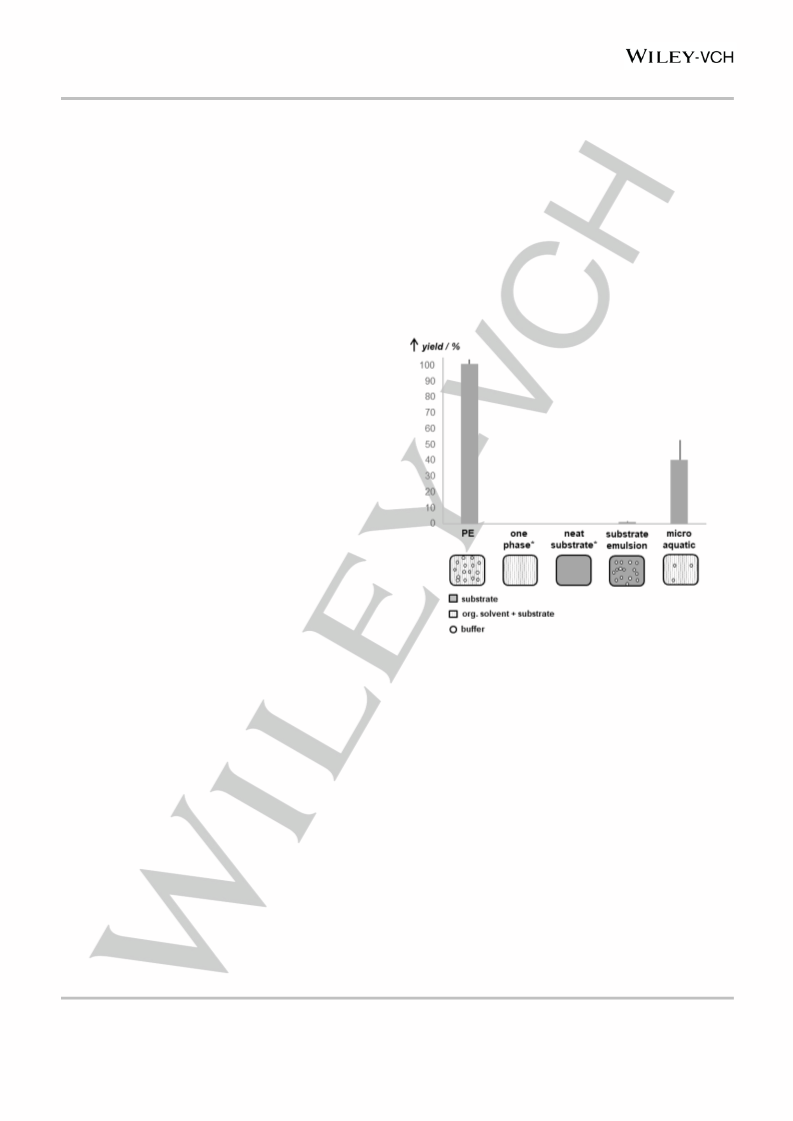
PE stabilized with modified cells (hydrophobic coating) yielded
148.6±5.7 mmol⋅L-1 (R)-benzoin within one hour (designating
100% in Figure 3), exceeding substrate emulsion
(2.1±0.3 mmol⋅L-1)
and
micro-aqueous
system
(59.0±19.7 mmol⋅L-1) by 70- and more than two-fold, respectively.
Considering that micro-aqueous systems have to date been
among the most efficient reaction systems for whole-cell
biocatalysis in non-conventional media [12], this is a formidable
performance. It might be explained by a facilitation of diffusion in
PE compared to diffusion in the only partially fluid micro-aquatic
systems.
Astonishingly, the PE stabilized with unmodified catalytic
active cells performed significantly better upon recycling than PE
stabilized with modified cells. A conversion drop of only ~13 %
occurred after the first recycling, after the fifth recycling
conversion was still at 52.0±3.7 %. This was more than 1.5-fold
better than in the PE stabilized with modified cells, regardless of
the type of silicone used for modification. Presently, we can only
speculate about the reasons. It might be that activity in the coated
systems dropped as a delayed effect of the coating procedure,
which involved solvent exposure for several hours and might have
weakened the cell envelope. Alternatively, the higher viscosity of
the PE stabilized with unmodified whole cells may have a
protective effect on the cells during the process.
In contrast, PE stabilized with modified cells, in particular
cells modified with hydrophobic silicone, showed the highest
reaction rate of all systems. It achieved maximum conversion
(77.5±5.7 %) within only 15 minutes (Figure S5 and S6,
respectively), while PE stabilized with unmodified cells reached
only ~86 % of maximum conversion in the same time. PE
stabilized with cells modified with hydrophilized silicone achieved
~92 % of the maximum conversion. The observation might be
explained by a facilitation of the access of hydrophobic molecules
to the cell due to their accumulation in the surficial silicone layer.
Finally, we evaluated the competitiveness of our bioactive
whole-cell PE through benchmarking their catalytic performance
against other promising reaction set-ups for BAL-catalyzed
benzoin condensation, namely whole-cells (unmodified or
modified with hydrophobic silicone) in pure organic solvents, in
neat substrates, and in micro-aquatic systems. In addition, we
tested a two-phase system consisting of an aqueous solution of
(unmodified) whole cells dispersed in pure substrate (substrate
emulsion). Like in the previous benchmarking of PE-stabilized
systems, we made sure that comparable amounts of enzyme
(cells) were present in all set-ups.
Figure 3. (R)-benzoin yields from catalytic active cells after one hour
reaction in various set-ups: Pickering emulsion stabilized by cells modified with
hydrophobic silicone (PE); pure CPME (one phase); undiluted benzaldehyde
(neat substrate); small amounts of buffer in benzaldehyde (substrate emulsion);
cell suspension in CPME with addition of a very small amount of buffer (micro-
aquatic). Error bars represent standard deviation of results from three
independent batches. Benzaldehyde concentration in PE, one phase and mirco
aquatic was 400 mmol L-1. Experiments were repeated with an equal amount of
unmodified E. coli cells* in one phase and neat substrate systems. Water activity
of either 0.37, or 0.79 was adjusted for both cells and benzaldehyde in neat
substrate systems. PE and substrate emulsions were formed at a water:sovlent
ratio of 1:5 (v/v). Water:solvent ratio in the micro aquatic was 1:111 (v/v).
In conclusion, Pickering emulsions using E. coli cells for
stabilization and simultaneous catalysis provided both efficient
and stable systems for biocatalyzed carboligation, overcoming
current limits in the use of whole-cell biocatalysts for synthesis.
Omission of extra particles and waiver of enzyme purification
makes the set-up considerably simpler than biocatalytic PE
currently in use. Formation of viscous multi-emulsions can, if
desired, easily be overcome through non-covalent cell-surface
coating with silicone. As a side effect such a modification can
further improve catalytic performance. We expect these findings
As illustrated in Figure 3, within a reaction time of one hour,
we observed measurable amounts of (R)-benzoin only in PE,
substrate emulsion, and the micro-aquatic system. Upon
This article is protected by copyright. All rights reserved.

 R?llig, Robert
R?llig, Robert
 Plikat, Christoph
Plikat, Christoph
 Ansorge-Schumacher, Marion B.
Ansorge-Schumacher, Marion B.